Poster Session C
Epidemiology, health policy and outcomes
Session: (1796–1826) Epidemiology & Public Health Poster III
1815: The Association of Body Mass Index with SARS-CoV-2 Infection (COVID-19) in Patients with Inflammatory Arthritis on Biologic Disease Modifying Anti-Rheumatic Drugs: Results from the Singapore National Biologics Register
Tuesday, November 14, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- PD
Preeti Dhanasekaran, BSc, MSc
National University of Singapore
Singapore, SingaporeDisclosure information not submitted.
Abstract Poster Presenter(s)
Preeti Dhanasekaran1, Margaret Ma2, Manjari Lahiri2, Li Wearn Koh3, Stanley Angkodjojo4, Andrew Yu Keat Khor5, James Jiaqi Liu6, Stephanie Wong7 and Siaw Ing Yeo6, 1National University of Singapore, Singapore, Singapore, 2National University Hospital, Singapore, Singapore, 3Tan Tock Seng Hospital, Braddell Heights, Singapore, 4Sengkang General Hospital / Singhealth, Singapore, Singapore, 5Changi General Hospital, Singapore, Singapore, 6Singapore General Hospital, Singapore, Singapore, 7Tan Tock Seng Hospital, Singapore, Singapore
Background/Purpose:
To determine the association of body mass index (BMI) with incident COVID-19 infection in patients with inflammatory arthritis (IA) using biologic disease-modifying anti-rheumatic drugs (bDMARDs) and describe the demographic and clinical characteristics of patients with severe COVID-19.
