Poster Session C
Systemic lupus erythematosus (SLE)
Session: (2257–2325) SLE – Diagnosis, Manifestations, & Outcomes Poster III
2290: Association of Mycophenolate and Azathioprine Use with Cognitive Function in Systemic Lupus Using a Bayesian Longitudinal Item-response Theory Model
Tuesday, November 14, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- CD
Chrisanna Dobrowolski, MD, MS
Icahn School of Medicine at Mount Sinai
New York, NY, United StatesDisclosure information not submitted.
Abstract Poster Presenter(s)
Chrisanna Dobrowolski1, Juan Pablo Diaz Martinez2, Michelle Barraclough3, Roberta Kretzmann4, Sherief Marzouk4, Kathleen Bingham3, Lesley Ruttan5, Dorcas Beaton6, Joan Wither3, Carmela Tartaglia7, Patti Katz8, Mahta Kakvan9, Nicole Anderson10, Dennisse Bonilla3 and Zahi Touma4, 1Icahn School of Medicine at Mount Sinai Hospital, New York, NY, 2Schroeder Arthritis Institute, University Health Network, Toronto, ON, Canada, 3University Health Network, Toronto, ON, Canada, 4University of Toronto, Toronto, ON, Canada, 5University Health Network, Toronto Rehabilitation Institute, Toronto, ON, Canada, 6Institute for Work & Health, Toronto, ON, Canada, 7Krembil Brain Institute, University Health Network, University of Toronto, Toronto, ON, Canada, 8University of California San Francisco, San Rafael, CA, 9University Health Network, University of Toronto, Toronto, ON, Canada, 10Schroeder Arthritis Institute, Krembil Research Institute, University Health Network and University of Toronto, Toronto, ON, Canada
Background/Purpose: Cognitive dysfunction (CD) is a common and often deleterious manifestation of systemic lupus erythematosus (SLE). CD is usually operationalized on the American College of Rheumatology Neuropsychological Battery (ACR-NB) as any z-score of ≤-1.5 on ≥2 domains or z ≤-2 in ≥1 domain; this approach necessarily omits information regarding affected cognitive domains. Using each individual ACR-NB z-scores, the aim of this study was to assess in further detail the association of azathioprine (AZA) and mycophenolate (MMF) use with performance in specific cognitive domains given that these medications have demonstrated overall neuroprotective qualities in prior studies.
Methods: Consecutive, adult SLE patients presenting to a single tertiary care center were considered for participation, providing demographic, clinical and psychiatric data, at multiple visits (0, 6, 12 months). Bayesian longitudinal item response theory (IRT) modelling using the cumulative dose of AZA and MMF, demographic and clinical variables as fixed effects and tests/domains and time/participant as random effects were undertaken to assess ACR-NB z-scores over time. Model One was fitted with an easiness parameter that was estimated for each domain and for each test nested within each domain (higher values represent a higher mean z-scores which represents better cognition). Model Two used the same structure as Model One but added a random slope for the cumulative dose of AZA to vary amongst domain and tests. Finally, leave-one-out cross-validation (loo-cv) was used to determine the best statistical model for our data.
