Poster Session C
Metabolic bone disease
Session: (1996–2018) Osteoporosis & Metabolic Bone Disease – Basic & Clinical Science Poster
2014: The Role of Orosomucoid 2 in the Regulation of Bone Remodeling by Inhibiting Osteoclastogenesis and Promoting Osteogenesis
Tuesday, November 14, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- CL
Chang-hoon Lee, MD, PhD
Wonkwang University Hospital
Iksan Jeonbuk, South KoreaDisclosure information not submitted.
Abstract Poster Presenter(s)
CHONG HYUK CHUNG1, Myeung-Su Lee1 and Chang-hoon Lee2, 1Wonkwang University Hospital, Iksan, South Korea, 2Wonkwang University Hospital, Iksan Jeonbuk, South Korea
Background/Purpose: Orosomucoid (ORM) is one of the acute phase reactant protein family and is expressed in hepatocytes and secreted into plasma under stress conditions such as tissue injury, infection, and inflammation. Three types of ORM are known to date (ORM1, 2 and 3), and ORM2 is mainly expressed in bone marrow cells. ORM2 has only some studies on hepatic metabolism, inflammation and immune regulation, and no studies on bone metabolism have been reported. The aim of this study is to reveal the role of ORM2 in the differentiation and function of osteoclasts and osteoblasts
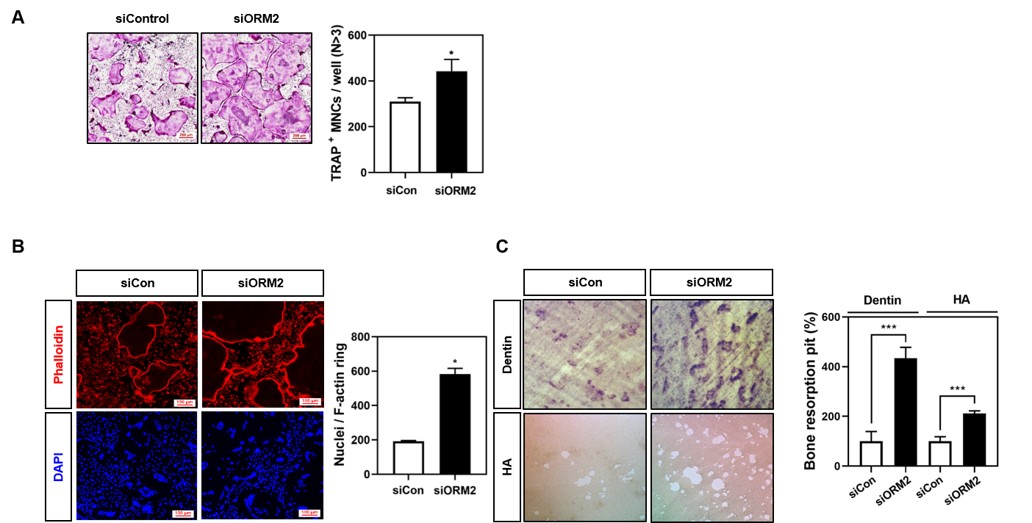
Methods: To determine the effect of ORM2 on RANKL-induced osteoclast differentiation and function, we performed TRAP staining, F-actin stating, and bone resorbing assay using ORM2 recombinant protein or ORM2 siRNA. Also, the effect of ORM2 on osteogenesis was confirmed by ALP and ARS assay. The intracellular mechanisms responsible for the dual regulation of osteoclastogenesis and osteogenesis of ORM2 were revealed by western blotting and quantitative real-time RT-PCR.