Methods:
This was a prospective inception cohort study of IA patients ≥ 21 years old initiating a bDMARD after July 2016 across 6 public-sector hospitals in Singapore. Data were collected via face-to-face questionnaires and abstracted from electronic medical records. Baseline characteristics were compared using chi-square test for categorical variables; and t-test and Mann-Whitney U test for continuous variables for normal and non-normal distributions respectively. The association of BMI with COVID-19 infection was analyzed using multivariate logistic regression, adjusting for demographics and other baseline characteristics.
Results:
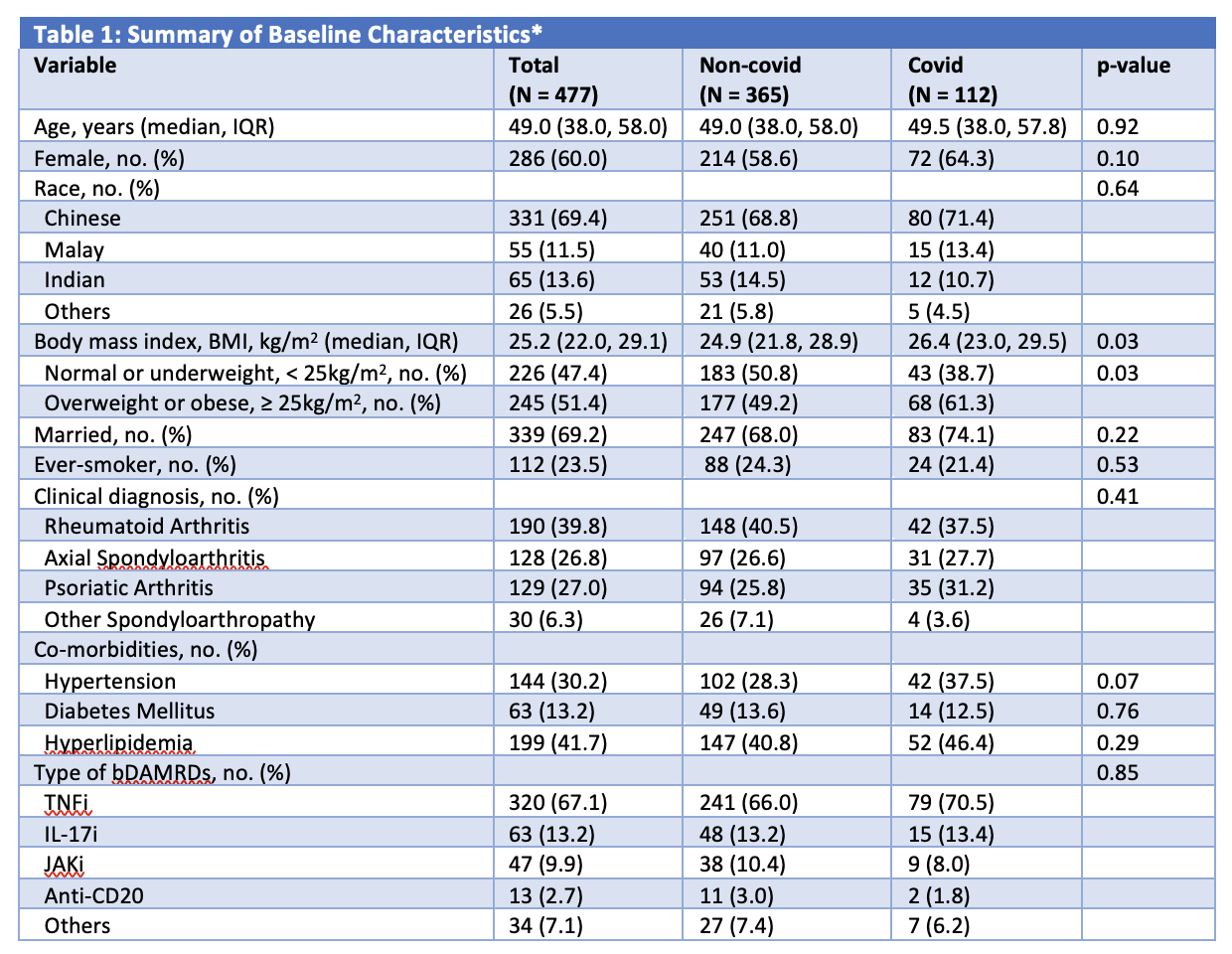
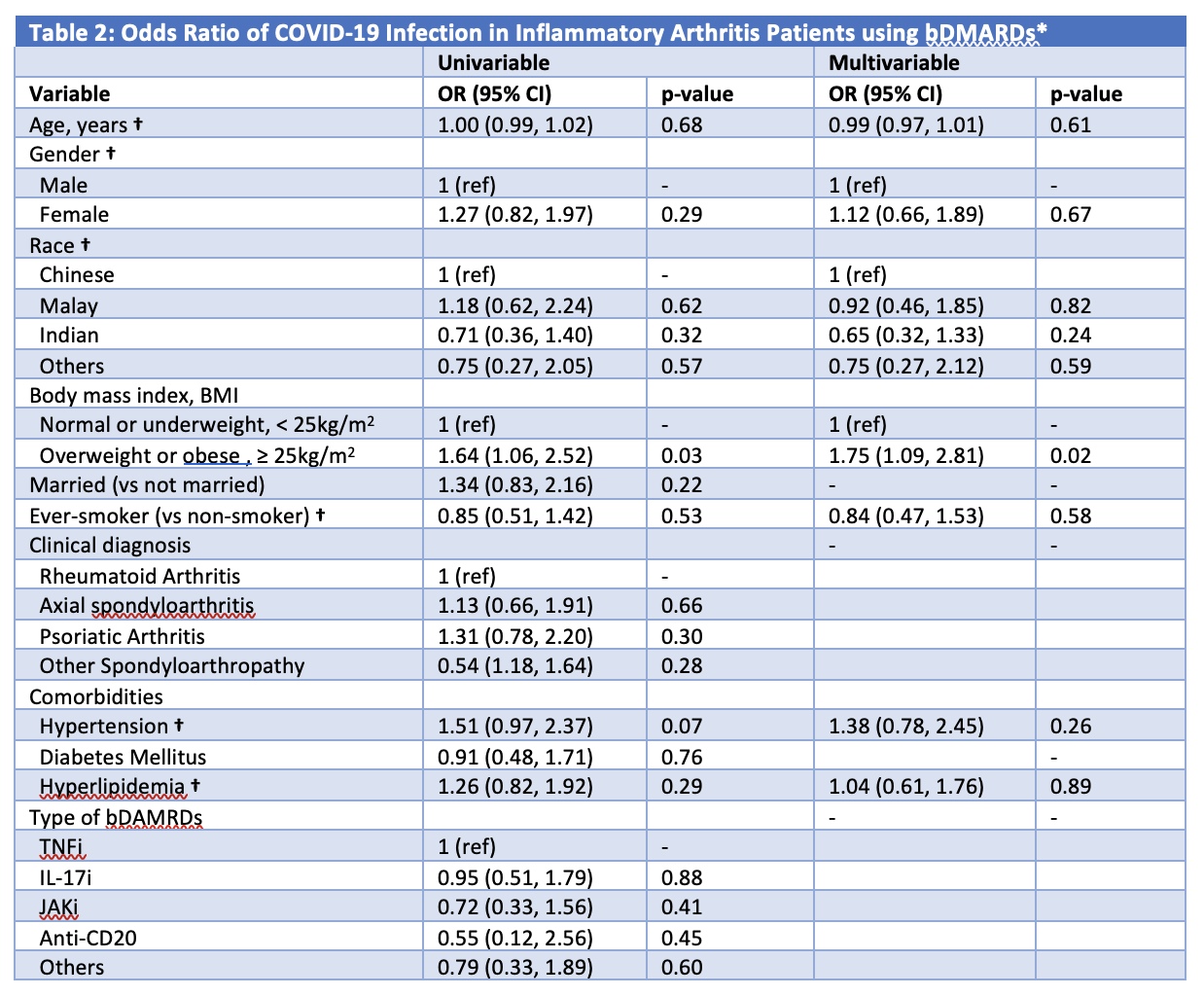
477 patients (60.0% female, 69.4% Chinese) with median (IQR) age of 49.0 (38.0, 58.0)years were included. 39.8% had rheumatoid arthritis, 27.0% had psoriatic arthritis and 33.1% had a spondyloarthropathy. The majority of patients (67.1%) were on tumor necrosis factor inhibitors (TNFi). 148 (31.0%) patients had incident SARS-CoV-2 infection. Compared to patients not infected with COVID-19, those who contracted COVID-19 had a higher BMI (24.9 vs 26.4 kg/m2, p = 0.03) (Table 1). Having a BMI of ≥ 25 kg/m2 (odds ratio [OR] 1.75; 95% CI (1.09, 2.81), p = 0.02) was independently associated with COVID-19 infection, after adjusting for age, sex, race, smoking status and co-morbidities (Table 2).
