Poster Session C
Systemic lupus erythematosus (SLE)
Session: (2257–2325) SLE – Diagnosis, Manifestations, & Outcomes Poster III
2274: Lupus Low Disease Activity State as an Attainable Target in Lupus Nephritis Associated with Reduced Risk of Relapse
Tuesday, November 14, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- CC
Chiu Wai Shirley Chan, MBBS
Division of Rheumatology and Clinial Immunology, Department of Medicine, the University of Hong Kong
Hong Kong, Hong Kong, ChinaDisclosure information not submitted.
Abstract Poster Presenter(s)
Chak Kwan Cheung1, Chak Sing Lau2 and Shirley Chiu Wai Chan3, 1Divison of Rheumatology and Clinical Immunology, Queen Mary Hospital, Department of Medicine, the University of Hong Kong, Hong Kong, Hong Kong, 2University of Hong Kong, Division of Rheumatology & Clinical Immunology, Department of Medicine, Queen Mary Hospital, Hong Kong, Hong Kong, 3Divison of Rheumatology and Clinical Immunology, Department of Medicine, Queen Mary Hospital, the University of Hong Kong, Hong Kong, Hong Kong
Background/Purpose: Lupus nephritis (LN) is a significant comorbidity affecting approximately up to 50-60% of patients with systemic lupus erythematosus (SLE). Complete renal response (CRR) and partial renal response (PRR) have been recommended as treatment targets in LN. Lupus low disease activity state (LLDAS) is an important target associated with improved clinical outcome in SLE. Its role in patients in LN has not been fully evaluated. This study aims to investigate the attainment rate, predictors and outcomes associated with LLDAS attainment in patients with LN.
