Poster Session C
Myopathic rheumatic diseases (polymyositis, dermatomyositis, inclusion body myositis)
Session: (1945–1972) Muscle Biology, Myositis & Myopathies – Basic & Clinical Science Poster III
1961: Anti-PM/Scl Autoantibodies in Juvenile Myositis Are Associated with a Distinct Phenotype Resembling Anti-synthetase Syndrome
Tuesday, November 14, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- MS
Matthew Sherman, MD, MHS
DAIT/NIAID/NIH
Washington, DC, United StatesDisclosure information not submitted.
Abstract Poster Presenter(s)
Matthew Sherman1, Payam Noroozi Farhad2, Edward Trieu3, Katherine Pak4, Iago Pinal-Fernandez4, Kakali Sarkar2, Megan Neely5, Ira Targoff6, Frederick Miller7, Andrew Mammen8 and Lisa Rider9, 1NIAMS/NIH, Washington, DC, 2Environmental Autoimmunity Group, National Institute of Environmental Health Sciences, National Institutes of Health, Bethesda, MD, 3Arthritis and Clinical Immunology, Oklahoma Medical Research Foundation, Oklahoma City, OK, 4National Institutes of Health, Bethesda, MD, 5Department of Biostatistics and Bioinformatics, Duke University, Durham, NC, 6Veterans Affairs Medical Center, University of Oklahoma Health Sciences Center, and Oklahoma Medical Research Foundation, Oklahoma City, OK, 7NIH, NIEHS, Chapel Hill, NC, 8NIH, Bethesda, MD, 9NIEHS, NIH, Bethesda, MD
Background/Purpose: Anti-PM/Scl autoantibodies (Abs) are an uncommon myositis-associated autoantibody (MAA) in juvenile myositis. The clinical features and outcomes associated with anti-PM/Scl Abs in juvenile myositis are largely uncharacterized.
