Poster Session C
Rheumatoid arthritis (RA)
Session: (1713–1733) RA – Animal Models Poster
1725: Distinct Perivascular and Intravascular Lymphatic Mast Cells and Their Role in Lymphatic Clearance, Joint Inflammation, and Bone Erosion in the TNF-Transgenic Murine Arthritis Model
Tuesday, November 14, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- YP
Yue Peng, MS
University of Rochester
Rochester, NY, United StatesDisclosure information not submitted.
Abstract Poster Presenter(s)
Yue Peng1, H. Mark Kenney2, Karen Bentley3, Lianping Xing4, Christopher T Ritchlin5 and Edward Schwarz3, 1University of Rochester Medical Center, Rochester, NY, 2University of Rochester School of Medicine and Dentistry, Henrietta, NY, 3University of Rochester, Rochester, NY, 4University of Rochester Medical Center, Webster, NY, 5University of Rochester Medical School, Allergy, Immunology & Rheumatology Division, Canandaigua, NY
Background/Purpose: Inflammatory-erosive arthritis is exacerbated by lymphatic dysfunction (1), and mast cells (MCs) regulate lymphatic vessel contractions via release of inflammatory and vasoactive mediators (2). In studies to directly assess this relationship, we identified a novel population of intravascular MCs embedded in the cellular architecture of murine joint-draining popliteal lymphatic vessels (PLVs) (3), along with perivascular MCs surrounding the PLV. Here, we investigated the phenotypic differences between PLV perivascular and intravascular MCs to elucidate their respective roles in regulating lymphatic function and inflammatory-erosive arthritis through genetic ablation and pharmacologic inhibition of MCs.
Methods: PLVs from WT, TNF-Tg, and KitW-sh/W-sh (cKit-/-) mice (4), which have a selective hematopoietic deficit in MCs, were harvested for whole mount immunofluorescent microscopy (WMIFM) and toluidine blue histochemistry to identify MCs. In silico analyses were performed on single-cell RNA sequencing (scRNAseq) datasets of PLV MCs (5) vs MC populations identified in the Mouse Cell Atlas (6). Near-infrared indocyanine green (NIR-ICG) imaging quantified lymphatic clearance function. Longitudinal micro-computed tomography (μCT) assessed bone erosions, while H&E and TRAP histology evaluated synovitis and osteoclasts of the afferent ankle. MC deficiency was studied in cKit-/- x TNF-Tg mice (n=3), while MC inhibition was investigated in TNF-Tg mice treated with cromolyn sodium (CS; 3.15mg/g/day/i.p., n=6) vssaline (n=5) for 3 weeks.
Results: WMIFM of MC-tryptase (MCT) confirmed increased numbers of perivascular MCs in TNF-Tg mice, and their complete absence in cKit-/-mice (Fig. 1). In silico scRNAseq analysis revealed a distinct population of PLV MCs vs known MCs in other mouse organs. WMIFM further demonstrated unique molecular signatures of perivascular (MCT+/MCTP1+/MCTP5-) vs intravascular (MCT+/MCTP1-/MCPT5-) MCs (Fig. 2). Remarkably, both CS treatment and cKit-/- x TNF-Tg mice exhibited decreased ICG clearance corresponding with increased bone erosions and synovitis vs their TNF-Tg controls (Fig. 3).
Conclusion: In this work, we characterized unique intravascular MCTP1- MCs in a cellular niche within the tissue structure [BK1] of murine joint-draining PLVs. These intravascular MCs are distinct from known MC subtypes, including perivascular MCTP1+ MCs, which are increased in TNF-Tg and absent in cKit-/- mice. Interestingly, both global MC deletion and inhibition decreased lymphatic function and exacerbated joint disease in TNF-Tg mice. These findings suggest subtype-specific MC regulation of lymphatic function through distinct cellular phenotypes, where MCTP1- intravascular MCs provide a protective role and selective deletion of MCTP1+ perivascular MCs restored lymphatic drainage and ameliorated inflammatory-erosive arthritis.
.jpg)
.jpg)
Y. Peng: None; H. Kenney: None; K. Bentley: None; L. Xing: None; C. Ritchlin: AbbVie, 2, 5, 6, Amgen, 2, BMS, 2, Eli Lilly, 2, Gilead, 2, Janssen, 2, Novartis, 2, Pfizer, 2, 5, 6, UCB, 2, 6; E. Schwarz: None.
Background/Purpose: Inflammatory-erosive arthritis is exacerbated by lymphatic dysfunction (1), and mast cells (MCs) regulate lymphatic vessel contractions via release of inflammatory and vasoactive mediators (2). In studies to directly assess this relationship, we identified a novel population of intravascular MCs embedded in the cellular architecture of murine joint-draining popliteal lymphatic vessels (PLVs) (3), along with perivascular MCs surrounding the PLV. Here, we investigated the phenotypic differences between PLV perivascular and intravascular MCs to elucidate their respective roles in regulating lymphatic function and inflammatory-erosive arthritis through genetic ablation and pharmacologic inhibition of MCs.