Severe COVID-19 was defined as infections resulting in hospitalization and/or death. Three (2.0%) of 148 patients had severe COVID-19 infection requiring hospitalization. All were male and two were Chinese. Two had rheumatoid arthritis while one had axial spondyloarthritis. All three had co-morbidities of hypertension and hyperlipidemia, and two had diabetes mellitus. At the time of severe COVID-19 infection, the patients were respectively on golimumab, tofacitinib and adalimumab biosimilar (Amgevita®).
Conclusion:
Higher BMI was associated with incident COVID-19 infection in this cohort. This could be due to upregulation of angiotensin-converting enzyme 2 (ACE2) receptors found in adipose tissues and impaired viral elimination as a result of disease-related and/ or obesity-induced blunted immune response and chronic inflammation [1]. Severe SARS-CoV-2 infections in this cohort were rare.Optimization of BMI may contribute to reducing risk and severity of COVID-19 infection.
[1] Demeulemeester, F., de Punder, K., van Heijningen, M., & van Doesburg, F. (2021). Obesity as a Risk Factor for Severe COVID-19 and Complications: A Review.Cells,10(4), 933. https://doi.org/10.3390/cells10040933
P. Dhanasekaran: None; M. Ma: None; M. Lahiri: None; L. Koh: None; S. Angkodjojo: None; A. Khor: None; J. Liu: None; S. Wong: None; S. Yeo: None.
Background/Purpose:
To determine the association of body mass index (BMI) with incident COVID-19 infection in patients with inflammatory arthritis (IA) using biologic disease-modifying anti-rheumatic drugs (bDMARDs) and describe the demographic and clinical characteristics of patients with severe COVID-19.
Methods:
This was a prospective inception cohort study of IA patients ≥ 21 years old initiating a bDMARD after July 2016 across 6 public-sector hospitals in Singapore. Data were collected via face-to-face questionnaires and abstracted from electronic medical records. Baseline characteristics were compared using chi-square test for categorical variables; and t-test and Mann-Whitney U test for continuous variables for normal and non-normal distributions respectively. The association of BMI with COVID-19 infection was analyzed using multivariate logistic regression, adjusting for demographics and other baseline characteristics.
Results:
477 patients (60.0% female, 69.4% Chinese) with median (IQR) age of 49.0 (38.0, 58.0)years were included. 39.8% had rheumatoid arthritis, 27.0% had psoriatic arthritis and 33.1% had a spondyloarthropathy. The majority of patients (67.1%) were on tumor necrosis factor inhibitors (TNFi). 148 (31.0%) patients had incident SARS-CoV-2 infection. Compared to patients not infected with COVID-19, those who contracted COVID-19 had a higher BMI (24.9 vs 26.4 kg/m2, p = 0.03) (Table 1). Having a BMI of ≥ 25 kg/m2 (odds ratio [OR] 1.75; 95% CI (1.09, 2.81), p = 0.02) was independently associated with COVID-19 infection, after adjusting for age, sex, race, smoking status and co-morbidities (Table 2).
Severe COVID-19 was defined as infections resulting in hospitalization and/or death. Three (2.0%) of 148 patients had severe COVID-19 infection requiring hospitalization. All were male and two were Chinese. Two had rheumatoid arthritis while one had axial spondyloarthritis. All three had co-morbidities of hypertension and hyperlipidemia, and two had diabetes mellitus. At the time of severe COVID-19 infection, the patients were respectively on golimumab, tofacitinib and adalimumab biosimilar (Amgevita®).
