Poster Session C
Periodic fever syndromes, autoinflammatory diseases, Still’s disease and MAS/HLH
Session: (1913–1944) Miscellaneous Rheumatic & Inflammatory Diseases Poster III
1935: Pre-exposure Prophylaxis with Tixegvimab/cilgavimab Is Effective in Limiting the Risk and Severity of COVID-19 in Patients with Auto Immune or Inflammatory Diseases at Increased Risk of Severe COVID-19
Tuesday, November 14, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- MT
Abstract Poster Presenter(s)
Marion THOMAS1, Maeva MASSON2, Raphaele Seror3, Samuel Bitoun4, Henry DUPUY5, Lazato ESTIBALIZ5, Christophe Richez6, Yannick ALLANORE7 and Jerome AVOUAC8, 1HOPITAL COCHIN AP-HP, Service de Rhumatologie, Paris, France, 2CHU Toulouse, Toulouse, France, 3University Hospital Paris Saclay, Le Kremlin-Bicêtre, France, 4CHU Bicêtre APHP, Le Kremlin-Bicêtre, France, 5CHU Bordeaux, Bordeaux, France, 6Université de Bordeaux, Bordeaux, France, 7Université Paris Cité, Paris, France, 8Rheumatology A Department, Hôpital Cochin, AP-HP Centre - Université Paris Cité, Paris, France
Background/Purpose: Patients with autoimmune or inflammatory diseases treated with immunosuppressants such as anti-CD20 are at increased risk for severe COVID-19 and have a high probability of insufficient response to vaccination. The monoclonal antibody combination tixagevimab/cilgavimab has received early access approval to reduce the frequency of symptomatic COVID-19 in immunocompromised patients at risk for severe COVID-19 and unresponsive to vaccination.We aim to evaluate the clinical efficacy of tixagevimab/cilgavimab in pre-exposure prophylaxis in patients with increased risk of severe COVID-19 in rheumatology.
Methods: In this multicenter observational study conducted between December 2021 and August 2022, we included patients with autoimmune or inflammatory diseases who received at least one intramuscular injection of tixagevimab/cilgavimab as pre-exposure prophylaxis in 3 French rheumatology units. Occurrence of COVID-19 was assessed during usual follow-up or by phone call. The endpoint was the incidence of COVID-19 and its severity.
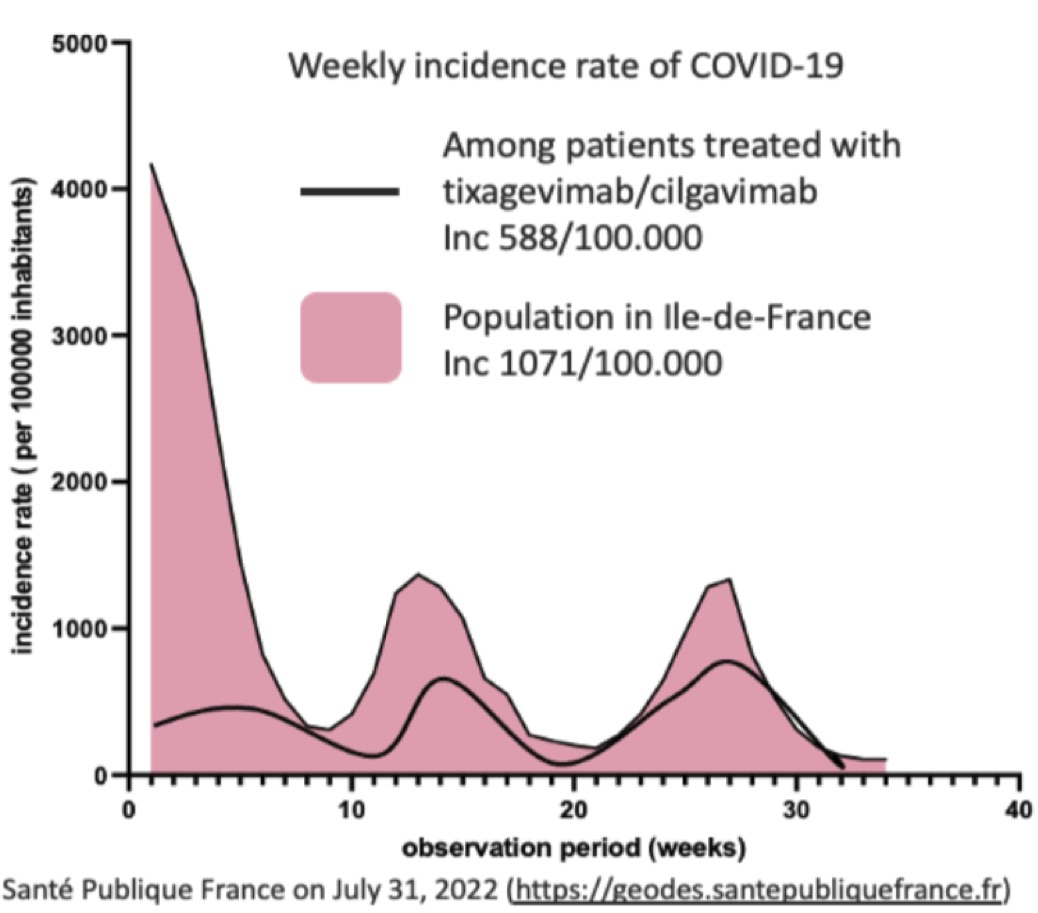