Methods: PLVs from WT, TNF-Tg, and KitW-sh/W-sh (cKit-/-) mice (4), which have a selective hematopoietic deficit in MCs, were harvested for whole mount immunofluorescent microscopy (WMIFM) and toluidine blue histochemistry to identify MCs. In silico analyses were performed on single-cell RNA sequencing (scRNAseq) datasets of PLV MCs (5) vs MC populations identified in the Mouse Cell Atlas (6). Near-infrared indocyanine green (NIR-ICG) imaging quantified lymphatic clearance function. Longitudinal micro-computed tomography (μCT) assessed bone erosions, while H&E and TRAP histology evaluated synovitis and osteoclasts of the afferent ankle. MC deficiency was studied in cKit-/- x TNF-Tg mice (n=3), while MC inhibition was investigated in TNF-Tg mice treated with cromolyn sodium (CS; 3.15mg/g/day/i.p., n=6) vssaline (n=5) for 3 weeks.
Results: WMIFM of MC-tryptase (MCT) confirmed increased numbers of perivascular MCs in TNF-Tg mice, and their complete absence in cKit-/-mice (Fig. 1). In silico scRNAseq analysis revealed a distinct population of PLV MCs vs known MCs in other mouse organs. WMIFM further demonstrated unique molecular signatures of perivascular (MCT+/MCTP1+/MCTP5-) vs intravascular (MCT+/MCTP1-/MCPT5-) MCs (Fig. 2). Remarkably, both CS treatment and cKit-/- x TNF-Tg mice exhibited decreased ICG clearance corresponding with increased bone erosions and synovitis vs their TNF-Tg controls (Fig. 3).
Conclusion: In this work, we characterized unique intravascular MCTP1- MCs in a cellular niche within the tissue structure [BK1] of murine joint-draining PLVs. These intravascular MCs are distinct from known MC subtypes, including perivascular MCTP1+ MCs, which are increased in TNF-Tg and absent in cKit-/- mice. Interestingly, both global MC deletion and inhibition decreased lymphatic function and exacerbated joint disease in TNF-Tg mice. These findings suggest subtype-specific MC regulation of lymphatic function through distinct cellular phenotypes, where MCTP1- intravascular MCs provide a protective role and selective deletion of MCTP1+ perivascular MCs restored lymphatic drainage and ameliorated inflammatory-erosive arthritis.

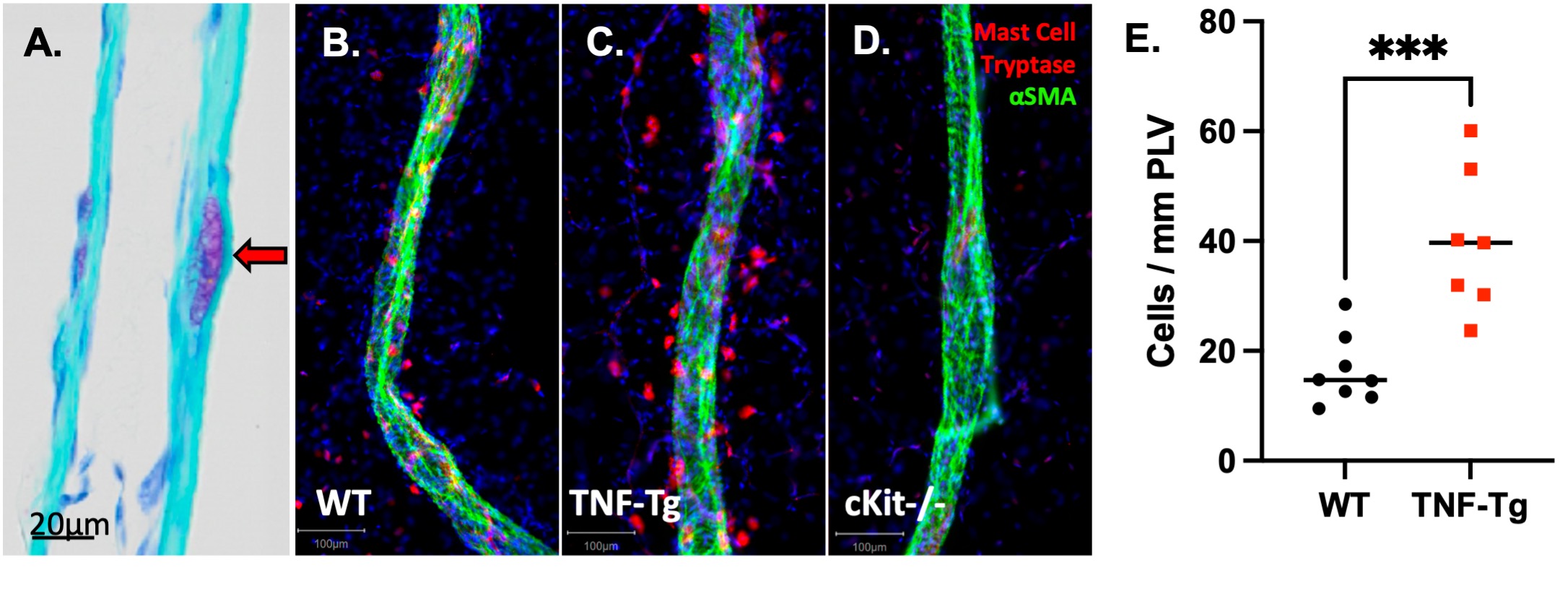
Figure 1. Identification of intravascular MCs and increased numbers of MCT+ perivascular MCs in TNF-Tg mice. Longitudinal sections of PLVs obtained from WT mice were subjected to toluidine blue staining, and a 200x micrograph is shown highlighting a novel intravascular MC embedded within the lymphatic vessel wall (red arrow), situated between the layers of lymphatic endothelial and lymphatic muscle cells (A) (3). PLVs were harvested for WMIFM of αSMA (green) and MCT (red), and representative 200x images are shown for WT (B),TNF-Tg (C), cKit-/-(D) mice, which demonstrated a complete absence of MCT+ MCs in cKit-/- as confirmation of the genetic ablation in this model. Quantification of perivascular MCT+ cells showed a significant increase in MCs surrounding inflamed TNF-Tg PLVs, normalized to vessel length (E, n=8). Statistics: Unpaired Mann Whitney test (E); ***p < 0.001.