Methods: Patients with juvenile myositis enrolled in NIH myositis natural history studies were screened for anti-PM/Scl and other myositis Abs. Demographics, clinical manifestations, medications, and outcomes of those with and without anti-PM/Scl Abs were compared using Chi-squared, Fisher's exact test, or Wilcoxon rank-sum test. Multivariable logistic regression with adjustment for year of diagnosis and myositis autoantibodies was performed for statistically significant variables from the univariable analyses. A two-sided p< 0.05 was considered significant.
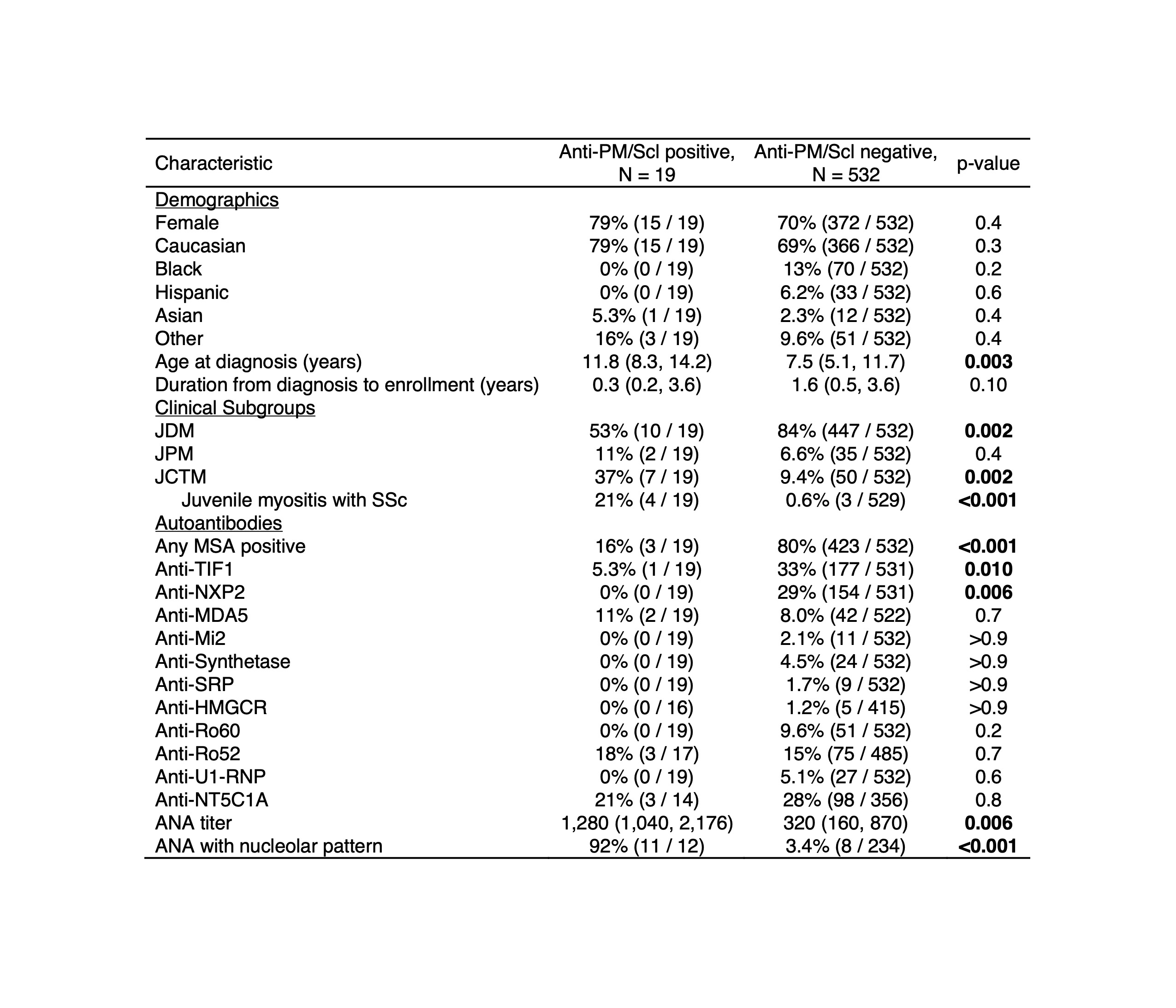
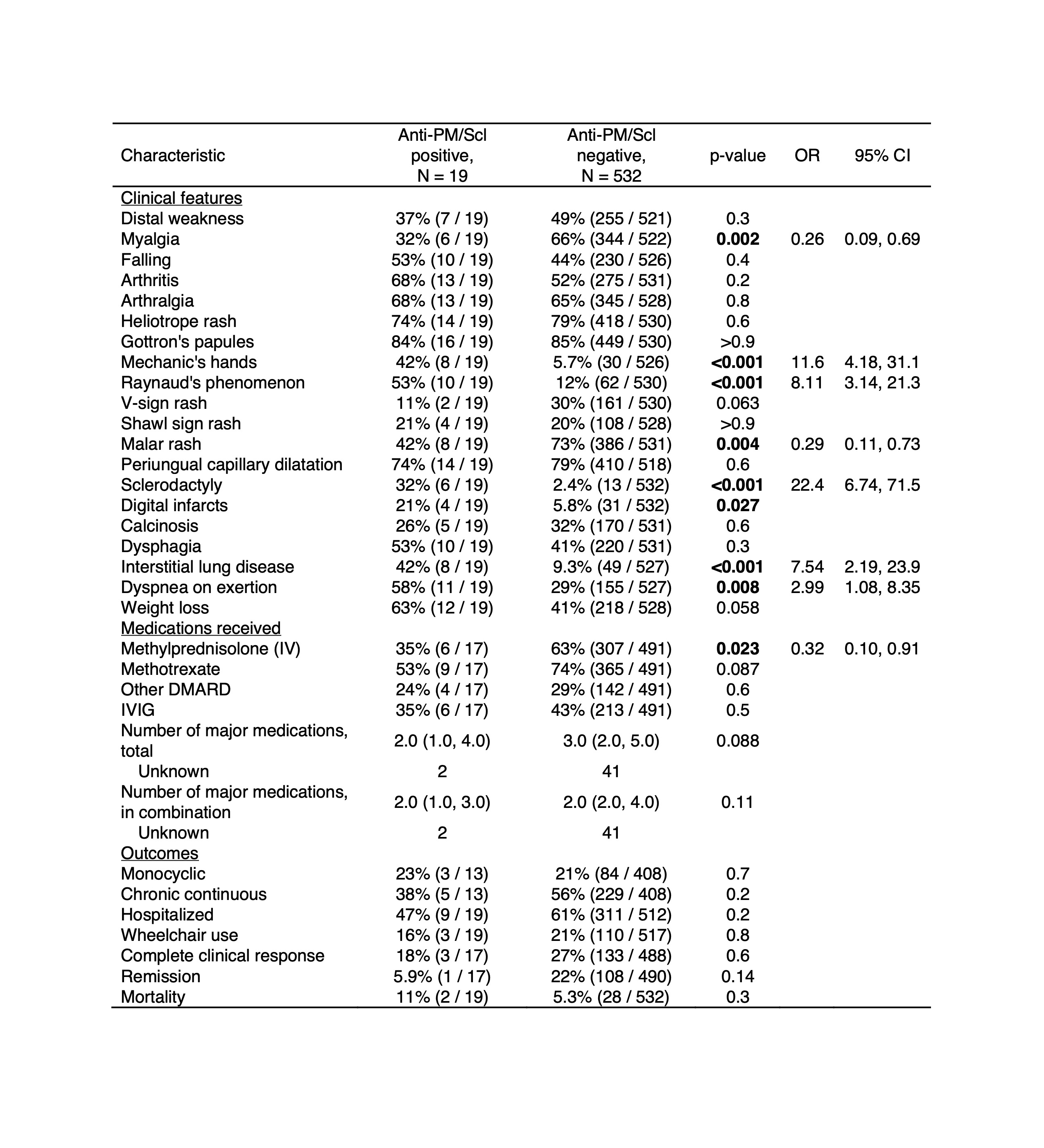
Results: Nineteen of 551 patients (3.4%) had anti-PM/Scl Abs by immunoprecipitation (Table 1). Among those with sera available for immunoblotting, 5 were positive for anti-PM/Scl-100, 1 was positive for anti-PM/Scl-75, 4 were positive for both antigens, and 3 were blot negative, likely reacting only to native protein. Patients with anti-PM/Scl Abs were older at diagnosis (11.8 vs 7.5 years, p=0.003) and more likely to have juvenile connective tissue disease myositis (37% vs 9.4%, p=0.002), frequently overlapping with systemic sclerosis (SSc) (21% vs 0.6%, p< 0.001). MSAs were less commonly associated with anti-PM/Scl Abs (16% vs 80%, p< 0.001). There was no difference in the frequency of anti-Ro52 