Poster Session C
Systemic lupus erythematosus (SLE)
Session: (2257–2325) SLE – Diagnosis, Manifestations, & Outcomes Poster III
2258: Brain Activity Patterns and Behavioural Performance in SLE Patients During a Spatial Working Memory and Sustained Attention Task
Tuesday, November 14, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
.png)
Zahi Touma, MD, PhD
University of Toronto
Toronto, ON, CanadaDisclosure(s): No financial relationships with ineligible companies to disclose
Abstract Poster Presenter(s)
Michelle Barraclough1, Shane McKie2, Alex Kafkas2, Ben Parker3, Juan Pablo Diaz Martinez4, Andrea Knight5, Kathleen Bingham1, Jiandong Su1, Mahta Kakvan6, Carolina Munoz-Grajales7, Carmela Tartaglia8, Lesley Ruttan9, Joan Wither1, Dennisse Bonilla1, Daniela Montaldi2, Rebecca Elliott2, Patti Katz10, Dorcas Beaton11, Robin Green9, Ian Bruce2 and Zahi Touma12, 1University Health Network, Toronto, ON, Canada, 2University of Manchester, Manchester, United Kingdom, 3Manchester University Hospitals NHS Foundation Trust, Manchester, United Kingdom, 4Schroeder Arthritis Institute, University Health Network, Toronto, ON, Canada, 5The Hospital for Sick Children, Toronto, ON, Canada, 6University Health Network, University of Toronto, Toronto, ON, Canada, 7UHN/TWH, Toronto, ON, Canada, 8Krembil Brain Institute, University Health Network, University of Toronto, Toronto, ON, Canada, 9University Health Network, Toronto Rehabilitation Institute, Toronto, ON, Canada, 10University of California San Francisco, San Rafael, CA, 11Institute for Work & Health, Toronto, ON, Canada, 12University of Toronto, Toronto, ON, Canada
Background/Purpose: Cognitive impairment (CI) is a significant problem in SLE but there is a disconnect between objective and subjective CI. This makes it difficult to fully understand and treat CI. Functional magnetic resonance imaging (fMRI) has shown compensatory brain mechanisms may help to maintain cognitive function but potentially cause fatigue leading to subjective CI. This in part could explain the objective/subjective CI disconnect but ways to measure this are needed. We devised a new fMRI paradigm examining attention, working memory, learning and long-term associative memory. The aim was to assess this new task by examining whether attentional performance and working memory decreases over repeated trials in an SLE cohort using our preliminary data.
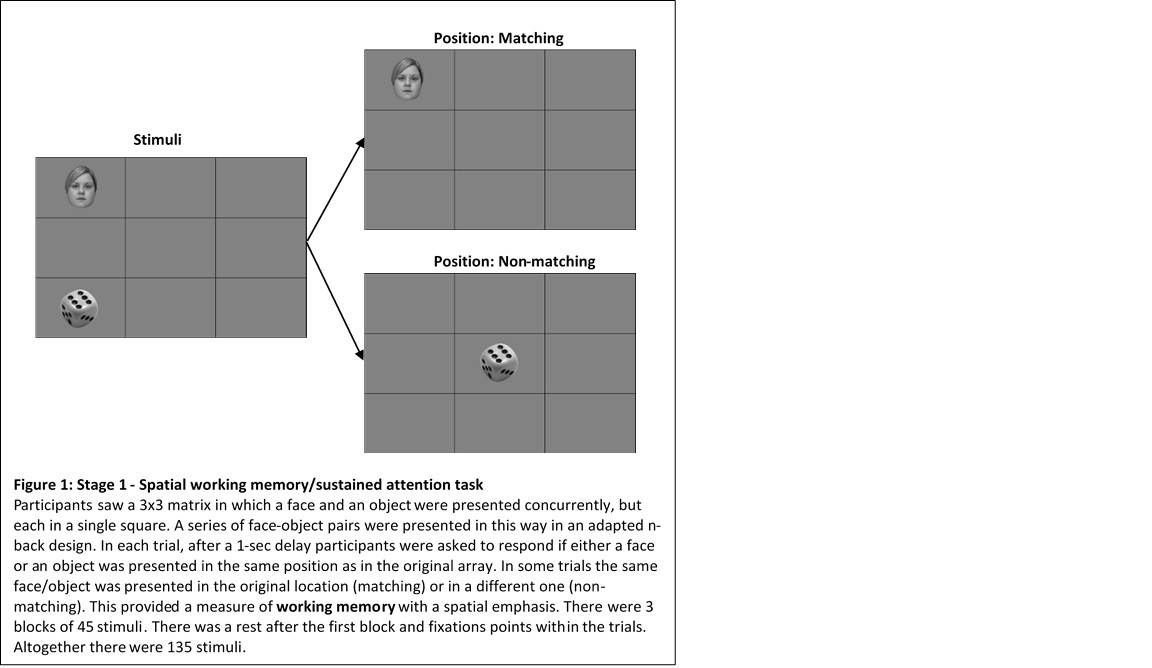
Methods: Recruited adult SLE patients met EULAR/ACR criteria. Demographic, clinical and psychiatric data and patient reported outcome measures were collected. Cognitive function was assessed using a modified version of the ACR Neuropsychological Battery. MR scans included two structural scans and two functional MRI scans done during stage 1 and 2 of a cognitive task. Stage 1 (working memory and attention, Figure 1) was split into three trial blocks of equal length and performance over the trials was examined using mean number correct (maximum of 45 per trial), mean response time per trial (ms) and fMRI blood-oxygen-level-dependent (BOLD) response signal to stage 1 of the task. Differences between the trial performances were examined using repeated measures ANOVA and the fMRI data was modelled using SPM12. fMRI interference was done at a cluster extent correction threshold of family-wise error-corrected (pFWEc)< 0.05.