Results: Tixagevimab/cilgavimab was administered to 115 patients, median age 62 years (52-71), with chronic arthritis (n=53), connective tissue disease (n=38) or vasculitis (n=11). The main background immunosuppressants were rituximab (n=98), methotrexate (n=48), mycophenolate mofetil (n=19), cyclophosphamide (n=15), azathioprine (n=12), and corticosteroids (n=62, median dose 5 mg, CI95% 5-8). During a median follow-up of 128 days (93-173), COVID-19 occurred in 23/115 patients (20%) with Omicron identified for the 8 genotyped patients. During the study period, the average weekly incidence was 1071 per 100.000 inhabitants in Ile-de-France vs. 588 per 100.000 in our patients. Patients who received a 2-injections regimen had a lower risk of infection than patients with a single injection (16/49, 33%, vs. 5/64, 8%, p=0.0012). The COVID-19+ patients did not differ from uninfected patients in terms of age, comorbidities, type and duration of underlying disease, extra-articular organ involvement or background immunosuppressants. All COVID-19 cases were non-severe, there were no deaths. The tolerance of injections was excellent, with no side effects observed.
Conclusion: In a population with autoimmune or inflammatory diseases at increased risk of severe COVID-19 with a poor response to vaccination, pre-exposure prophylaxis by tixagevimab/cilgavimab limited the risk of infection and the severity of COVID-19. This study supports the use of COVID-19 serological tests in this patient population in order to detect those who do not respond adequately to vaccination since pre-exposure treatments and/or early treatments are available.
M. THOMAS: None; M. MASSON: None; R. Seror: None; S. Bitoun: None; H. DUPUY: None; L. ESTIBALIZ: None; C. Richez: AbbVie/Abbott, 2, 6, Amgen, 6, AstraZeneca, 2, 6, Bristol-Myers Squibb(BMS), 6, Eli Lilly, 6, 12, receipt of drugs, GlaxoSmithKlein(GSK), 2, 6, Novartis, 2, 6, Pfizer, 2, 6; Y. ALLANORE: AbbVie/Abbott, 2, Alpine Immunoscience, 5, AstraZeneca, 2, Bayer, 2, Boehringer-Ingelheim, 2, Janssen, 2, Medsenic, 2, 5, Mylan, 2, OSE Immunotherapeutics, 5, Prometeus, 2, Roche, 2, Sanofi, 2; J. AVOUAC: AbbVie, 1, 2, 4, 6, BMS, 4, 5, 6, Fresenius Kabi, 4, 5, Galapagos, 1, 2, 4, 6, Lilly, 6, Novartis, 5, 6, Pfizer, 5, 6, Sanofi, 4, 6.