Abs among patients with or without anti-PM/Scl Abs. ANA titer was higher (1:1280 vs 1:320, p=0.006) and a nucleolar pattern (92% vs 3.4%, p< 0.001) was more common among those with anti-PM/Scl Abs. Patients with anti-PM/Scl Abs had higher odds of certain clinical features (Table 2), including sclerodactyly (OR 22.4; 95% CI 6.74-71.5), mechanic's hands (OR 11.6; 95% CI 4.18-31.1), Raynaud's phenomenon (OR 8.11; 95% CI 3.14-21.3), and ILD (OR 7.54; 95% CI 2.19-23.9) and lower odds of malar rash (OR 0.29; 95% CI 0.11-0.73). There was also a lower odds of treatment with IV methylprednisolone (OR 0.32; 95% CI 0.10-0.91) for those with anti-PM/Scl Abs.
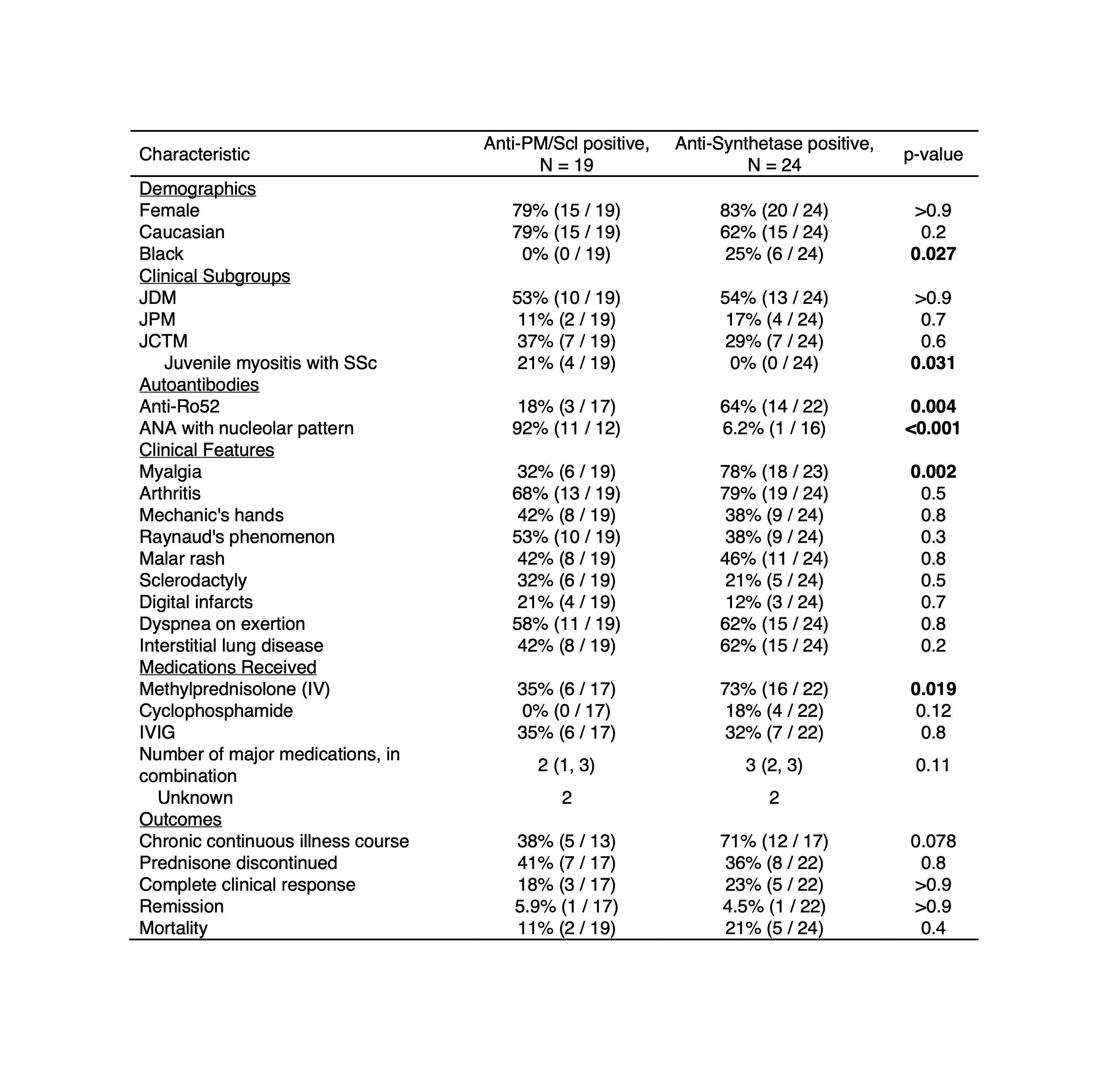
Given the similarity in clinical features to anti-synthetase syndrome, patients with anti-PM/Scl were compared to those with anti-aminoacyl tRNA synthetase (ARS) Abs (Table 3). Anti-ARS Abs were present in 24 (4.3%) patients, similar in frequency to anti-PM/Scl Abs. Among patients with anti-PM/Scl Abs, none was Black (0% vs 25%, p=0.027) and overlap with SSc was more common (21% vs 0%, p=0.031). Those with anti-PM/Scl Abs less often had co-occurring anti-Ro52 Abs (18% vs 64%, p=0.004) and myalgia (32% vs 78%, p=0.002). The presence of ANA with nucleolar pattern (92% vs 6.2%, p< 0.001) was more frequent in anti-PM/Scl Ab-positive patients.
