Poster Session C
Periodic fever syndromes, autoinflammatory diseases, Still’s disease and MAS/HLH
Session: (1913–1944) Miscellaneous Rheumatic & Inflammatory Diseases Poster III
1925: Importance of the Patient Pain Experience in Disease Activity Assessment in the Adult SAPHO and Chronic Nonbacterial Osteomyelitis Study
Tuesday, November 14, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- AL
Aleksander Lenert, MD, MS, FRCPC (he/him/his)
University of Iowa
Iowa City, IA, United StatesDisclosure information not submitted.
Abstract Poster Presenter(s)
Aleksander Lenert1, Robyn Domsic2, Karmela Kim Chan3, Melissa Oliver4, Jisna Paul5, Courtney Kremer6, Emma Leisinger1, Helena Abodeely1, Sandy Hong6, Arundathi Jayatilleke7, Petar Lenert8, T. Shawn Sato1, Yongdong (Dan) Zhao9, Jonathan Templin1, Mary Vaughan-Sarrazin1, Daniel Solomon10 and Polly Ferguson11, 1University of Iowa, Iowa City, IA, 2University of Pittsburgh, Pittsburgh, PA, 3Hospital For Special Surgery, New York, NY, 4Indiana University, Indianapolis, IN, 5Ohio State University Hospital, Columbus, OH, 6University of Iowa Stead Family Children's Hospital, Iowa City, IA, 7Temple University, Philadelphia, PA, 8University of Iowa Hospitals and Clinics, Iowa City, IA, 9University of Washington, Seattle, WA, 10Brigham and Women's Hospital, Boston, MA, 11University of Iowa Carver College of Medicine, Iowa City, IA
Background/Purpose: Musculoskeletal pain is a key symptom experienced by patients with SAPHO and chronic nonbacterial osteomyelitis (SAPHO-CNO), yet its relation to disease activity assessment remains unknown. We sought to determine the relation between patient-reported outcome (PRO) measures for pain, and the physician and patient assessments of disease activity in adults with SAPHO-CNO.
Methods: Adults (≥18 years of age) with SAPHO-CNO enrolled in the SAPHO-CNO study (SCS), a prospective longitudinal observational study, completed 5 PRO instruments for the assessment of pain: 1) global pain in SAPHO-CNO by the numeric rating scale (NRS, 0-10); 2) pain scale of the Short Form 36-item survey (SF-36) (0-100); 3) Patient-Reported Outcomes Measurement Information System (PROMIS) pain interference short form (SF) (6a v1.1); 4) PROMIS nociceptive pain quality SF (5a v2.0); and 5) PROMIS neuropathic pain quality SF (5a v2.0). Subjects completed a patient global assessment (NRS, 0-10) for overall and musculoskeletal (MSK) disease activity in SAPHO-CNO. Physician-assessed disease activity was measured by the physician global scale (NRS, 0-10) for overall and MSK symptoms. Correlational analyses using Spearman's rank correlation coefficient (rho, r) were performed between 1) physician-assessed and patient-reported disease activity (for overall and MSK), and 2) disease activity (by physician and patient) and PRO pain measures. P-values less than 0.05 were considered significant.