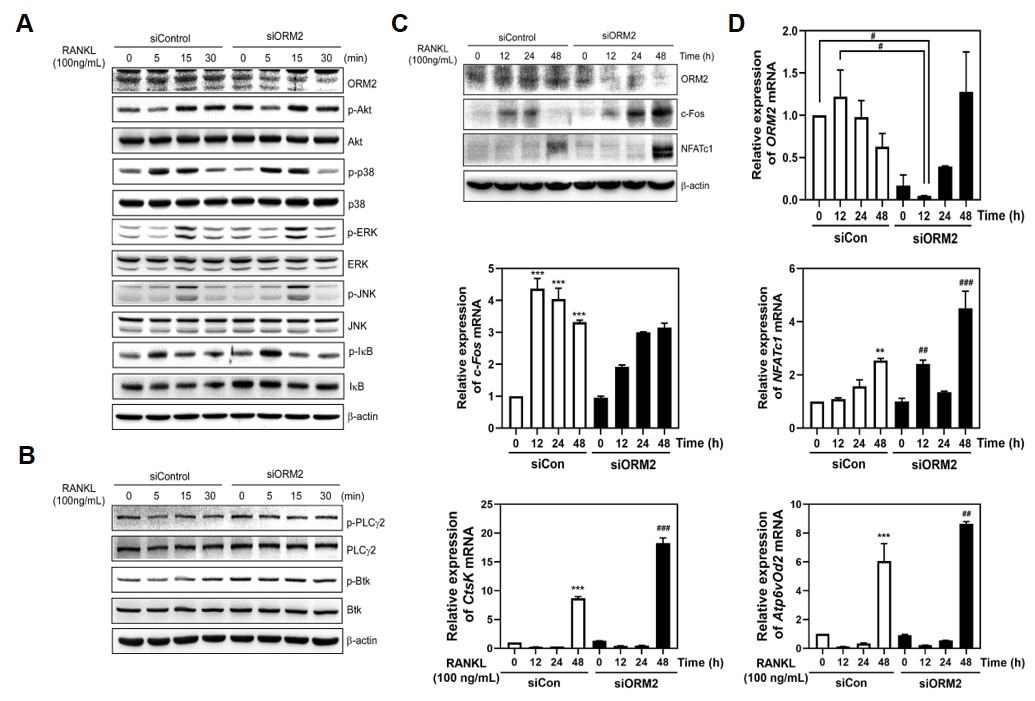
Results: We found that ORM2 is a potential target for osteoporosis therapeutics, as treatment with this agent enhances osteoblast differentiation and bone growth and suppresses osteoclast differentiation and bone resorption by performing gain- and loss-of-function studies. During ORM2-mediated regulation of osteoclastogenesis, phosphorylation of early signal transducers such as p38, JNK, Akt, IB, PLC2, and Btk was affected, which in turn altered the mRNA and protein levels of c-Fos and NFATc1. ORM2 also increased ALP, Alizarin Red-mineralization activity, and the expression of osteoblastogenic gene markers, such as Runx2, osteocalcin (OCN), and ALP in mouse calvarial primary osteoblasts, and activated the p38-Runx2 pathway, which enhanced osteoblast differentiation.
Conclusion: we suggest that ORM2 may be a promising candidate for gene therapy for bone metabolic diseases, and further serve as a potentially important biomarker in the field of bone disease diagnosis.
.jpg)
C. CHUNG: None; M. Lee: None; C. Lee: None.
Background/Purpose: Orosomucoid (ORM) is one of the acute phase reactant protein family and is expressed in hepatocytes and secreted into plasma under stress conditions such as tissue injury, infection, and inflammation. Three types of ORM are known to date (ORM1, 2 and 3), and ORM2 is mainly expressed in bone marrow cells. ORM2 has only some studies on hepatic metabolism, inflammation and immune regulation, and no studies on bone metabolism have been reported. The aim of this study is to reveal the role of ORM2 in the differentiation and function of osteoclasts and osteoblasts
Methods: To determine the effect of ORM2 on RANKL-induced osteoclast differentiation and function, we performed TRAP staining, F-actin stating, and bone resorbing assay using ORM2 recombinant protein or ORM2 siRNA. Also, the effect of ORM2 on osteogenesis was confirmed by ALP and ARS assay. The intracellular mechanisms responsible for the dual regulation of osteoclastogenesis and osteogenesis of ORM2 were revealed by western blotting and quantitative real-time RT-PCR.
Results: We found that ORM2 is a potential target for osteoporosis therapeutics, as treatment with this agent enhances osteoblast differentiation and bone growth and suppresses osteoclast differentiation and bone resorption by performing gain- and loss-of-function studies. During ORM2-mediated regulation of osteoclastogenesis, phosphorylation of early signal transducers such as p38, JNK, Akt, IB, PLC2, and Btk was affected, which in turn altered the mRNA and protein levels of c-Fos and NFATc1. ORM2 also increased ALP, Alizarin Red-mineralization activity, and the expression of osteoblastogenic gene markers, such as Runx2, osteocalcin (OCN), and ALP in mouse calvarial primary osteoblasts, and activated the p38-Runx2 pathway, which enhanced osteoblast differentiation.
Conclusion: we suggest that ORM2 may be a promising candidate for gene therapy for bone metabolic diseases, and further serve as a potentially important biomarker in the field of bone disease diagnosis.
.jpg)
ORM2 expression was increased during RANKL-induced osteoclast differentiation and the administration of recombinant ORM2 protein restrained the formation of osteoclast and bone resorption function.

The knock down of ORM2 improved osteoclast differentiation and bone resorbing function.

The knock down of ORM2 leaded to osteoclast differentiation by increasing RANKL-mediated MAPKs (ERK, JNK), NF-KB, Akt and Btk-PLCr2 calcium signaling.
C. CHUNG: None; M. Lee: None; C. Lee: None.