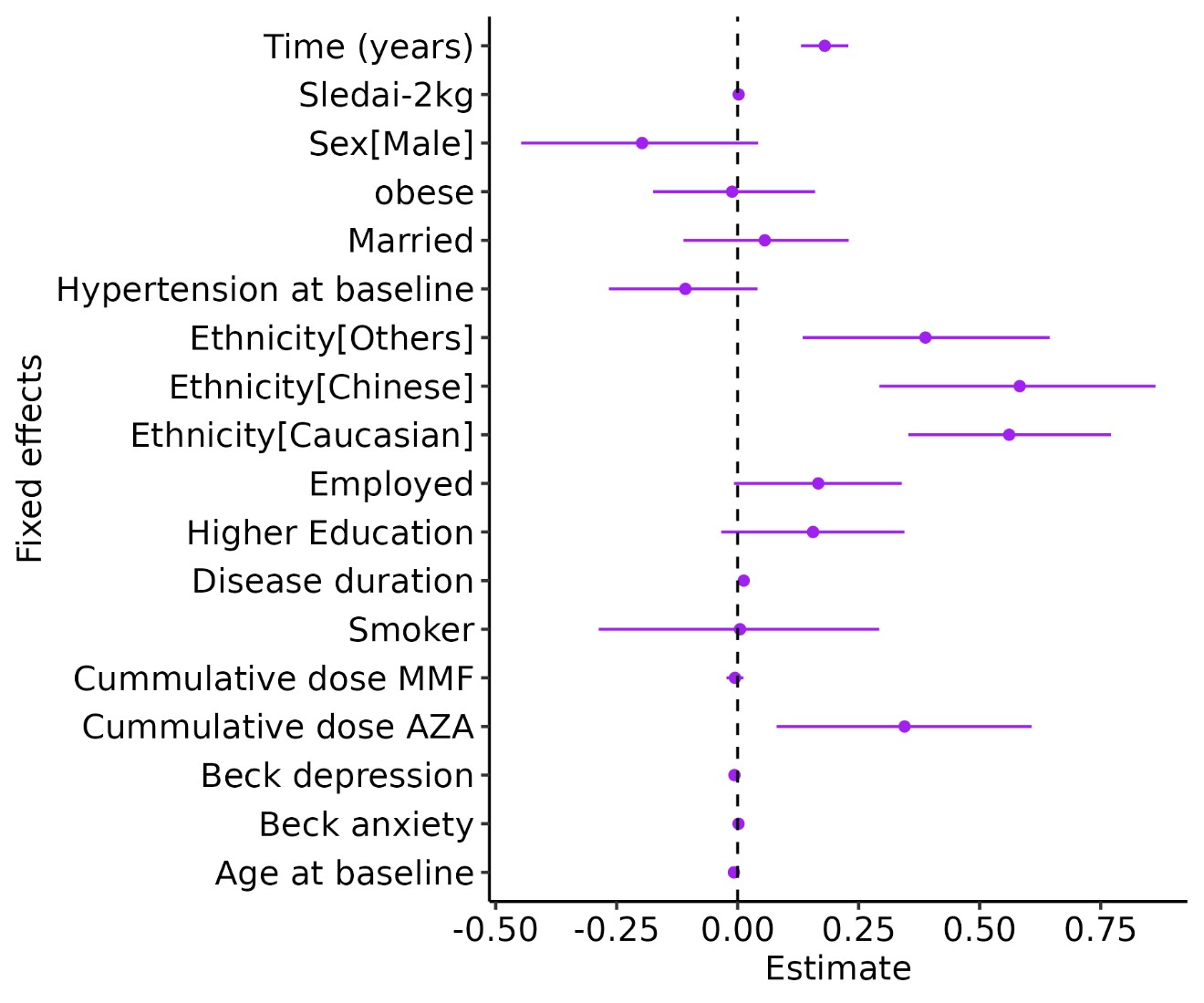
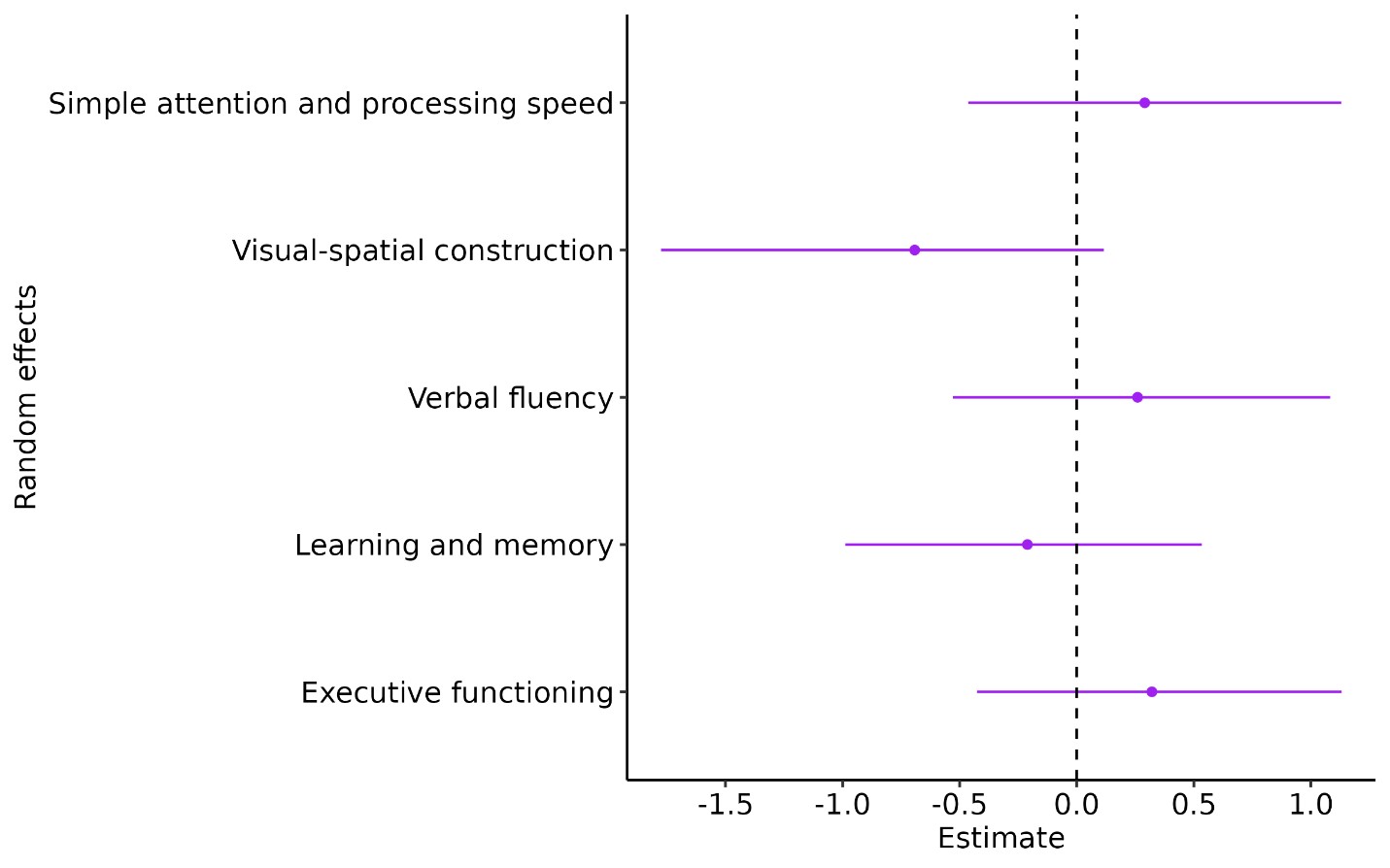
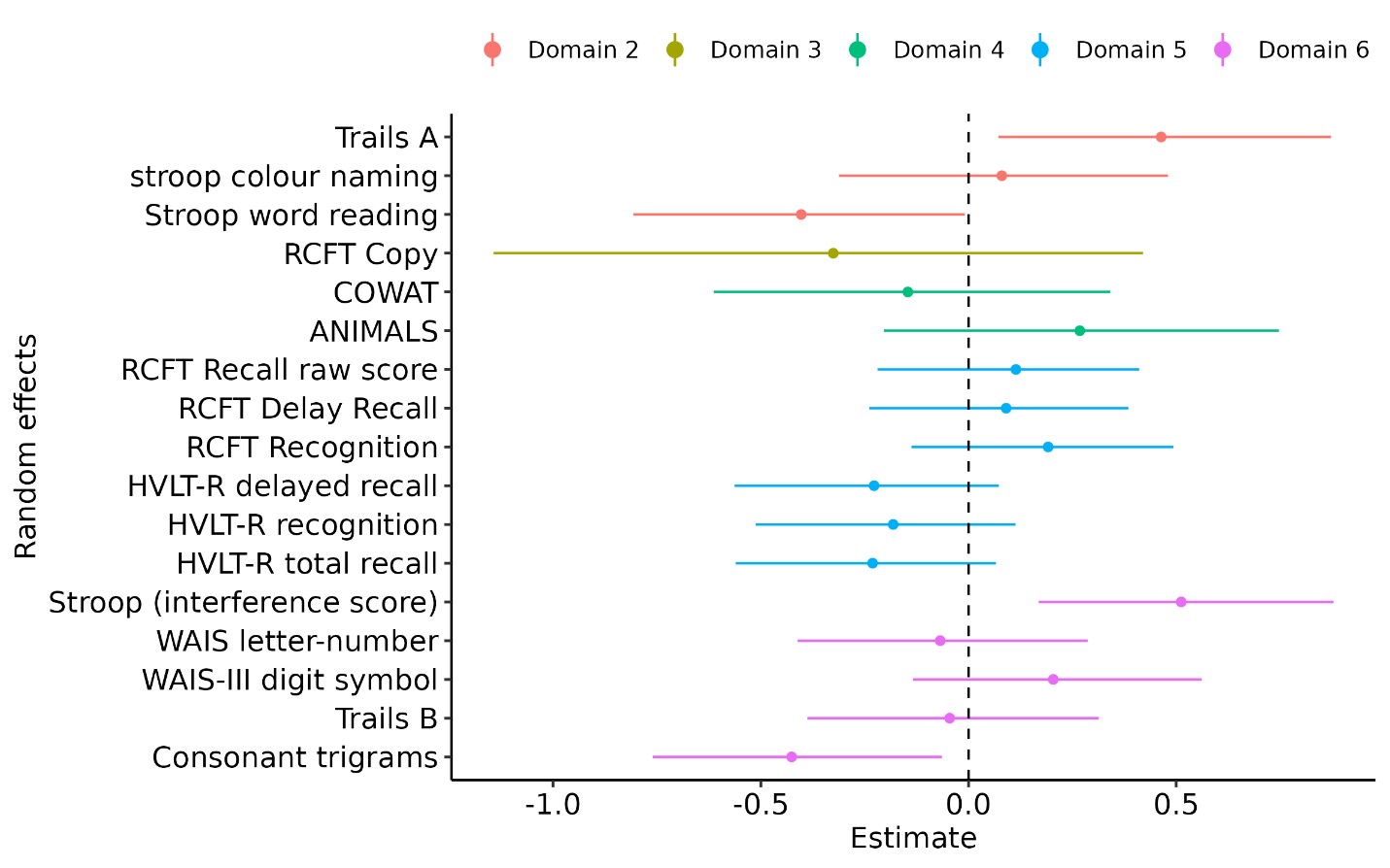
Results: Three hundred participants representing 676 patient visits completed the study. The mean overall z-score with all covariates set to zero (ie. intercept) was -0.84 (95% CrI -1.73, 0.04). Cumulative AZA dose (decigrams/kg) was associated with a z-score increase of 0.34 (95% CrI -0.08, 0.61) on average (Figure 1). Time was also associated with a z-score increase, meaning that on average, z-scores improved as time passed. MMF use was not associated with a protective effect against CD (Figure 1). The easiness parameters for visual-spatial construction domain had the lowest estimate (Figure 2). On average, this