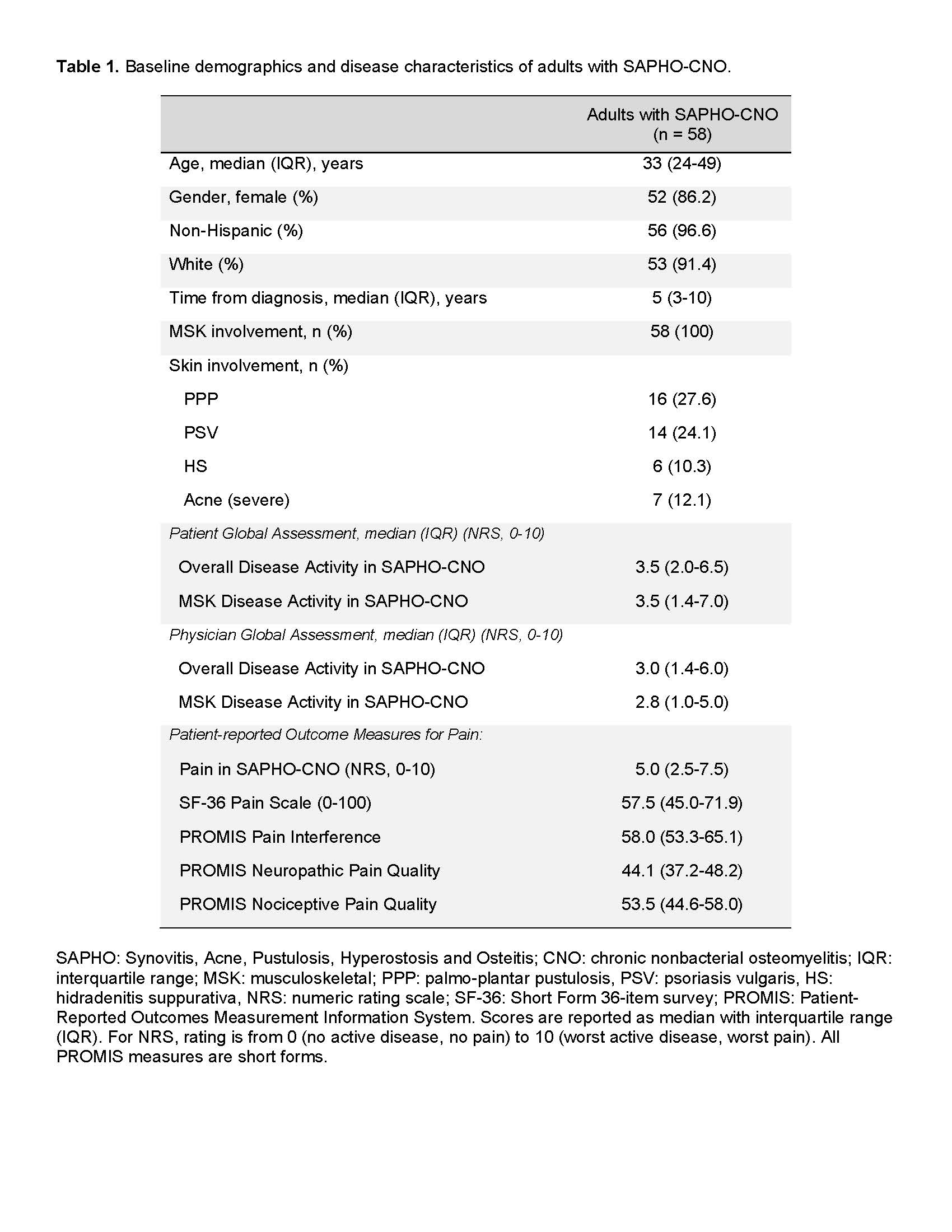
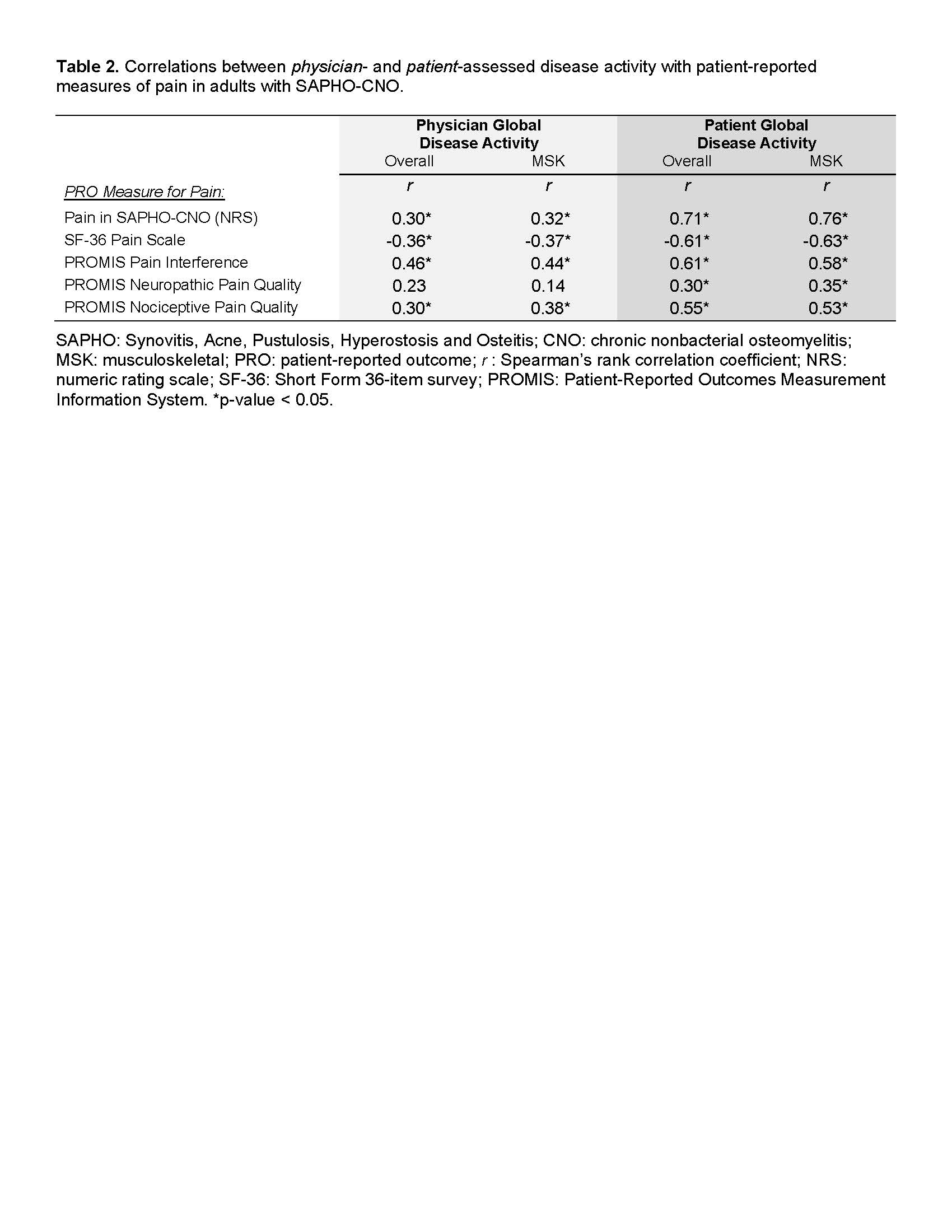
Results: Fifty-eight subjects in the SCS completed their assessments at baseline and were included in this cross-sectional analysis. Baseline characteristics of the adult SAPHO-CNO cohort are summarized in Table 1. We observed weak correlation between physician-assessed and patient-reported overall (r(56)=0.37, p=0.005) and MSK (r(56)=0.34, p=0.009) disease activity in SAPHO-CNO. Next, we observed a weak correlation for 4 out of 5 PRO pain measures and physician-assessed disease activity, except for PROMIS pain interference which showed moderate correlation (r ~0.44-0.46) with physician-assessed overall and MSK disease activity (Table 2). In contrast, there was moderate/good correlation for 3 out of 5 PRO pain measures, and strong correlation of pain by NRS with overall (r = 0.71) and MSK (r = 0.76) disease activity by the patient (Table 2). The PROMIS neuropathic scale showed weak correlation with patient-reported disease activity; additionally, this scale had a high floor effect (29.3% of subjects had the lowest score).
Conclusion: The physician global assessment of disease activity in adult SAPHO-CNO does not fully capture patient pain and likely misses important disease experiences from the patient perspective. For pain quality assessment, the PROMIS neuropathic pain scale may not be suitable. Incorporating patient-reported pain measures will be important for the development of a future composite disease activity index in adult SAPHO-CNO.