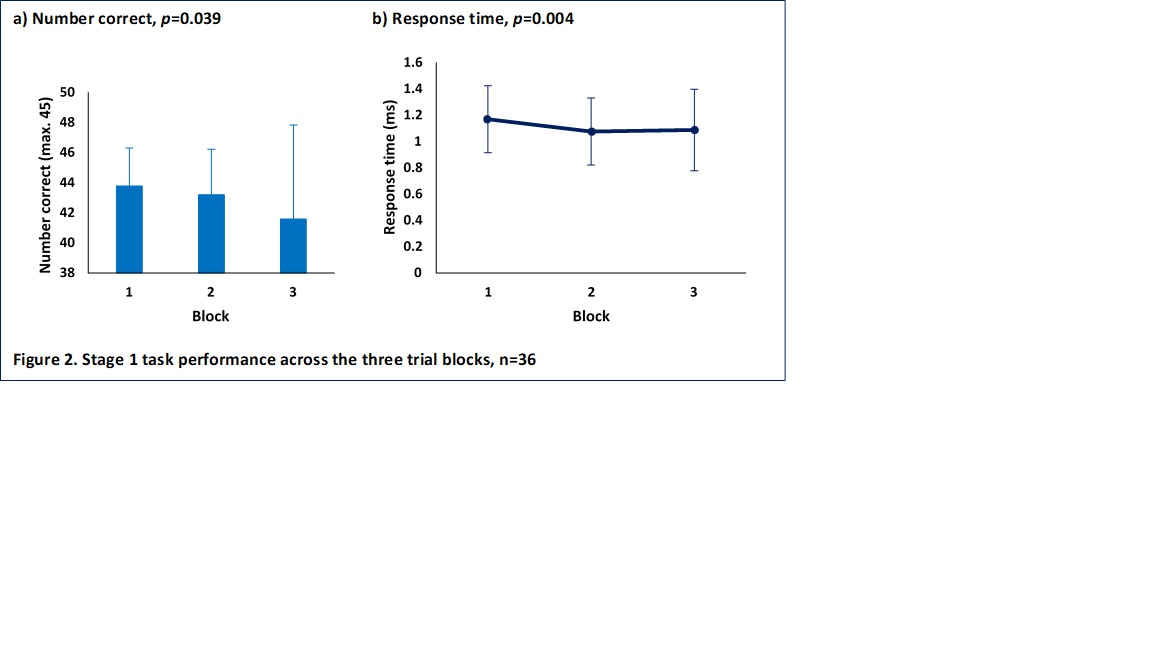
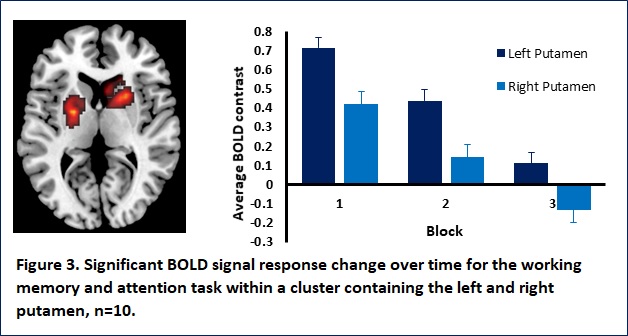
Results: Participants characteristics (n=36) were as follows; mean age 40 ±13, SLE disease duration 15 years ±10, SLEDAI-2K 4.3 ±4.8 and SDI 1 ±1.8. The majority were female (n=27, 75%), Caucasian (n=23, 63%) and educated to college level or higher (n=28, 78%). Current medication use was as follows; immunosuppressants (n=23, 64%), antimalarials (n=29, 81%), glucocorticoids (n=15, 42%) and biologics (n=6, 17%). Mean scores on the Beck Depression Inventory-II and Beck Anxiety Inventory were 14 ±12 and 33 ±11 respectively. CI was seen in 9 (35%). Participant performance on the working memory task worsened over time; response time was slowest during the first trial and quickest during the second (Figure 2). A significant cluster (900 voxels; pFWEc=0.006) containing the left and right putamen was found for the task BOLD response change over time. Within this cluster the BOLD response decreased from trial 1 to trial 3 (Figure 3).