Conclusion:
Higher BMI was associated with incident COVID-19 infection in this cohort. This could be due to upregulation of angiotensin-converting enzyme 2 (ACE2) receptors found in adipose tissues and impaired viral elimination as a result of disease-related and/ or obesity-induced blunted immune response and chronic inflammation [1]. Severe SARS-CoV-2 infections in this cohort were rare.Optimization of BMI may contribute to reducing risk and severity of COVID-19 infection.
[1] Demeulemeester, F., de Punder, K., van Heijningen, M., & van Doesburg, F. (2021). Obesity as a Risk Factor for Severe COVID-19 and Complications: A Review.Cells,10(4), 933. https://doi.org/10.3390/cells10040933

*Missing values – 6 (BMI), 3 (Smoking), 5 (Hypertension), 5 (Hyperlipidemia), 5 (Diabetes Mellitus). P-values denote statistical differences between the two groups of non-covid and covid-infected. Chi-square test was used for categorical variables, independent samples t-test for normally distributed continuous variables and Mann-Whitney U test for non-normally distributed continuous variables; to calculate p-values.
bDMARD: biological disease modifying anti-rheumatic drug
TNFi, Tumor necrosis factor inhibitors – adalimumab (Humira®), golimumab, infliximab, etanercept, adalimumab biosimilar (Amgevita®) and infliximab biosimilar (Remsima®); IL-17i, Interleukin-17 inhibitor – secukinumab, ixekizumab; JAKi, Janus kinase inhibitor – tofacitinib, baricitinib; Anti-CD20 – rituximab, rituximab biosimilar (Truxima®); Others include tocilizumab (Interleukin (IL)-6 inhibitor), gulselkumab and ustekinumab (IL-12/23 inhibitors) and abatacept (Cytotoxic T lymphocyte-associated antigen 4-immunoglobulin (CTLA4-Ig)).
bDMARD: biological disease modifying anti-rheumatic drug
TNFi, Tumor necrosis factor inhibitors – adalimumab (Humira®), golimumab, infliximab, etanercept, adalimumab biosimilar (Amgevita®) and infliximab biosimilar (Remsima®); IL-17i, Interleukin-17 inhibitor – secukinumab, ixekizumab; JAKi, Janus kinase inhibitor – tofacitinib, baricitinib; Anti-CD20 – rituximab, rituximab biosimilar (Truxima®); Others include tocilizumab (Interleukin (IL)-6 inhibitor), gulselkumab and ustekinumab (IL-12/23 inhibitors) and abatacept (Cytotoxic T lymphocyte-associated antigen 4-immunoglobulin (CTLA4-Ig)).

bDMARD: biological disease modifying anti-rheumatic drug
* Variables were included in the multivariable model if they were significant at P ≤ 0.2 on univariate analysis. † Included in the multivariable model despite not statistically significant in the univariable analysis due to significant correlation with BMI and need to be adjusted for.
TNFi, Tumor necrosis factor inhibitors – adalimumab (Humira®), golimumab, infliximab, etanercept, adalimumab biosimilar (Amgevita®) and infliximab biosimilar (Remsima®); IL-17i, Interleukin-17 inhibitor – secukinumab, ixekizumab; JAKi, Janus kinase inhibitor – tofacitinib, baricitinib; Anti-CD20 – rituximab, rituximab biosimilar (Truxima®); Others include tocilizumab (IL-6 inhibitor), gulselkumab and ustekinumab (IL-12/23 inhibitors) and abatacept (Cytotoxic T lymphocyte-associated antigen 4-immunoglobulin (CTLA4-Ig)).
* Variables were included in the multivariable model if they were significant at P ≤ 0.2 on univariate analysis. † Included in the multivariable model despite not statistically significant in the univariable analysis due to significant correlation with BMI and need to be adjusted for.
TNFi, Tumor necrosis factor inhibitors – adalimumab (Humira®), golimumab, infliximab, etanercept, adalimumab biosimilar (Amgevita®) and infliximab biosimilar (Remsima®); IL-17i, Interleukin-17 inhibitor – secukinumab, ixekizumab; JAKi, Janus kinase inhibitor – tofacitinib, baricitinib; Anti-CD20 – rituximab, rituximab biosimilar (Truxima®); Others include tocilizumab (IL-6 inhibitor), gulselkumab and ustekinumab (IL-12/23 inhibitors) and abatacept (Cytotoxic T lymphocyte-associated antigen 4-immunoglobulin (CTLA4-Ig)).
P. Dhanasekaran: None; M. Ma: None; M. Lahiri: None; L. Koh: None; S. Angkodjojo: None; A. Khor: None; J. Liu: None; S. Wong: None; S. Yeo: None.