Conclusion: Anti-PM/Scl Abs, unlike other MAAs, were associated with a well-defined phenotype resembling anti-synthetase syndrome. Patients with anti-PM/Scl Abs, compared to those with anti-ARS Abs, were not Black, had an ANA with nucleolar pattern, and less frequently were positive for anti-Ro52 Abs.
Acknowledgements: Intramural Research Program of NIAMS and NIEHS of the NIH.
M. Sherman: None; P. Noroozi Farhad: None; E. Trieu: None; K. Pak: None; I. Pinal-Fernandez: None; K. Sarkar: None; M. Neely: None; I. Targoff: Oklahoma Medical Research Foundation Clinical Immunology Laboratory, 2, UpToDate (Wolters Kluwer), 9; F. Miller: Argenx, 1, Cabaletta, 1, Pfizer, 2, 2; A. Mammen: None; L. Rider: AstraZeneca, 5, Bristol-Myers Squibb(BMS), 5, Hope Pharmaceuticals, 5.
Background/Purpose: Anti-PM/Scl autoantibodies (Abs) are an uncommon myositis-associated autoantibody (MAA) in juvenile myositis. The clinical features and outcomes associated with anti-PM/Scl Abs in juvenile myositis are largely uncharacterized.
Methods: Patients with juvenile myositis enrolled in NIH myositis natural history studies were screened for anti-PM/Scl and other myositis Abs. Demographics, clinical manifestations, medications, and outcomes of those with and without anti-PM/Scl Abs were compared using Chi-squared, Fisher's exact test, or Wilcoxon rank-sum test. Multivariable logistic regression with adjustment for year of diagnosis and myositis autoantibodies was performed for statistically significant variables from the univariable analyses. A two-sided p< 0.05 was considered significant.
