Poster Session C
Rheumatoid arthritis (RA)
Session: (2141–2176) RA – Treatment Poster III
2143: Efficacy of Filgotinib in Patients with Rheumatoid Arthritis: Week 156 Results from a Long-term Extension Study
Tuesday, November 14, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall

MAYA BUCH, MD, PhD
UNIVERSITY OF MANCHESTER
MANCHESTER, United KingdomDisclosure information not submitted.
Abstract Poster Presenter(s)
Maya Buch1, Daniel Aletaha2, Roberto F. Caporali3, Bernard G. Combe4, Hendrik Schulze-Koops5, Jacques-Eric Gottenberg6, Yoshiya Tanaka7, Ricardo Blanco8, Tsutomu Takeuchi9, Edmund V. Ekoka Omoruyi10, Katrien Van Beneden11, Vijay Rajendran12, Chris Watson13, Francesco De Leonardis14 and Paul Emery15, 1University of Manchester and NIHR Manchester Biomedical Research Centre, Manchester, United Kingdom, 2Division of Rheumatology, Department of Medicine III, Medical University of Vienna, Vienna, Austria, Wien, Austria, 3Department of Clinical Sciences and Community Health, University of Milan, and Department of Rheumatology and Medical Sciences, ASST Gaetano Pini-CTO, Milano, Italy, 4Department of Rheumatology, Montpellier University, Montpellier, France, 5Division of Rheumatology and Clinical Immunology, Department of Internal Medicine IV, Ludwig-Maximilians–University Munich, Munich, Germany, 6Rheumatology Department, Strasbourg University Hospital, Strasbourg, France, 7University of Occupational and Environmental Health, Kitakyushu, Japan, 8Hospital Universitario Marqués de Valdecilla, IDIVAL, Santander, Spain, 9Keio University School of Medicine and Saitama Medical University, Tokyo, Japan, 10Biostatistics, Galapagos NV, Mechelen, Belgium, 11Medical Safety, Galapagos NV, Mechelen, Belgium, 12Clinical Research, Galapagos NV, Mechelen, Belgium, 13Medical Affairs, Galapagos Biotech Ltd., Cambridge, United Kingdom, 14Medical Affairs, Galapagos GmbH, Basel, Switzerland, 15Leeds Institute of Rheumatic and Musculoskeletal Medicine, University of Leeds, and NIHR Leeds Biomedical Research Centre, Leeds Teaching Hospitals NHS Trust, Leeds, United Kingdom
Background/Purpose: In the treatment of RA, JAK inhibitors are a valuable option to meet remission or low disease activity (LDA) treatment targets following an inadequate response (IR) or intolerance to ≥ 1 conventional synthetic disease-modifying antirheumatic drug (DMARD). Filgotinib (FIL) is a JAK1 preferential inhibitor available in two doses for the treatment of moderate to severe RA. The objective of this analysis was to evaluate long-term efficacy of two doses of FIL in clinically relevant pt populations. Response rates for Boolean remission 2.0 were reported as an exploratory objective.
Methods: In this interim analysis, efficacy (nonresponder imputation) of FIL 200 mg (FIL200) and 100 mg (FIL100) was assessed from long-term extension (LTE) baseline (BL) to Week (W) 156 in patients (pts) with an IR to methotrexate (MTX-IR) and biologics (bDMARD-IR), enrolled from FINCH 1 (NCT02889796) and 2 (NCT02873936) parent studies, respectively, receiving ≥ 1 FIL dose in FINCH 4 (NCT03025308).
