Poster Session C
Rheumatoid arthritis (RA)
Session: (2095–2140) RA – Diagnosis, Manifestations, and Outcomes Poster III
2123: Dose Trabecular Bone Score (TBS) Add Predictive Value to Fracture Risk Assessment (FRAX) in Rheumatoid Arthritis?
Tuesday, November 14, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- JC
Jia-Feng Chen, MD (he/him/his)
Chang Gung Memorial Hospital
Kaohsiung city, TaiwanDisclosure information not submitted.
Abstract Poster Presenter(s)
Jia-Feng Chen1, Tien-Tsai Cheng2 and Hong-Yo Kang3, 1Kaohsiung Chang Gung Memorial Hospital and Chang Gung University College of Medicine, Kaohsiung, Taiwan, 2Department of Internal Medicine, Kaohsiung Chang Gung Memorial Hospital and Chang Gung University, College of Medicine, Kaohsiung, Taiwan, 3Graduate Institute of Clinical Medical Sciences, Chang Gung University, College of Medicine, Kaohsiung, Taiwan
Background/Purpose: Trabecular bone score (TBS) of lumbar spine is a dual X-ray densitometry (DXA)-based tool to evaluate bone microarchitecture, providing information on bone quality in metabolic bone diseases. The aim of this study is to assess whether TBS provides additional benefit to Fracture Risk Assessment (FRAX) tool to predict fractures in rheumatoid arthritis (RA) patients.
Methods: This is a retrospective study enrolling RA patients who fulfilled ACR/EULAR 2010 criteria from September 2014 to April 2021. We recruited participants aged between 40-90 and excluded those with malignancy. At enrollment, we measured bone mineral density (BMD) by Dual X-ray densitometry (DXA) at hip and lumbar spine, and assessed TBS at the lumbar spine. The 10-year risk of major osteoporotic fractures and TBS-adjusted risks were calculated on the FRAX website (https://frax.shef.ac.uk/tbs/index.aspx), and the FRAX scores were adjusted by glucocorticoid dose. In current studies, three FRAX models were evaluated, including conventional FRAX score, FRAX adjusted by TBS (TBS-FRAX) and FRAX without input of BMD (FRAX-noBMD). All patients were followed for at least three years to monitor fractures. We defined prevalent vertebral fracture by the morphometric change of vertebral bodies on the X-rays at baselines, according to Genant's semiquantitative assessment. New fracture was defined by any clinical fragility fractures by medical records or vertebral body morphometrical changes after comparing baseline and final X-ray images by independent radiologists. Receiver operating characteristic (ROC) curves were illustrated to compare prediction models.
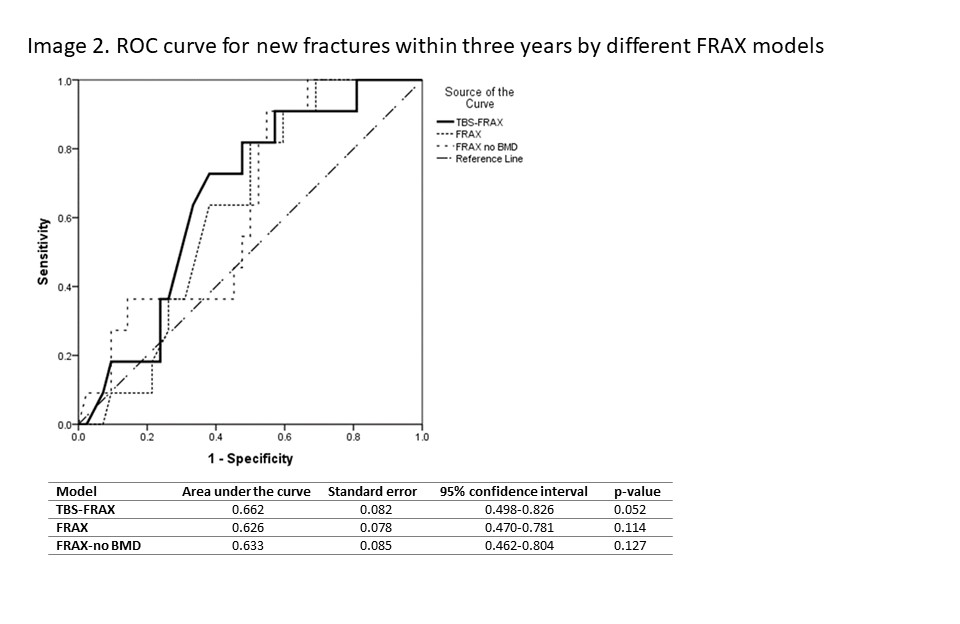
Results: Fifty-three RA participants were enrolled in current study, with a mean age of 55.8± 9.6 years and female predominant (83.0%). Among them, 14 (26.4%) had prevalent vertebral fractures at baseline, while 11 (20.8%) developed new fractures during follow-up. For prevalent vertebral fracture, the area under ROC curves (AUCs) of TBS-FRAX, FRAX, FRAX-noBMD were 0.874, 0.842, and 0.881(all p-value < 0.001), respectively, and there was no statistical significance between different models (Image 1). Regarding new fracture, the AUCs were 0.662, 0.626, and 0.633, and the p-values were 0.052, 0.114, and 0.127, respectively. Also, no statistical significance was displayed between models (Image 2).