Results: Nineteen of 551 patients (3.4%) had anti-PM/Scl Abs by immunoprecipitation (Table 1). Among those with sera available for immunoblotting, 5 were positive for anti-PM/Scl-100, 1 was positive for anti-PM/Scl-75, 4 were positive for both antigens, and 3 were blot negative, likely reacting only to native protein. Patients with anti-PM/Scl Abs were older at diagnosis (11.8 vs 7.5 years, p=0.003) and more likely to have juvenile connective tissue disease myositis (37% vs 9.4%, p=0.002), frequently overlapping with systemic sclerosis (SSc) (21% vs 0.6%, p< 0.001). MSAs were less commonly associated with anti-PM/Scl Abs (16% vs 80%, p< 0.001). There was no difference in the frequency of anti-Ro52 Abs among patients with or without anti-PM/Scl Abs. ANA titer was higher (1:1280 vs 1:320, p=0.006) and a nucleolar pattern (92% vs 3.4%, p< 0.001) was more common among those with anti-PM/Scl Abs. Patients with anti-PM/Scl Abs had higher odds of certain clinical features (Table 2), including sclerodactyly (OR 22.4; 95% CI 6.74-71.5), mechanic's hands (OR 11.6; 95% CI 4.18-31.1), Raynaud's phenomenon (OR 8.11; 95% CI 3.14-21.3), and ILD (OR 7.54; 95% CI 2.19-23.9) and lower odds of malar rash (OR 0.29; 95% CI 0.11-0.73). There was also a lower odds of treatment with IV methylprednisolone (OR 0.32; 95% CI 0.10-0.91) for those with anti-PM/Scl Abs.
Given the similarity in clinical features to anti-synthetase syndrome, patients with anti-PM/Scl were compared to those with anti-aminoacyl tRNA synthetase (ARS) Abs (Table 3). Anti-ARS Abs were present in 24 (4.3%) patients, similar in frequency to anti-PM/Scl Abs. Among patients with anti-PM/Scl Abs, none was Black (0% vs 25%, p=0.027) and overlap with SSc was more common (21% vs 0%, p=0.031). Those with anti-PM/Scl Abs less often had co-occurring anti-Ro52 Abs (18% vs 64%, p=0.004) and myalgia (32% vs 78%, p=0.002). The presence of ANA with nucleolar pattern (92% vs 6.2%, p< 0.001) was more frequent in anti-PM/Scl Ab-positive patients.
Conclusion: Anti-PM/Scl Abs, unlike other MAAs, were associated with a well-defined phenotype resembling anti-synthetase syndrome. Patients with anti-PM/Scl Abs, compared to those with anti-ARS Abs, were not Black, had an ANA with nucleolar pattern, and less frequently were positive for anti-Ro52 Abs.
Acknowledgements: Intramural Research Program of NIAMS and NIEHS of the NIH.

TABLE 1: Demographics and clinicoserologic subgroups of juvenile myositis patients with and without anti-PM/Scl autoantibodies. Abbreviations: ANA, antinuculear antibody; JCTM, juvenile connective tissue myositis; JDM, juvenile dermatomyositis; JPM, juvenile polymyositis; MSA, myositis-specific autoantibody; SSc, systemic sclerosis.

TABLE 2: Clinical features, medications received, and outcomes of juvenile myositis patients with and without anti-PM/Scl autoantibodies. Abbreviations: DMARD, disease-modifying antirheumatic drug; IV, intravenous; IVIG, intravenous immunoglobulin; MTX, methotrexate.

TABLE 3: Comparison of demographics, clinical subgroups, myositis-associated autoantibodies, clinical features, medications received, and outcomes between juvenile myositis patients with anti-PM/Scl and anti-synthetase autoantibodies. Abbreviations: ANA, antinuculear antibody; DMARD, disease-modifying antirheumatic drug; IV, intravenous; IVIG, intravenous immunoglobulin; JCTM, juvenile connective tissue myositis; JDM, juvenile dermatomyositis; JPM, juvenile polymyositis; SSc, systemic sclerosis.
M. Sherman: None; P. Noroozi Farhad: None; E. Trieu: None; K. Pak: None; I. Pinal-Fernandez: None; K. Sarkar: None; M. Neely: None; I. Targoff: Oklahoma Medical Research Foundation Clinical Immunology Laboratory, 2, UpToDate (Wolters Kluwer), 9; F. Miller: Argenx, 1, Cabaletta, 1, Pfizer, 2, 2; A. Mammen: None; L. Rider: AstraZeneca, 5, Bristol-Myers Squibb(BMS), 5, Hope Pharmaceuticals, 5.



