Poster Session C
Rheumatoid arthritis (RA)
Session: (2095–2140) RA – Diagnosis, Manifestations, and Outcomes Poster III
2096: Association of Left Ventricular Mass with Interleukin-17 in Rheumatoid Arthritis Patients Without Clinical Heart Failure
Tuesday, November 14, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- EP
Elizabeth Park, MD, MSc (she/her/hers)
Columbia University Medical Center
New York, NY, United StatesDisclosure(s): Pfizer: Grant/Research Support (Terminated, July 3, 2023)
Abstract Poster Presenter(s)
Elizabeth Park1, Kazato Ito2, Christopher Depender1, Jon Giles3 and Joan Bathon3, 1Columbia University Irving Medical Center, New York, NY, 2Columbia University Medical Center, New York, NY, 3Columbia University, New York, NY
Background/Purpose: Elevated left ventricular (LV) mass (LVM) is an important precursor to clinical heart failure (HF) in the general population. In fact, rheumatoid arthritis (RA) patients without HF demonstrate higher LVM than non-RA patients without HF. However, few studies have established direct associations between LVM and inflammatory cytokines in RA patients without clinical HF. Interleukin-17 (IL-17) is an important contributor to LV structural abnormalities in RA, given its dual pathologic roles in myocardial fibrosis and articular cartilage breakdown. Here we present previously unreported associations between IL-17, obtained from a large inflammatory cytokine panel, and LVM in a RA cohort without clinical HF.
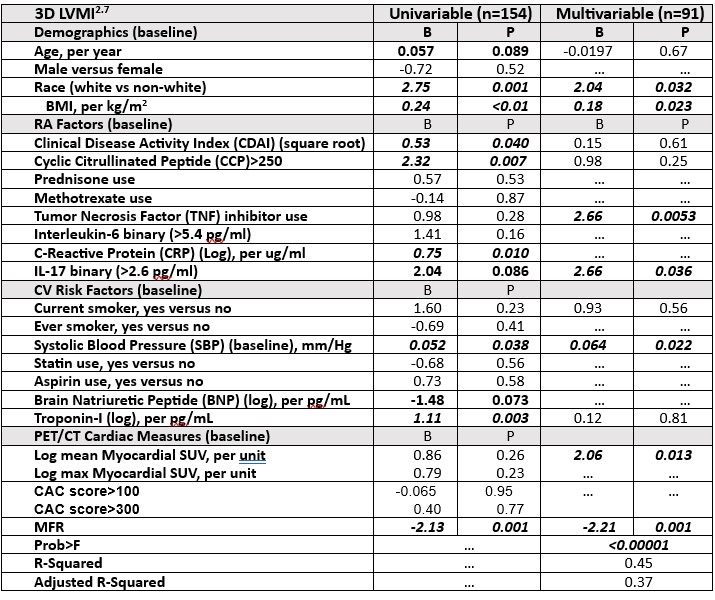
Methods: RA patients (n=158) without clinical HF underwent RA and cardiovascular (CV) clinical assessment, including myocardial inflammation and myocardial flow reserve (MFR) measured on 18F-Fluorodeoxyglucosecardiac positron emission tomography–computed tomography (FDG-PET/CT), as well as transthoracic echocardiography (TTE) at baseline. A subset (n=60) returned for follow-up TTE 3-6 years later. Real-time 3-dimensional (3D) TTE was utilized to calculate LVM (indexed to height (m2.7); LVMI). Baseline plasma from all RA patients was assayed using the Rule Based Medicine (RBM)/Inflammation MAP v 1.1, a 54-marker Discovery Panel, assaying a broad group of inflammatory proteins. Multivariable (MV) regression models with LVMI as the outcome were constructed, adjusting for biologically plausible confounders identified from univariable regressions.