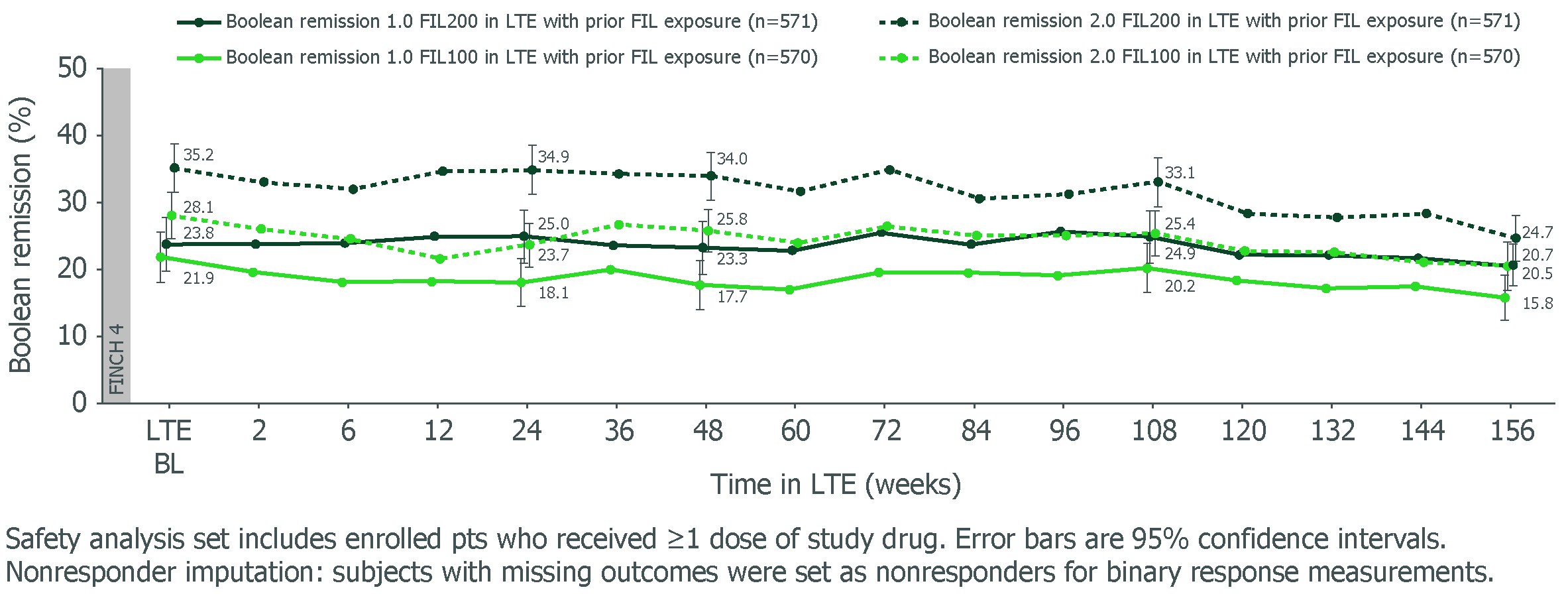
Results: Study design, BL characteristics and W48 outcomes for MTX-IR1 and bDMARD-IR2 pts were reported previously. For MTX-IR and bDMARD-IR pts who received FIL200 or FIL100 in the parent study, W156 remission rates using Boolean 1.0 criteria were 20.5% and 15.8%, and 18.2% and 8.9%, respectively. Adopting the Boolean 2.0 criteria slightly increased remission rates for FIL200 and FIL100 at W156: +4.2% and +4.9% for MTX-IR pts, and +1.5% and +2.4% for bDMARD-IR pts, respectively. For pts rerandomized to FIL on entering the LTE, Boolean 2.0 criteria also increased remission rates vs Boolean 1.0. Both MTX-IR (Figure) and bDMARD-IR pts maintained long-term Boolean remission through W156 with FIL200 and FIL100, irrespective of prior FIL. Results for Disease Activity Score in 28 joints using C-reactive protein (DAS28-CRP) < 2.6, Clinical Disease Activity Index (CDAI) ≤ 2.8 and Simplified Disease Activity Index (SDAI) ≤ 3.3, and mean change from parent study BL in Health Assessment Questionnaire–Disability Index (HAQ-DI) and pain are shown (Table). Similar trends in efficacy were seen for LDA and ACR response criteria.
Conclusion: In FINCH 4, both FIL200 and FIL100 showed sustained efficacy up to W156 in clinically relevant pt populations. Boolean 2.0 criteria classified more pts in remission, in line with the range reported in the validation study.3
References:
1. Combe B, et al. Arthritis Rheumatol 2021;73(Suppl 9):POS1697
2. Buch M, et al. Arthritis Rheumatol 2021;73(Suppl 9):POS1696
3. Studenic P, et al. Arthritis Rheumatol 2023;82:74–80
.jpg)
M. Buch: AbbVie, 2, 6, 12, All paid to host institution, Boehringer Ingelheim, 2, 6, 12, Paid to host institution, Galapagos, 2, 6, 12, Paid to host institution, Gilead, 2, 5, 6, 12, Paid to host institution, Lilly, 2, 6, 12, All paid to host institution, National Insitute for Health and Care Research (NIHR), 3, 12, Maya H Buch is a National Institute for Health and Care Research (NIHR) Senior Investigator. The views expressed are those of the authors and not nece, Pfizer, 2, 12, Paid to host institution; D. Aletaha: AbbVie, 2, 5, 6, Amgen, 2, 5, 6, Janssen, 2, 6, Lilly, 2, 5, 6, Merck, 2, 6, Novartis, 2, 5, 6, Pfizer, 2, 6, Roche, 2, 5, 6, Sandoz, 2, 6, Sanofi, 5, Sobi, 5; R. Caporali: AbbVie, 2, 6, Amgen, 2, 6, BMS, 2, 6, Celltrion, 2, 6, Fresenius Kabi, 2, Galapagos, 2, 6, Janssen, 2, 6, Lilly, 2, 6, MSD, 2, 6, Novartis, 2, 6, Pfizer, 2, 6, Roche, 2, Sandoz, 2, 6, UCB, 2, 6; B. Combe: AbbVie, 2, 6, BMS, 6, Celltrion, 2, Eli Lilly, 1, 2, 6, Galapagos, 2, 6, Gilead, 2, Janssen, 2, 6, MSD, 6, Nordic Pharma, 5, Novartis, 1, Pfizer, 6, Roche-Chugai, 2, 6; H. Schulze-Koops: AbbVie, 2, 6, Eli Lilly, 2, 6, Galapagos, 2, 6, Pfizer, 2, 6; J. Gottenberg: AbbVie, 2, BMS, 2, 5, Galapagos, 2, Gilead, 2, Lilly, 2, MSD, 2, Novartis, 2, Pfizer, 2, 5; Y. Tanaka: AbbVie, 6, AstraZeneca, 6, BMS, 