Conclusion: FRAX alone demonstrates good prediction ability for prevalent vertebral fractures, so TBS-adjusted FRAX does not add much value to the outcome. The performance of test models is not optimal for new fractures, and adding TBS might improve prediction ability with a borderline of statistical significance.
.jpg)
J. Chen: None; T. Cheng: None; H. Kang: None.
Background/Purpose: Trabecular bone score (TBS) of lumbar spine is a dual X-ray densitometry (DXA)-based tool to evaluate bone microarchitecture, providing information on bone quality in metabolic bone diseases. The aim of this study is to assess whether TBS provides additional benefit to Fracture Risk Assessment (FRAX) tool to predict fractures in rheumatoid arthritis (RA) patients.
Methods: This is a retrospective study enrolling RA patients who fulfilled ACR/EULAR 2010 criteria from September 2014 to April 2021. We recruited participants aged between 40-90 and excluded those with malignancy. At enrollment, we measured bone mineral density (BMD) by Dual X-ray densitometry (DXA) at hip and lumbar spine, and assessed TBS at the lumbar spine. The 10-year risk of major osteoporotic fractures and TBS-adjusted risks were calculated on the FRAX website (https://frax.shef.ac.uk/tbs/index.aspx), and the FRAX scores were adjusted by glucocorticoid dose. In current studies, three FRAX models were evaluated, including conventional FRAX score, FRAX adjusted by TBS (TBS-FRAX) and FRAX without input of BMD (FRAX-noBMD). All patients were followed for at least three years to monitor fractures. We defined prevalent vertebral fracture by the morphometric change of vertebral bodies on the X-rays at baselines, according to Genant's semiquantitative assessment. New fracture was defined by any clinical fragility fractures by medical records or vertebral body morphometrical changes after comparing baseline and final X-ray images by independent radiologists. Receiver operating characteristic (ROC) curves were illustrated to compare prediction models.
Results: Fifty-three RA participants were enrolled in current study, with a mean age of 55.8± 9.6 years and female predominant (83.0%). Among them, 14 (26.4%) had prevalent vertebral fractures at baseline, while 11 (20.8%) developed new fractures during follow-up. For prevalent vertebral fracture, the area under ROC curves (AUCs) of TBS-FRAX, FRAX, FRAX-noBMD were 0.874, 0.842, and 0.881(all p-value < 0.001), respectively, and there was no statistical significance between different models (Image 1). Regarding new fracture, the AUCs were 0.662, 0.626, and 0.633, and the p-values were 0.052, 0.114, and 0.127, respectively. Also, no statistical significance was displayed between models (Image 2).
Conclusion: FRAX alone demonstrates good prediction ability for prevalent vertebral fractures, so TBS-adjusted FRAX does not add much value to the outcome. The performance of test models is not optimal for new fractures, and adding TBS might improve prediction ability with a borderline of statistical significance.
.jpg)
Comparison between ROC curves: TBS-FRAX vs. FRAX, p=0.42; TBS-FRAX vs. FRAX-no BMD, p=0.86; FRAX vs. FRAX-no BMD, p=0.24. Abbreviations: Receiver operating characteristic (ROC); Fracture Risk Assessment (FRAX); Trabecular bone score (TBS); bone mineral density (BMD).

Comparison between ROC curves: TBS-FRAX vs. FRAX, p=0.26; TBS-FRAX vs. FRAX-no BMD, p=0.64; FRAX vs. FRAX-no BMD, p=0.88. Abbreviations: Receiver operating characteristic (ROC); Fracture Risk Assessment (FRAX); Trabecular bone score (TBS); bone mineral density (BMD).
J. Chen: None; T. Cheng: None; H. Kang: None.