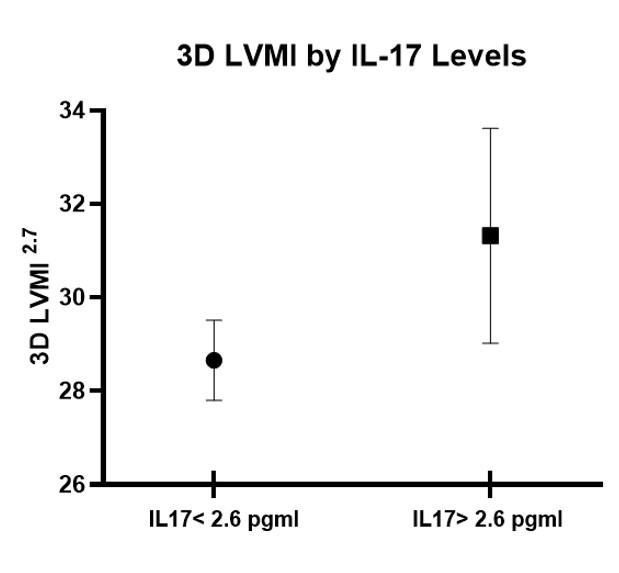
Results: The baseline cohort was predominantly middle aged, female, with moderate RA disease activity, and multiple CV risk factors. LVMI did not change significantly from baseline to follow-up. Up to 14% of the baseline cohort had IL-17 levels >2.6 pg/ml (lower limit of quantitation). In a MV model adjusted for demographics including BMI, RA disease activity, seropositivity, TNF inhibitor use, blood pressure, smoking, troponin levels, myocardial inflammation and MFR, baseline IL-17 levels >2.6 were associated with a higher baseline LVMI (Table 1; MV model). Figure 1 demonstrates adjusted LVMI stratified by IL-17 levels (based on aforementioned MV regression model). IL-17 levels were not significantly associated in univariable and MV models with myocardial measures such as MFR and myocardial inflammation (mean standardized uptake value; SUV), as well as baseline and follow-up diastolic dysfunction, and baseline/annual rate of change in ejection fraction (Tables not shown).
Conclusion: In an RA cohort without clinical HF, higher IL-17 levels were associated with higher LVMI, while adjusting for important RA and CV covariates. Whether anti IL-17 therapy could ameliorate these LV mass abnormalities, needs to be explored in future clinical studies.
E. Park: Pfizer, 5; K. Ito: None; C. Depender: None; J. Giles: AbbVie, 2, Eli Lilly, 2, Gilead, 2, Novartis, 2, Pfizer, 2; J. Bathon: None.
Background/Purpose: Elevated left ventricular (LV) mass (LVM) is an important precursor to clinical heart failure (HF) in the general population. In fact, rheumatoid arthritis (RA) patients without HF demonstrate higher LVM than non-RA patients without HF. However, few studies have established direct associations between LVM and inflammatory cytokines in RA patients without clinical HF. Interleukin-17 (IL-17) is an important contributor to LV structural abnormalities in RA, given its dual pathologic roles in myocardial fibrosis and articular cartilage breakdown. Here we present previously unreported associations between IL-17, obtained from a large inflammatory cytokine panel, and LVM in a RA cohort without clinical HF.
Methods: RA patients (n=158) without clinical HF underwent RA and cardiovascular (CV) clinical assessment, including myocardial inflammation and myocardial flow reserve (MFR) measured on 18F-Fluorodeoxyglucosecardiac positron emission tomography–computed tomography (FDG-PET/CT), as well as transthoracic echocardiography (TTE) at baseline. A subset (n=60) returned for follow-up TTE 3-6 years later. Real-time 3-dimensional (3D) TTE was utilized to calculate LVM (indexed to height (m2.7); LVMI). Baseline plasma from all RA patients was assayed using the Rule Based Medicine (RBM)/Inflammation MAP v 1.1, a 54-marker Discovery Panel, assaying a broad group of inflammatory proteins. Multivariable (MV) regression models with LVMI as the outcome were constructed, adjusting for biologically plausible confounders identified from univariable regressions.
Results: The baseline cohort was predominantly middle aged, female, with moderate RA disease activity, and multiple CV risk factors. LVMI did not change significantly from baseline to follow-up. Up to 14% of the baseline cohort had IL-17 levels >2.6 pg/ml (lower limit of quantitation). In a MV model adjusted for demographics including BMI, RA disease activity, seropositivity, TNF inhibitor use, blood pressure, smoking, troponin levels, myocardial inflammation and MFR, baseline IL-17 levels >2.6 were associated with a higher baseline LVMI (Table 1; MV model). Figure 1 demonstrates adjusted LVMI stratified by IL-17 levels (based on aforementioned MV regression model). IL-17 levels were not significantly associated in univariable and MV models with myocardial measures such as MFR and myocardial inflammation (mean standardized uptake value; SUV), as well as baseline and follow-up diastolic dysfunction, and baseline/annual rate of change in ejection fraction (Tables not shown).
Conclusion: In an RA cohort without clinical HF, higher IL-17 levels were associated with higher LVMI, while adjusting for important RA and CV covariates. Whether anti IL-17 therapy could ameliorate these LV mass abnormalities, needs to be explored in future clinical studies.

Table 1. Univariable and Multivariable Regression Table for LVMI (2.7)

Figure 1. 3D LVMI by IL-17 levels, adjusted for RA and CV covariates
E. Park: Pfizer, 5; K. Ito: None; C. Depender: None; J. Giles: AbbVie, 2, Eli Lilly, 2, Gilead, 2, Novartis, 2, Pfizer, 2; J. Bathon: None.



