Poster Session C
Imaging
Session: (1862–1894) Imaging of Rheumatic Diseases Poster II
1878: MRI-determined Intramuscular Fat Changes During Oral Glucocorticoid Treatment: Findings from a Feasibility Study
Tuesday, November 14, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- TR
Thurkka Rajeswaran, MBBS, MRCP
University of Leeds
Leeds, United KingdomDisclosure information not submitted.
Abstract Poster Presenter(s)
Thurkka Rajeswaran1, Emma Harris2, hannah Mathieson1, John Biglands3, Paul Stewart1, Ai Lyn Tan1 and Sarah Mackie4, 1University of Leeds, Leeds, United Kingdom, 2The Open University, Milton Keynes, United Kingdom, 3NIHR Leeds Biomedical Research Centre and Medical Physics and Engineering , Leeds Teaching Hospitals NHS Trust Leeds, Leeds, United Kingdom, 4Leeds Institute of Rheumatic and Musculoskeletal Medicine, University of Leeds, Leeds, UK; Leeds Biomedical Research Centre, Leeds Teaching Hospitals NHS Trust, Leeds, United Kingdom
Background/Purpose: Oral glucocorticoid therapy has many adverse effects including insulin resistance and myopathy. Long-term glucocorticoid therapy causes a "sarcopenic obesity" phenotype of adiposity with muscle wasting. The type IIb ("fast twitch") muscle fibres affected by glucocorticoid myopathy make up a greater proportion of hamstrings than of quadriceps. Hamstring muscles also contains a higher proportion of intramuscular fat than quadriceps. In population studies intramuscular fat is a marker of "unhealthy aging" and is associated with elevated morbidity and mortality. We performed a study to determine the feasibility and sensitivity to change of various assessments of muscle for future research studies.
Methods: Modalities for assessment of muscles included MRI (using Siemens MAGNETOM Verio 3T MR Scanner) of proximal leg muscles(hamstrings; semitendinosus, semimembranosus, biceps femoris and quadriceps;rectus femoris, vastus lateralis, vastus medialis,vastus intermedius). A 40 slice volume interpolated breath-hold examination (VIBE), 2-point Dixon, image series was acquired with the inferior slice located at the insertion point of the distal rectus femoris muscle into the tendon. Muscle volume, and single slice intramuscular fat fraction estimates, were acquired from the same VIBE Dixon acquisition. Muscle strength was assessed by biodex/isokinetic dynamometry.Physical performance was assessed via an extended timed get up and go test. Feasibility was assessed in a group of patients treated with oral glucocorticoid therapy for various rheumatological indications and compared to data from age matched controls. Sensitivity to change was assessed in a subset of patients taking < 4 weeks glucocorticoid therapy at first assessment, by repeating the feasible measures after 4-6 months of glucocorticoid therapy.
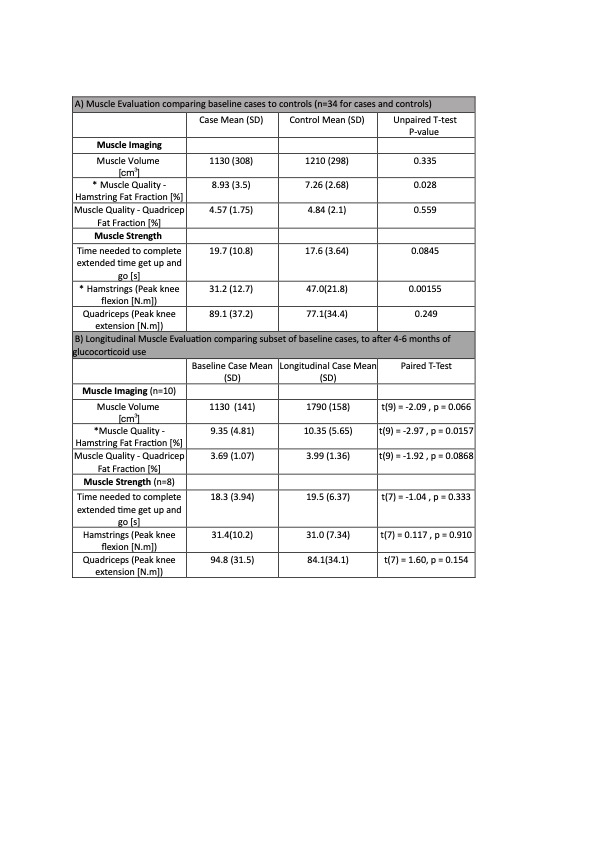
Results: 34 glucocorticoid-treated patients (19 cases of polymyalgia rheumatica (PMR), 4 of giant cell arteritis (GCA), 5 of PMR and GCA, and 6 of other inflammatory conditions) were matched for age (range 45 to 86yrs) and sex (26/34 female) to 34 healthy controls. Compared to controls, patients treated with glucocorticoids had lower hamstring muscle strength and higher fat fraction, but similar muscle volume and physical performance (Table 1A). After 4-6 months of oral glucocorticoid therapy, there was a significant increase in hamstrings fat fraction compared to visit 1, and no significant change in the other measured variables (Table 1B).