Conclusion: Our new cognitive task is working as expected, with participants' performance worsening over time as participants become less engaged/cognitively fatigued. These results were mirrored by the fMRI results. Response time improved from trial one to two/three as the task was learnt. Final results will include data from healthy controls enabling us to determine if the decrease in performance is similar to healthy controls or if SLE patients become cognitively fatigued more quickly. We will also, examine the results from stage 2, the learning and long-term associative memory task.
M. Barraclough: None; S. McKie: None; A. Kafkas: None; B. Parker: AbbVie, 6, Eli Lilly, 6, Fresenius-Kabi, 6, Genzyme/Sanofi, 5, GSK, 5, Roche, 6; J. Diaz Martinez: None; A. Knight: Pfizer, 6; K. Bingham: None; J. Su: None; M. Kakvan: None; C. Munoz-Grajales: None; C. Tartaglia: None; L. Ruttan: None; J. Wither: AstraZeneca, 1, 6, Pfizer, 12, Indirect salary support through a Chair award to the Division of Rheumatology at the University of Toronto; D. Bonilla: None; D. Montaldi: None; R. Elliott: None; P. Katz: None; D. Beaton: None; R. Green: None; I. Bruce: AstraZeneca, 1, 2, 5, 6, Aurinia, 2, GSK, 1, 2, 5, 6, Janssen, 5, 6, Lilly, 1, UCB, 6; Z. Touma: AstraZeneca, 2, GSK, 2.
Background/Purpose: Cognitive impairment (CI) is a significant problem in SLE but there is a disconnect between objective and subjective CI. This makes it difficult to fully understand and treat CI. Functional magnetic resonance imaging (fMRI) has shown compensatory brain mechanisms may help to maintain cognitive function but potentially cause fatigue leading to subjective CI. This in part could explain the objective/subjective CI disconnect but ways to measure this are needed. We devised a new fMRI paradigm examining attention, working memory, learning and long-term associative memory. The aim was to assess this new task by examining whether attentional performance and working memory decreases over repeated trials in an SLE cohort using our preliminary data.
Methods: Recruited adult SLE patients met EULAR/ACR criteria. Demographic, clinical and psychiatric data and patient reported outcome measures were collected. Cognitive function was assessed using a modified version of the ACR Neuropsychological Battery. MR scans included two structural scans and two functional MRI scans done during stage 1 and 2 of a cognitive task. Stage 1 (working memory and attention, Figure 1) was split into three trial blocks of equal length and performance over the trials was examined using mean number correct (maximum of 45 per trial), mean response time per trial (ms) and fMRI blood-oxygen-level-dependent (BOLD) response signal to stage 1 of the task. Differences between the trial performances were examined using repeated measures ANOVA and the fMRI data was modelled using SPM12. fMRI interference was done at a cluster extent correction threshold of family-wise error-corrected (pFWEc)< 0.05.
Results: Participants characteristics (n=36) were as follows; mean age 40 ±13, SLE disease duration 15 years ±10, SLEDAI-2K 4.3 ±4.8 and SDI 1 ±1.8. The majority were female (n=27, 75%), Caucasian (n=23, 63%) and educated to college level or higher (n=28, 78%). Current medication use was as follows; immunosuppressants (n=23, 64%), antimalarials (n=29, 81%), glucocorticoids (n=15, 42%) and biologics (n=6, 17%). Mean scores on the Beck Depression Inventory-II and Beck Anxiety Inventory were 14 ±12 and 33 ±11 respectively. CI was seen in 9 (35%). Participant performance on the working memory task worsened over time; response time was slowest during the first trial and quickest during the second (Figure 2). A significant cluster (900 voxels; pFWEc=0.006) containing the left and right putamen was found for the task BOLD response change over time. Within this cluster the BOLD response decreased from trial 1 to trial 3 (Figure 3).
Conclusion: Our new cognitive task is working as expected, with participants' performance worsening over time as participants become less engaged/cognitively fatigued. These results were mirrored by the fMRI results. Response time improved from trial one to two/three as the task was learnt. Final results will include data from healthy controls enabling us to determine if the decrease in performance is similar to healthy controls or if SLE patients become cognitively fatigued more quickly. We will also, examine the results from stage 2, the learning and long-term associative memory task.



M. Barraclough: None; S. McKie: None; A. Kafkas: None; B. Parker: AbbVie, 6, Eli Lilly, 6, Fresenius-Kabi, 6, Genzyme/Sanofi, 5, GSK, 5, Roche, 6; J. Diaz Martinez: None; A. Knight: Pfizer, 6; K. Bingham: None; J. Su: None; M. Kakvan: None; C. Munoz-Grajales: None; C. Tartaglia: None; L. Ruttan: None; J. Wither: AstraZeneca, 1, 6, Pfizer, 12, Indirect salary support through a Chair award to the Division of Rheumatology at the University of Toronto; D. Bonilla: None; D. Montaldi: None; R. Elliott: None; P. Katz: None; D. Beaton: None; R. Green: None; I. Bruce: AstraZeneca, 1, 2, 5, 6, Aurinia, 2, GSK, 1, 2, 5, 6, Janssen, 5, 6, Lilly, 1, UCB, 6; Z. Touma: AstraZeneca, 2, GSK, 2.