A. Lenert: None; R. Domsic: Aisa Pharma, Inc., 2, CSL Behring, 1; K. Chan: None; M. Oliver: None; J. Paul: None; C. Kremer: None; E. Leisinger: None; H. Abodeely: None; S. Hong: None; A. Jayatilleke: None; P. Lenert: None; T. Sato: None; Y. Zhao: None; J. Templin: None; M. Vaughan-Sarrazin: None; D. Solomon: CorEvitas, 5, Janssen, 5, moderna, 5, Novartis, 5, UpToDate, 9; P. Ferguson: None.
Background/Purpose: Musculoskeletal pain is a key symptom experienced by patients with SAPHO and chronic nonbacterial osteomyelitis (SAPHO-CNO), yet its relation to disease activity assessment remains unknown. We sought to determine the relation between patient-reported outcome (PRO) measures for pain, and the physician and patient assessments of disease activity in adults with SAPHO-CNO.
Methods: Adults (≥18 years of age) with SAPHO-CNO enrolled in the SAPHO-CNO study (SCS), a prospective longitudinal observational study, completed 5 PRO instruments for the assessment of pain: 1) global pain in SAPHO-CNO by the numeric rating scale (NRS, 0-10); 2) pain scale of the Short Form 36-item survey (SF-36) (0-100); 3) Patient-Reported Outcomes Measurement Information System (PROMIS) pain interference short form (SF) (6a v1.1); 4) PROMIS nociceptive pain quality SF (5a v2.0); and 5) PROMIS neuropathic pain quality SF (5a v2.0). Subjects completed a patient global assessment (NRS, 0-10) for overall and musculoskeletal (MSK) disease activity in SAPHO-CNO. Physician-assessed disease activity was measured by the physician global scale (NRS, 0-10) for overall and MSK symptoms. Correlational analyses using Spearman's rank correlation coefficient (rho, r) were performed between 1) physician-assessed and patient-reported disease activity (for overall and MSK), and 2) disease activity (by physician and patient) and PRO pain measures. P-values less than 0.05 were considered significant.
Results: Fifty-eight subjects in the SCS completed their assessments at baseline and were included in this cross-sectional analysis. Baseline characteristics of the adult SAPHO-CNO cohort are summarized in Table 1. We observed weak correlation between physician-assessed and patient-reported overall (r(56)=0.37, p=0.005) and MSK (r(56)=0.34, p=0.009) disease activity in SAPHO-CNO. Next, we observed a weak correlation for 4 out of 5 PRO pain measures and physician-assessed disease activity, except for PROMIS pain interference which showed moderate correlation (r ~0.44-0.46) with physician-assessed overall and MSK disease activity (Table 2). In contrast, there was moderate/good correlation for 3 out of 5 PRO pain measures, and strong correlation of pain by NRS with overall (r = 0.71) and MSK (r = 0.76) disease activity by the patient (Table 2). The PROMIS neuropathic scale showed weak correlation with patient-reported disease activity; additionally, this scale had a high floor effect (29.3% of subjects had the lowest score).
Conclusion: The physician global assessment of disease activity in adult SAPHO-CNO does not fully capture patient pain and likely misses important disease experiences from the patient perspective. For pain quality assessment, the PROMIS neuropathic pain scale may not be suitable. Incorporating patient-reported pain measures will be important for the development of a future composite disease activity index in adult SAPHO-CNO.

Table 1. Baseline demographics and disease characteristics of adults with SAPHO-CNO.

Table 2. Correlations between physician- and patient-assessed disease activity with patient-reported measures of pain in adults with SAPHO-CNO.
A. Lenert: None; R. Domsic: Aisa Pharma, Inc., 2, CSL Behring, 1; K. Chan: None; M. Oliver: None; J. Paul: None; C. Kremer: None; E. Leisinger: None; H. Abodeely: None; S. Hong: None; A. Jayatilleke: None; P. Lenert: None; T. Sato: None; Y. Zhao: None; J. Templin: None; M. Vaughan-Sarrazin: None; D. Solomon: CorEvitas, 5, Janssen, 5, moderna, 5, Novartis, 5, UpToDate, 9; P. Ferguson: None.