Methods: Patients with biopsy-proven LN during 2010-2020 in Queen Mary Hospital were included. Baseline demographics, blood parameters and urinalysis results were recorded. Renal response and LLDAS attainment were assessed at 12 months after LN diagnosis. CRR was defined as proteinuria ≤0.5g/day with normal estimate glomerular filtration rate (eGFR); PRR was defined as a reduction in proteinuria by ≥50% with near normal eGFR. LLDAS was attained by meeting: (1) SLE Disease Activity Index ≤4 with no major organ activity; (2) no new lupus disease; (3) physician global assessment ≤1; (4) prednisolone dose ≤7.5mg; (5) standard maintenance immunosuppressants1. Treatment response was defined as proteinuria reduction of ≥50% or to sub-nephrotic range. Relapse was defined as a biopsy-proven active LN on histology after an initial treatment response. Time-to-relapse survival analysis was performed to compare the significance of CRR/PRR and LLDAS attainment.
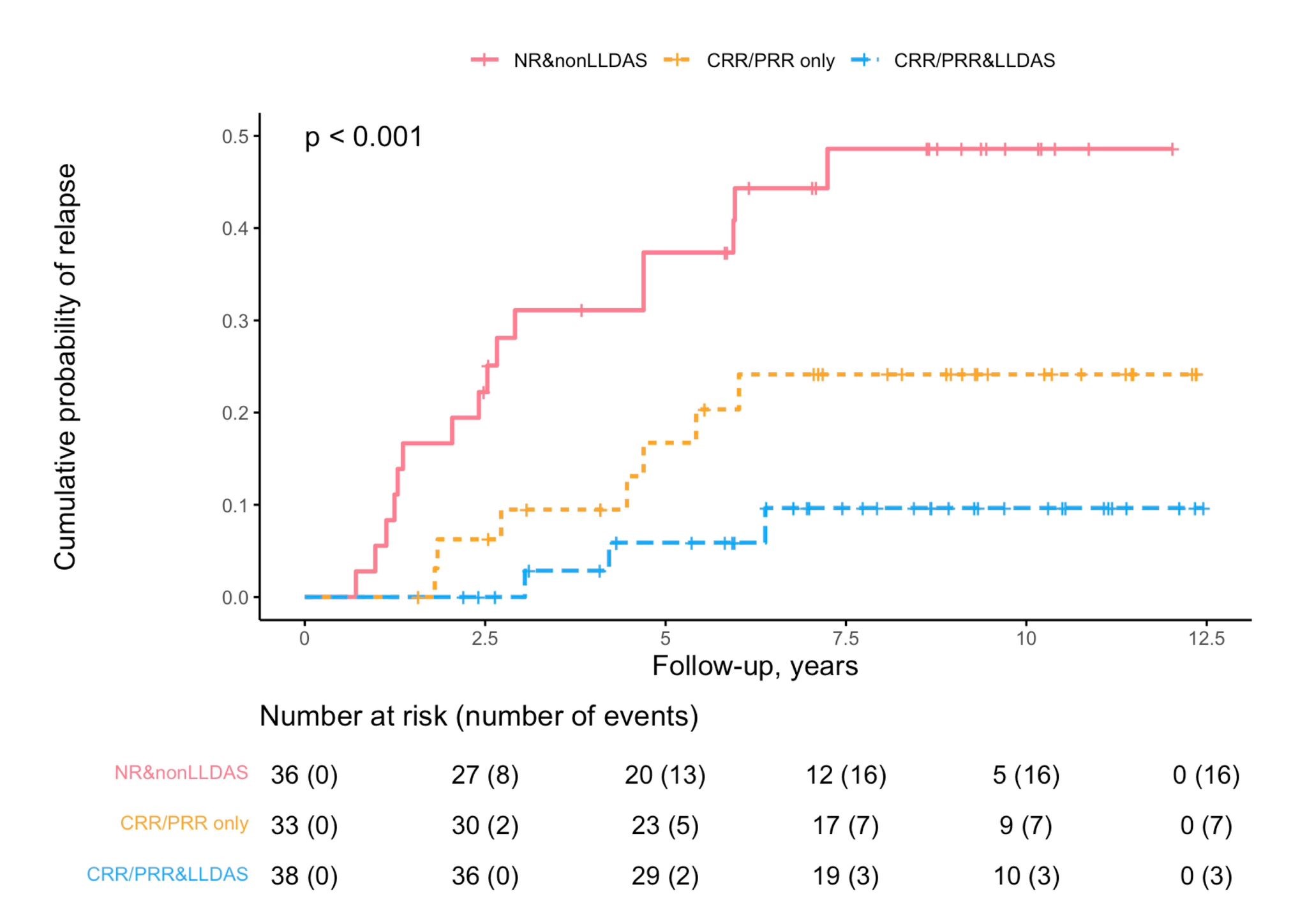
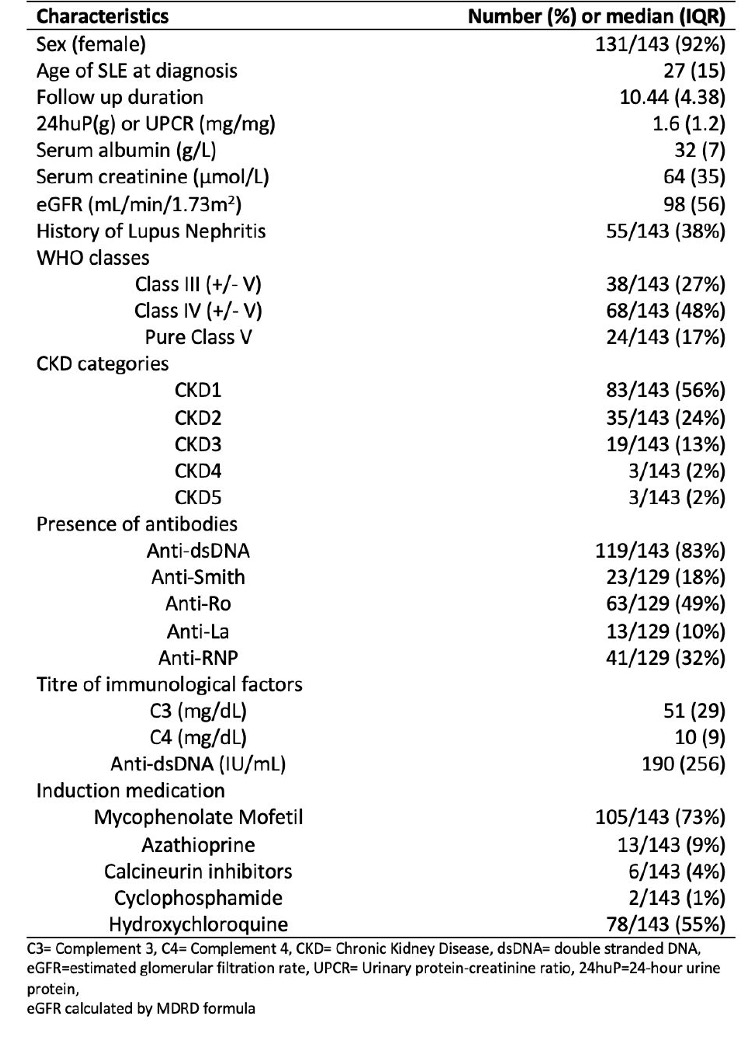
Results: A total of 143 LN patients were included with a median follow-up duration of 10.4 years (Table 1). At 12 months, 57 (40%), 14 (10%) and 69 (48%) patients achieved CRR, PRR and LLDAS, respectively. Although 39 (27%) patients attained both CRR/PRR and LLDAS, a significant number of 30 (21%) patients reached LLDAS without meeting CRR/PRR (Figure 1). Among 136 patients who achieved the pre-defined treatment response, 30 (22%) patients developed LN relapse after a median of 2.98 years. Patients reaching either CRR/PRR or LLDAS had a significantly lower risk of relapse (CRR/PRR: HR = 0.34, p = 0.02 ; LLDAS: HR = 0.28, p = 0.003). The attainment of both CRR/PRR and LLDAS was associated with the lowest risk of relapse (Figure 2).