domain decreased the z-score by -0.69 (95% CrI -1.77, 0.11). Of note, we found that simple attention and processing speed had a null effect on the mean z-score (Figure 2) but when examining each test within this domain we found that Trails A had a positive effect while STROOP word reading decreased the mean z-score (Figure 3). Loo-cv showed Model One (without adding cumulative AZA dose as a random slope) to be the best fit for our data.
Conclusion: This analysis demonstrated that cognition may improve over time in patients with SLE. It also highlighted which cognitive domains and tests on the ACR-NB are more likely to be affected. Similar to our prior study, increasing cumulative AZA dose was associated with a protective effect against CD, while MMF use was not. This is the first longitudinal IRT model that examines trajectories of individual ACR-NB z-scores and relationship with immunosuppressant use. Additional studies are warranted to further investigate visual-spatial function in SLE, as well as the relationship of AZA and SLE-CD.
C. Dobrowolski: None; J. Diaz Martinez: None; M. Barraclough: None; R. Kretzmann: None; S. Marzouk: None; K. Bingham: None; L. Ruttan: None; D. Beaton: None; J. Wither: AstraZeneca, 1, 6, Pfizer, 12, Indirect salary support through a Chair award to the Division of Rheumatology at the University of Toronto; C. Tartaglia: None; P. Katz: None; M. Kakvan: None; N. Anderson: None; D. Bonilla: None; Z. Touma: AstraZeneca, 2, GSK, 2.