Background/Purpose: Patients with autoimmune or inflammatory diseases treated with immunosuppressants such as anti-CD20 are at increased risk for severe COVID-19 and have a high probability of insufficient response to vaccination. The monoclonal antibody combination tixagevimab/cilgavimab has received early access approval to reduce the frequency of symptomatic COVID-19 in immunocompromised patients at risk for severe COVID-19 and unresponsive to vaccination.We aim to evaluate the clinical efficacy of tixagevimab/cilgavimab in pre-exposure prophylaxis in patients with increased risk of severe COVID-19 in rheumatology.
Methods: In this multicenter observational study conducted between December 2021 and August 2022, we included patients with autoimmune or inflammatory diseases who received at least one intramuscular injection of tixagevimab/cilgavimab as pre-exposure prophylaxis in 3 French rheumatology units. Occurrence of COVID-19 was assessed during usual follow-up or by phone call. The endpoint was the incidence of COVID-19 and its severity.
Results: Tixagevimab/cilgavimab was administered to 115 patients, median age 62 years (52-71), with chronic arthritis (n=53), connective tissue disease (n=38) or vasculitis (n=11). The main background immunosuppressants were rituximab (n=98), methotrexate (n=48), mycophenolate mofetil (n=19), cyclophosphamide (n=15), azathioprine (n=12), and corticosteroids (n=62, median dose 5 mg, CI95% 5-8). During a median follow-up of 128 days (93-173), COVID-19 occurred in 23/115 patients (20%) with Omicron identified for the 8 genotyped patients. During the study period, the average weekly incidence was 1071 per 100.000 inhabitants in Ile-de-France vs. 588 per 100.000 in our patients. Patients who received a 2-injections regimen had a lower risk of infection than patients with a single injection (16/49, 33%, vs. 5/64, 8%, p=0.0012). The COVID-19+ patients did not differ from uninfected patients in terms of age, comorbidities, type and duration of underlying disease, extra-articular organ involvement or background immunosuppressants. All COVID-19 cases were non-severe, there were no deaths. The tolerance of injections was excellent, with no side effects observed.
Conclusion: In a population with autoimmune or inflammatory diseases at increased risk of severe COVID-19 with a poor response to vaccination, pre-exposure prophylaxis by tixagevimab/cilgavimab limited the risk of infection and the severity of COVID-19. This study supports the use of COVID-19 serological tests in this patient population in order to detect those who do not respond adequately to vaccination since pre-exposure treatments and/or early treatments are available.

M. THOMAS: None; M. MASSON: None; R. Seror: None; S. Bitoun: None; H. DUPUY: None; L. ESTIBALIZ: None; C. Richez: AbbVie/Abbott, 2, 6, Amgen, 6, AstraZeneca, 2, 6, Bristol-Myers Squibb(BMS), 6, Eli Lilly, 6, 12, receipt of drugs, GlaxoSmithKlein(GSK), 2, 6, Novartis, 2, 6, Pfizer, 2, 6; Y. ALLANORE: AbbVie/Abbott, 2, Alpine Immunoscience, 5, AstraZeneca, 2, Bayer, 2, Boehringer-Ingelheim, 2, Janssen, 2, Medsenic, 2, 5, Mylan, 2, OSE Immunotherapeutics, 5, Prometeus, 2, Roche, 2, Sanofi, 2; J. AVOUAC: AbbVie, 1, 2, 4, 6, BMS, 4, 5, 6, Fresenius Kabi, 4, 5, Galapagos, 1, 2, 4, 6, Lilly, 6, Novartis, 5, 6, Pfizer, 5, 6, Sanofi, 4, 6.