6, Boehringer-Ingelheim, 6, Chugai, 5, 6, Eisai, 5, 6, Eli Lilly, 6, Gilead, 6, GSK, 6, Mitsubishi-Tanabe, 5, Pfizer, 6, Taiho, 6, Taisho, 5, 6; R. Blanco: AbbVie, 5, 6, Amgen, 6, AstraZeneca, 2, BMS, 6, Eli Lilly, 6, Galapagos, 2, 6, Janssen, 2, 6, MSD, 6, Novartis, 2, 6, Pfizer, 2, 6, Roche, 5, 6, Sanofi, 6; T. Takeuchi: AbbVie, 2, 5, 6, AYUMI, 5, Bristol-Myers Squibb, 6, Chugai, 2, 5, 6, Daiichi Sankyo, 5, Eisai, 5, 6, Eli Lilly Japan, 2, 6, Gilead, 2, 6, Janssen, 6, Mitsubishi-Tanabe, 2, 5, 6, ONO, 5, Pfizer Japan, 6, Taiho, 2; E. Ekoka Omoruyi: Galapagos, 2, Janssen, 2, UCB, 11; K. Van Beneden: Galapagos, 3, 11; V. Rajendran: Galapagos, 3, 11; C. Watson: Galapagos, 3, 11; F. De Leonardis: Galapagos, 3; P. Emery: Boehringer Ingelheim, 2, Eli Lilly, 2, Novartis, 2.
Background/Purpose: In the treatment of RA, JAK inhibitors are a valuable option to meet remission or low disease activity (LDA) treatment targets following an inadequate response (IR) or intolerance to ≥ 1 conventional synthetic disease-modifying antirheumatic drug (DMARD). Filgotinib (FIL) is a JAK1 preferential inhibitor available in two doses for the treatment of moderate to severe RA. The objective of this analysis was to evaluate long-term efficacy of two doses of FIL in clinically relevant pt populations. Response rates for Boolean remission 2.0 were reported as an exploratory objective.
Methods: In this interim analysis, efficacy (nonresponder imputation) of FIL 200 mg (FIL200) and 100 mg (FIL100) was assessed from long-term extension (LTE) baseline (BL) to Week (W) 156 in patients (pts) with an IR to methotrexate (MTX-IR) and biologics (bDMARD-IR), enrolled from FINCH 1 (NCT02889796) and 2 (NCT02873936) parent studies, respectively, receiving ≥ 1 FIL dose in FINCH 4 (NCT03025308).
Results: Study design, BL characteristics and W48 outcomes for MTX-IR1 and bDMARD-IR2 pts were reported previously. For MTX-IR and bDMARD-IR pts who received FIL200 or FIL100 in the parent study, W156 remission rates using Boolean 1.0 criteria were 20.5% and 15.8%, and 18.2% and 8.9%, respectively. Adopting the Boolean 2.0 criteria slightly increased remission rates for FIL200 and FIL100 at W156: +4.2% and +4.9% for MTX-IR pts, and +1.5% and +2.4% for bDMARD-IR pts, respectively. For pts rerandomized to FIL on entering the LTE, Boolean 2.0 criteria also increased remission rates vs Boolean 1.0. Both MTX-IR (Figure) and bDMARD-IR pts maintained long-term Boolean remission through W156 with FIL200 and FIL100, irrespective of prior FIL. Results for Disease Activity Score in 28 joints using C-reactive protein (DAS28-CRP) < 2.6, Clinical Disease Activity Index (CDAI) ≤ 2.8 and Simplified Disease Activity Index (SDAI) ≤ 3.3, and mean change from parent study BL in Health Assessment Questionnaire–Disability Index (HAQ-DI) and pain are shown (Table). Similar trends in efficacy were seen for LDA and ACR response criteria.