Background/Purpose: Cognitive dysfunction (CD) is a common and often deleterious manifestation of systemic lupus erythematosus (SLE). CD is usually operationalized on the American College of Rheumatology Neuropsychological Battery (ACR-NB) as any z-score of ≤-1.5 on ≥2 domains or z ≤-2 in ≥1 domain; this approach necessarily omits information regarding affected cognitive domains. Using each individual ACR-NB z-scores, the aim of this study was to assess in further detail the association of azathioprine (AZA) and mycophenolate (MMF) use with performance in specific cognitive domains given that these medications have demonstrated overall neuroprotective qualities in prior studies.
Methods: Consecutive, adult SLE patients presenting to a single tertiary care center were considered for participation, providing demographic, clinical and psychiatric data, at multiple visits (0, 6, 12 months). Bayesian longitudinal item response theory (IRT) modelling using the cumulative dose of AZA and MMF, demographic and clinical variables as fixed effects and tests/domains and time/participant as random effects were undertaken to assess ACR-NB z-scores over time. Model One was fitted with an easiness parameter that was estimated for each domain and for each test nested within each domain (higher values represent a higher mean z-scores which represents better cognition). Model Two used the same structure as Model One but added a random slope for the cumulative dose of AZA to vary amongst domain and tests. Finally, leave-one-out cross-validation (loo-cv) was used to determine the best statistical model for our data.
Results: Three hundred participants representing 676 patient visits completed the study. The mean overall z-score with all covariates set to zero (ie. intercept) was -0.84 (95% CrI -1.73, 0.04). Cumulative AZA dose (decigrams/kg) was associated with a z-score increase of 0.34 (95% CrI -0.08, 0.61) on average (Figure 1). Time was also associated with a z-score increase, meaning that on average, z-scores improved as time passed. MMF use was not associated with a protective effect against CD (Figure 1). The easiness parameters for visual-spatial construction domain had the lowest estimate (Figure 2). On average, this domain decreased the z-score by -0.69 (95% CrI -1.77, 0.11). Of note, we found that simple attention and processing speed had a null effect on the mean z-score (Figure 2) but when examining each test within this domain we found that Trails A had a positive effect while STROOP word reading decreased the mean z-score (Figure 3). Loo-cv showed Model One (without adding cumulative AZA dose as a random slope) to be the best fit for our data.
Conclusion: This analysis demonstrated that cognition may improve over time in patients with SLE. It also highlighted which cognitive domains and tests on the ACR-NB are more likely to be affected. Similar to our prior study, increasing cumulative AZA dose was associated with a protective effect against CD, while MMF use was not. This is the first longitudinal IRT model that examines trajectories of individual ACR-NB z-scores and relationship with immunosuppressant use. Additional studies are warranted to further investigate visual-spatial function in SLE, as well as the relationship of AZA and SLE-CD.

Figure 1. Posterior uncertainty intervals for the fixed effects in our model. X axis represents the distribution of the beta coefficients. Y axis represents the parameters where inference is made. The dark shaded region represents the point estimate from a frequentist point of view and the light shaded region represents the 95% uncertainty interval (CrI), i.e., with 95% of probability the estimate would lie in that region.

Figure 2. Posterior uncertainty intervals for the ACR Neuropsychiatric Battery domains (as random effects). X axis represents the mean z-score change and Y axis represents the parameters where inference is made. The dot on the graph represents the point estimate and the line represents the 95% CrI.

Figure 3. Posterior uncertainty intervals for the ACR Neuropsychiatric Battery tests (as random effects). X axis represents the estimated mean z-score change over time and Y axis represents the cognitive test parameters where an inference is made. Each color represents the cognitive domain where the test is nested. The dot on the graph represents the point estimate and the line represents the 95% CrI.
C. Dobrowolski: None; J. Diaz Martinez: None; M. Barraclough: None; R. Kretzmann: None; S. Marzouk: None; K. Bingham: None; L. Ruttan: None; D. Beaton: None; J. Wither: AstraZeneca, 1, 6, Pfizer, 12, Indirect salary support through a Chair award to the Division of Rheumatology at the University of Toronto; C. Tartaglia: None; P. Katz: None; M. Kakvan: None; N. Anderson: None; D. Bonilla: None; Z. Touma: AstraZeneca, 2, GSK, 2.



