Poster Session C
Epidemiology, health policy and outcomes
Session: (1840–1861) Health Services Research Poster III
1841: Clinical Trial Perceptions Among Male Patients with Systemic Lupus Erythematosus (SLE): Georgians Organized Against Lupus (GOAL) Cohort
Tuesday, November 14, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- HM
Hilton Mozee, MPH
Emory University School of Medicine
Atlanta, GA, United StatesDisclosure information not submitted.
Abstract Poster Presenter(s)
Hilton Mozee1, Charmayne Dunlop-Thomas1, Gaobin Bao1, Kim Schofield2, Jessica Williams3 and S. Sam Lim1, 1Emory University, Atlanta, GA, 2Georgia State Rep. HD 63, Atlanta, GA, 3Emory University School of Medicine, Atlanta, GA
Background/Purpose: Minority patients are disproportionately affected by SLE. Yet, they are underrepresented in SLE clinical trials compared to White patients. Specifically, patients that identify as male and Black are significantly understudied. We explored racial and gender differences in clinical trial perceptions among a cohort of predominantly Black patients with SLE in Atlanta, Georgia.
Methods: Data from the Georgians Organized Against Lupus (GOAL) cohort were used for this analysis. The GOAL cohort consists of validated SLE patients living in Atlanta, Georgia. Since 2012, SLE patients have been surveyed annually regarding demographics, SLE natural history, treatment, healthcare utilization, and psychosocial factors. For this study, only male patients were assessed. The 2022 GOAL survey included ad-hoc questions that assessed clinical trial knowledge, willingness to participate, and hesitancy in clinical trial enrollment. The Systemic Lupus Activity Questionnaire (SLAQ), Self-administered Brief Index of Lupus Damage (SA-BILD), and self-efficacy, depression, and perceived stress measures were included in this study. Descriptive analysis was used for comparisons.
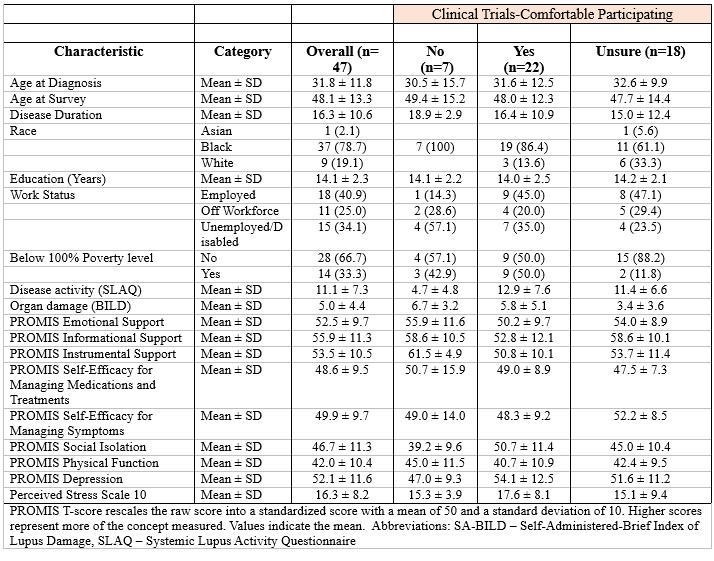
Results: A total of 47 males responded to the 2022 GOAL Survey; the majority were Black (79% Black, 19% White, 2% Asian) with a mean age 32 years (SD 11.8), mean education of 14 years (SD 2.3), and majority unemployed (59%). The mean disease duration was 16 years (SD 10.6), mean disease activity score was 11 (SD 7.3), and mean organ damage score was 5 (SD 4.4). We found that 47% of male respondents (n=19 Black, n=3 White) were willing to participate in lupus clinical trials. In contrast, about 30% were unwilling to participate, and 38% were unsure if they would participate in a clinical trial. Male willingness to participate was associated with higher disease activity, living in poverty (50%), less instrumental support), and more social isolation, as compared to those unwilling or unsure (Table). Education, marital status, work status, insurance type, physical function, depression, and perceived stress were not associated with willingness to participate in clinical trials among male respondents.
Conclusion: Among a predominantly Black cohort of males with SLE, we found that respondents with more disease activity, fewer resources, and social support appeared more willing to participate in clinical trials. Further efforts are necessary to educate and provide social support to individuals with SLE that are unsure or resistant to clinical trial participation, especially among underrepresented groups, including males. Similar studies have found that clinical trial education, recruitment through lupus support groups, and diversity of clinical trial staff are potential ways to increase trial participation. Further studies of males with SLE are necessary to validate our study findings and provide more knowledge on factors associated with clinical trial hesitancy among this population.
H. Mozee: None; C. Dunlop-Thomas: None; G. Bao: None; K. Schofield: None; J. Williams: None; S. Lim: None.
Background/Purpose: Minority patients are disproportionately affected by SLE. Yet, they are underrepresented in SLE clinical trials compared to White patients. Specifically, patients that identify as male and Black are significantly understudied. We explored racial and gender differences in clinical trial perceptions among a cohort of predominantly Black patients with SLE in Atlanta, Georgia.
Methods: Data from the Georgians Organized Against Lupus (GOAL) cohort were used for this analysis. The GOAL cohort consists of validated SLE patients living in Atlanta, Georgia. Since 2012, SLE patients have been surveyed annually regarding demographics, SLE natural history, treatment, healthcare utilization, and psychosocial factors. For this study, only male patients were assessed. The 2022 GOAL survey included ad-hoc questions that assessed clinical trial knowledge, willingness to participate, and hesitancy in clinical trial enrollment. The Systemic Lupus Activity Questionnaire (SLAQ), Self-administered Brief Index of Lupus Damage (SA-BILD), and self-efficacy, depression, and perceived stress measures were included in this study. Descriptive analysis was used for comparisons.
Results: A total of 47 males responded to the 2022 GOAL Survey; the majority were Black (79% Black, 19% White, 2% Asian) with a mean age 32 years (SD 11.8), mean education of 14 years (SD 2.3), and majority unemployed (59%). The mean disease duration was 16 years (SD 10.6), mean disease activity score was 11 (SD 7.3), and mean organ damage score was 5 (SD 4.4). We found that 47% of male respondents (n=19 Black, n=3 White) were willing to participate in lupus clinical trials. In contrast, about 30% were unwilling to participate, and 38% were unsure if they would participate in a clinical trial. Male willingness to participate was associated with higher disease activity, living in poverty (50%), less instrumental support), and more social isolation, as compared to those unwilling or unsure (Table). Education, marital status, work status, insurance type, physical function, depression, and perceived stress were not associated with willingness to participate in clinical trials among male respondents.
Conclusion: Among a predominantly Black cohort of males with SLE, we found that respondents with more disease activity, fewer resources, and social support appeared more willing to participate in clinical trials. Further efforts are necessary to educate and provide social support to individuals with SLE that are unsure or resistant to clinical trial participation, especially among underrepresented groups, including males. Similar studies have found that clinical trial education, recruitment through lupus support groups, and diversity of clinical trial staff are potential ways to increase trial participation. Further studies of males with SLE are necessary to validate our study findings and provide more knowledge on factors associated with clinical trial hesitancy among this population.

H. Mozee: None; C. Dunlop-Thomas: None; G. Bao: None; K. Schofield: None; J. Williams: None; S. Lim: None.