Conclusion: In FINCH 4, both FIL200 and FIL100 showed sustained efficacy up to W156 in clinically relevant pt populations. Boolean 2.0 criteria classified more pts in remission, in line with the range reported in the validation study.3
References:
1. Combe B, et al. Arthritis Rheumatol 2021;73(Suppl 9):POS1697
2. Buch M, et al. Arthritis Rheumatol 2021;73(Suppl 9):POS1696
3. Studenic P, et al. Arthritis Rheumatol 2023;82:74–80

Figure. Boolean remission 1.0 and 2.0 for MTX-IR pts in FINCH 4 LTE through W156 (safety analysis set, nonresponder imputation)
.jpg)
Table. Efficacy in MTX-IR and bDMARD-IR pts through W156 in FINCH 4 (safety analysis set; nonresponder imputation)
M. Buch: AbbVie, 2, 6, 12, All paid to host institution, Boehringer Ingelheim, 2, 6, 12, Paid to host institution, Galapagos, 2, 6, 12, Paid to host institution, Gilead, 2, 5, 6, 12, Paid to host institution, Lilly, 2, 6, 12, All paid to host institution, National Insitute for Health and Care Research (NIHR), 3, 12, Maya H Buch is a National Institute for Health and Care Research (NIHR) Senior Investigator. The views expressed are those of the authors and not nece, Pfizer, 2, 12, Paid to host institution; D. Aletaha: AbbVie, 2, 5, 6, Amgen, 2, 5, 6, Janssen, 2, 6, Lilly, 2, 5, 6, Merck, 2, 6, Novartis, 2, 5, 6, Pfizer, 2, 6, Roche, 2, 5, 6, Sandoz, 2, 6, Sanofi, 5, Sobi, 5; R. Caporali: AbbVie, 2, 6, Amgen, 2, 6, BMS, 2, 6, Celltrion, 2, 6, Fresenius Kabi, 2, Galapagos, 2, 6, Janssen, 2, 6, Lilly, 2, 6, MSD, 2, 6, Novartis, 2, 6, Pfizer, 2, 6, Roche, 2, Sandoz, 2, 6, UCB, 2, 6; B. Combe: AbbVie, 2, 6, BMS, 6, Celltrion, 2, Eli Lilly, 1, 2, 6, Galapagos, 2, 6, Gilead, 2, Janssen, 2, 6, MSD, 6, Nordic Pharma, 5, Novartis, 1, Pfizer, 6, Roche-Chugai, 2, 6; H. Schulze-Koops: AbbVie, 2, 6, Eli Lilly, 2, 6, Galapagos, 2, 6, Pfizer, 2, 6; J. Gottenberg: AbbVie, 2, BMS, 2, 5, Galapagos, 2, Gilead, 2, Lilly, 2, MSD, 2, Novartis, 2, Pfizer, 2, 5; Y. Tanaka: AbbVie, 6, AstraZeneca, 6, BMS, 6, Boehringer-Ingelheim, 6, Chugai, 5, 6, Eisai, 5, 6, Eli Lilly, 6, Gilead, 6, GSK, 6, Mitsubishi-Tanabe, 5, Pfizer, 6, Taiho, 6, Taisho, 5, 6; R. Blanco: AbbVie, 5, 6, Amgen, 6, AstraZeneca, 2, BMS, 6, Eli Lilly, 6, Galapagos, 2, 6, Janssen, 2, 6, MSD, 6, Novartis, 2, 6, Pfizer, 2, 6, Roche, 5, 6, Sanofi, 6; T. Takeuchi: AbbVie, 2, 5, 6, AYUMI, 5, Bristol-Myers Squibb, 6, Chugai, 2, 5, 6, Daiichi Sankyo, 5, Eisai, 5, 6, Eli Lilly Japan, 2, 6, Gilead, 2, 6, Janssen, 6, Mitsubishi-Tanabe, 2, 5, 6, ONO, 5, Pfizer Japan, 6, Taiho, 2; E. Ekoka Omoruyi: Galapagos, 2, Janssen, 2, UCB, 11; K. Van Beneden: Galapagos, 3, 11; V. Rajendran: Galapagos, 3, 11; C. Watson: Galapagos, 3, 11; F. De Leonardis: Galapagos, 3; P. Emery: Boehringer Ingelheim, 2, Eli Lilly, 2, Novartis, 2.