Conclusion: Patients taking oral glucocorticoids for rheumatic diseases had greater hamstrings intramuscular fat than matched controls, and reduced hamstrings strength. These differences could have been due to the drug and/or the disease being treated. With increasing duration of glucocorticoid therapy there was a further increase in hamstring fat fraction despite no significant change in muscle strength. Intramuscular fat infiltration could be a sensitive modality for detecting metabolic alterations resulting from long-term glucocorticoid therapy. Further studies are needed to investigate the potential of MRI as a potential endpoint for clinical trials of glucocorticoid-sparing treatments.
T. Rajeswaran: None; E. Harris: None; h. Mathieson: None; J. Biglands: None; P. Stewart: Sparrow Therapeutics, 2; A. Tan: Abbvie, 1, 6, Gilead, 6, Janssen, 6, Lilly, 6, Novartis, 6, Pfizer, 6, UCB, 6; S. Mackie: AbbVie/Abbott, 2, AstraZeneca, 2, GlaxoSmithKlein(GSK), 3, 12, Investigator, National Institute for Health and Care Research, 5, 12, investigator on STERLING-PMR trial, funded by NIHR; patron of the charity PMRGCAuk, Pfizer, 2, 6, Roche, 2, 6, 12, Support from Roche/Chugai to attend EULAR2019 in person, Sanofi, 2, 12, Investigator, Sparrow, 12, Investigator, UCB and Novartis, 6, Vifor, 6.
Background/Purpose: Oral glucocorticoid therapy has many adverse effects including insulin resistance and myopathy. Long-term glucocorticoid therapy causes a "sarcopenic obesity" phenotype of adiposity with muscle wasting. The type IIb ("fast twitch") muscle fibres affected by glucocorticoid myopathy make up a greater proportion of hamstrings than of quadriceps. Hamstring muscles also contains a higher proportion of intramuscular fat than quadriceps. In population studies intramuscular fat is a marker of "unhealthy aging" and is associated with elevated morbidity and mortality. We performed a study to determine the feasibility and sensitivity to change of various assessments of muscle for future research studies.
Methods: Modalities for assessment of muscles included MRI (using Siemens MAGNETOM Verio 3T MR Scanner) of proximal leg muscles(hamstrings; semitendinosus, semimembranosus, biceps femoris and quadriceps;rectus femoris, vastus lateralis, vastus medialis,vastus intermedius). A 40 slice volume interpolated breath-hold examination (VIBE), 2-point Dixon, image series was acquired with the inferior slice located at the insertion point of the distal rectus femoris muscle into the tendon. Muscle volume, and single slice intramuscular fat fraction estimates, were acquired from the same VIBE Dixon acquisition. Muscle strength was assessed by biodex/isokinetic dynamometry.Physical performance was assessed via an extended timed get up and go test. Feasibility was assessed in a group of patients treated with oral glucocorticoid therapy for various rheumatological indications and compared to data from age matched controls. Sensitivity to change was assessed in a subset of patients taking < 4 weeks glucocorticoid therapy at first assessment, by repeating the feasible measures after 4-6 months of glucocorticoid therapy.
Results: 34 glucocorticoid-treated patients (19 cases of polymyalgia rheumatica (PMR), 4 of giant cell arteritis (GCA), 5 of PMR and GCA, and 6 of other inflammatory conditions) were matched for age (range 45 to 86yrs) and sex (26/34 female) to 34 healthy controls. Compared to controls, patients treated with glucocorticoids had lower hamstring muscle strength and higher fat fraction, but similar muscle volume and physical performance (Table 1A). After 4-6 months of oral glucocorticoid therapy, there was a significant increase in hamstrings fat fraction compared to visit 1, and no significant change in the other measured variables (Table 1B).
Conclusion: Patients taking oral glucocorticoids for rheumatic diseases had greater hamstrings intramuscular fat than matched controls, and reduced hamstrings strength. These differences could have been due to the drug and/or the disease being treated. With increasing duration of glucocorticoid therapy there was a further increase in hamstring fat fraction despite no significant change in muscle strength. Intramuscular fat infiltration could be a sensitive modality for detecting metabolic alterations resulting from long-term glucocorticoid therapy. Further studies are needed to investigate the potential of MRI as a potential endpoint for clinical trials of glucocorticoid-sparing treatments.

1a) Muscle Evaluation comparing baseline cases to controls (n=34 for cases and controls)
1b) Longitudinal Muscle Evaluation comparing subset of baseline cases, to after 4-6 months of glucocorticoid use
1b) Longitudinal Muscle Evaluation comparing subset of baseline cases, to after 4-6 months of glucocorticoid use
T. Rajeswaran: None; E. Harris: None; h. Mathieson: None; J. Biglands: None; P. Stewart: Sparrow Therapeutics, 2; A. Tan: Abbvie, 1, 6, Gilead, 6, Janssen, 6, Lilly, 6, Novartis, 6, Pfizer, 6, UCB, 6; S. Mackie: AbbVie/Abbott, 2, AstraZeneca, 2, GlaxoSmithKlein(GSK), 3, 12, Investigator, National Institute for Health and Care Research, 5, 12, investigator on STERLING-PMR trial, funded by NIHR; patron of the charity PMRGCAuk, Pfizer, 2, 6, Roche, 2, 6, 12, Support from Roche/Chugai to attend EULAR2019 in person, Sanofi, 2, 12, Investigator, Sparrow, 12, Investigator, UCB and Novartis, 6, Vifor, 6.