Conclusion: We advocate LLDAS as a target for LN patients as LLDAS attainment lowers the risk of future relapse.
.jpg)
C. Cheung: None; C. Lau: AstraZeneca, 6, 12, external expert for SLE sterring committee 11.2022; S. Chan: None.
Background/Purpose: Lupus nephritis (LN) is a significant comorbidity affecting approximately up to 50-60% of patients with systemic lupus erythematosus (SLE). Complete renal response (CRR) and partial renal response (PRR) have been recommended as treatment targets in LN. Lupus low disease activity state (LLDAS) is an important target associated with improved clinical outcome in SLE. Its role in patients in LN has not been fully evaluated. This study aims to investigate the attainment rate, predictors and outcomes associated with LLDAS attainment in patients with LN.
Methods: Patients with biopsy-proven LN during 2010-2020 in Queen Mary Hospital were included. Baseline demographics, blood parameters and urinalysis results were recorded. Renal response and LLDAS attainment were assessed at 12 months after LN diagnosis. CRR was defined as proteinuria ≤0.5g/day with normal estimate glomerular filtration rate (eGFR); PRR was defined as a reduction in proteinuria by ≥50% with near normal eGFR. LLDAS was attained by meeting: (1) SLE Disease Activity Index ≤4 with no major organ activity; (2) no new lupus disease; (3) physician global assessment ≤1; (4) prednisolone dose ≤7.5mg; (5) standard maintenance immunosuppressants1. Treatment response was defined as proteinuria reduction of ≥50% or to sub-nephrotic range. Relapse was defined as a biopsy-proven active LN on histology after an initial treatment response. Time-to-relapse survival analysis was performed to compare the significance of CRR/PRR and LLDAS attainment.
Results: A total of 143 LN patients were included with a median follow-up duration of 10.4 years (Table 1). At 12 months, 57 (40%), 14 (10%) and 69 (48%) patients achieved CRR, PRR and LLDAS, respectively. Although 39 (27%) patients attained both CRR/PRR and LLDAS, a significant number of 30 (21%) patients reached LLDAS without meeting CRR/PRR (Figure 1). Among 136 patients who achieved the pre-defined treatment response, 30 (22%) patients developed LN relapse after a median of 2.98 years. Patients reaching either CRR/PRR or LLDAS had a significantly lower risk of relapse (CRR/PRR: HR = 0.34, p = 0.02 ; LLDAS: HR = 0.28, p = 0.003). The attainment of both CRR/PRR and LLDAS was associated with the lowest risk of relapse (Figure 2).
Conclusion: We advocate LLDAS as a target for LN patients as LLDAS attainment lowers the risk of future relapse.
.jpg)
Figure 1: Attainment of CRR/PRR and/ or LLDAS at 12 months.

Figure 2: Risk of LN relapse.

Table 1: Baseline characteristics of 143 patients with biopsy-proven lupus nephritis
C. Cheung: None; C. Lau: AstraZeneca, 6, 12, external expert for SLE sterring committee 11.2022; S. Chan: None.