.jpg)
Figure 2. Identification of a unique MCT+/MCTP1-/MCPT5- PLV intravascular MC population. In silico datasets were used to extract MCs from a comprehensive scRNAseq dataset of 242k cells encompassing various organs in the Mouse Cell Atlas (6). The extraction was performed based on the expression of MC marker genes (Mcpt4, Cma1, Cpa3, Tpsb2, Kit, Fcer1a, Gata2) to subset MCs from other cell types. These MCs were subsequently integrated with the MCs from our previously published scRNAseq dataset of PLV tissue (5), reclustered, and shown as a UMAP. Notably, the PLV MCs (arrow) exhibited a distinct transcriptional pattern distinct from other MCs identified in the Mouse Cell Atlas (A). WMIFM was performed on PLVs for αSMA, MCT, MCPT1, and MCPT5, as described in Figure 1. A 400x image highlights the MCT+ intravascular MC (B, red arrow) embedded in gaps between αSMA+ lymphatic muscle cells, while a 200x image with 400x inset (white box) exhibits the MCPT1+ perivascular MC (pink arrow) and MCPT1- intravascular MC (white arrow) that exists within the αSMA- void (C). A 200x image with 400x inset (white box) demonstrates that both perivascular (red arrow) and intravascular (white arrow) MCs are MCPT5-negative (D).
.jpg)
Figure 3. Global MC deletion and inhibition with cromolyn sodium decrease lymphatic clearance and exacerbate inflammatory-erosive arthritis in TNF-Tg mice. KitW-sh/W-sh (cKit-/-) and TNF-Tg mice were crossed to generate TNF-Tg x cKit-/- cohorts (both C57BL/6 background). Male mice at 4.5 months of age underwent NIR-ICG imaging, which demonstrated a combinatorial reduction of lymphatic function in the setting of inflammation (TNF-Tg) and MC ablation (cKit-/-) (A). In TNF-Tg mice, the enhanced lymphatic deficiency with MC deletion was associated with increased bone erosions in the talus (green bone) of the afferent ankle joint, quantified by micro-CT with represented images provided (B, C). Histomorphometric analysis revealed a corresponding increase in synovial inflammation (D, H&E) and number of osteoclasts (E, TRAP) in the talus region of MC deficient TNF-Tg x cKit-/- mice, as shown by representative images (F). To evaluate the effects of MC inhibition, 4-month-old female TNF-Tg mice (n=10) were randomized to 3-weeks of placebo (PBS) or cromolyn sodium (CS) treatment (n = 5 mice/group; 10 limbs) based on baseline lymphatic clearance by NIR-ICG imaging. Terminal NIR-ICG and micro-CT imaging revealed significantly reduced lymphatic clearance (G) and midfoot bone volumes (H, I; green talus as representative image) with CS, respectively. Coinciding with the increased bone erosions, histomorphometry also showed increased synovial (J) and TRAP+ osteoclast area (K) with representative images provided (L). Statistics: unpaired t-test; *p<0.05, **p<0.01, ***p<0.001, ****p<0.0001.
Y. Peng: None; H. Kenney: None; K. Bentley: None; L. Xing: None; C. Ritchlin: AbbVie, 2, 5, 6, Amgen, 2, BMS, 2, Eli Lilly, 2, Gilead, 2, Janssen, 2, Novartis, 2, Pfizer, 2, 5, 6, UCB, 2, 6; E. Schwarz: None.



