Poster Session C
Systemic lupus erythematosus (SLE)
Session: (2257–2325) SLE – Diagnosis, Manifestations, & Outcomes Poster III
2269: Evaluating Global Patterns in Treatment and Prevalence of Glucocorticoid Related Comorbidities in Systemic Lupus Erythematosus: An International Study
Tuesday, November 14, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- CW
Chris Wincup, MBBS, BSc, MRCP
King's College Hospital
London, United KingdomDisclosure information not submitted.
Abstract Poster Presenter(s)
Amelia Holloway1, Sook Yan Lee2, Elena Nikiphorou3, Ioannis Parodis4, Simone Appenzeller5, Naveen R6, Jessica Day7, Mrudula Joshi8, Sreoshy Saha9, Syahrul Sazliyana Shaharir10, Wanruchada Katchamart11, Phonpen Akarawatcharangura Goo12, Lisa Traboco13, Yi-Ming Chen14, Parikshit Sen15, James Lilleker16, Arvind Nune17, John Pauling18, Ai Lyn Tan19, Nelly Ziade20, Marcin Milchert21, Abraham Edgar Gracia-Ramos22, Carlo Caballero23, COVAD Study Group24, Vikas Agarwal6, Rohit Aggarwal25, Latika Gupta26 and Chris Wincup1, 1King's College Hospital, London, United Kingdom, 2King's College Hospital, London, United Kingdom, 3King's College London, London, United Kingdom, 4Karolinska Institutet, Stockholm, Sweden, 5UNICAMP, Campinas, Brazil, 6Sanjay Gandhi Postgraduate Institute of Medical Sciences (SGPGIMS), Lucknow, India, 7Walter and Eliza Hall Institute, Melbourne, Australia, 8Byramjee Jeejeebhoy Government Medical College and Sassoon General Hospitals, Pune, India, 9Mymensingh Medical College, Faridpur, Bangladesh, 10Faculty of Medicine, Universiti Kebangsaan Malaysia, 56000, Cheras, Kuala Lumpur, Malaysia, 11Mahidol University, Bangkok, Thailand, 12Department of Medicine, Queen Savang Vadhana Memorial Hospital, Chonburi, Thailand, 13University of the Philippines - Manila, St Luke's Medical Center - Bonifacio Global City, Paranaque, Manila, Philippines, 14Taichung Veterans General Hospital, Taichung, Taiwan, 15Maulana Azad Medical College, 2-Bahadurshah Zafar Marg, New Delhi, Delhi-110002, India., Dalhi, India, 16Centre for Musculoskeletal Research, Division of Musculoskeletal and Dermatological Sciences, School of Biological Sciences, Faculty of Biology, Medicine and Health, Manchester Academic Health Science Centre, The University of Manchester, Manchester, UK; Manchester Centre for Clinical Neurosciences, Salford Royal NHS Foundation Trust, Salford, UK. Orcid ID: 0000-0002-9230-4137., Manchester, United Kingdom, 17Southport & Ormskirk NHS Foundation Trust, Liverpool, United Kingdom, 18North Bristol NHS Trust, Bristol, United Kingdom, 19University of Leeds, Leeds, United Kingdom, 20Saint-Joseph University, Beirut, Lebanon, 21Department of Internal Medicine, Rheumatology, Diabetology, Geriatrics and Clinical Immunology, Pomeranian Medical University in Szczecin, Szczecin, Poland, 22Department of Internal Medicine, General Hospital, National Medical Center "La Raza", Instituto Mexicano del Seguro Social, Av. Jacaranda S/N, Col. La Raza, Del. Azcapotzalco, C.P. 02990, Mexico City, Mexico, 23Universidad del Norte, Barranquilla, Colombia, 24-, -, 25University of Pittsburgh, Pittsburgh, PA, 26Royal Wolverhampton Trust, Wolverhampton/University of Manchester, United Kingdom
Background/Purpose: Regional disparities in the management of SLE are frequently described. Governance, funding, logistic barriers, and physician choice may be important determinants though data is scare from many underrepresented regions thus limiting our understanding and ability to appraise this. Steroids are a key contributor to damage and the use of steroid sparing medication has an important role in reducing these complications. In this study, we sought to evaluate global patterns in the treatment of SLE and identify the prevalence of steroid related comorbidities.
Methods: We identified SLE patients from the COVAD 2 database, an international study of over 20,000 respondents. Data collection included, demographics, comorbidities, treatment including; corticosteroids (CS), antimalarials, immunosuppressants (IS), cyclophosphamide and biologics. Country Human Development Index (HDI) classification, a composite index formulated by the United Nations to rank countries into tiers of development, was utilised. Statistical analysis comprised of Chi square for comparison of categorical values and t-test for comparison between groups. Significance was defined as p< 0.05.
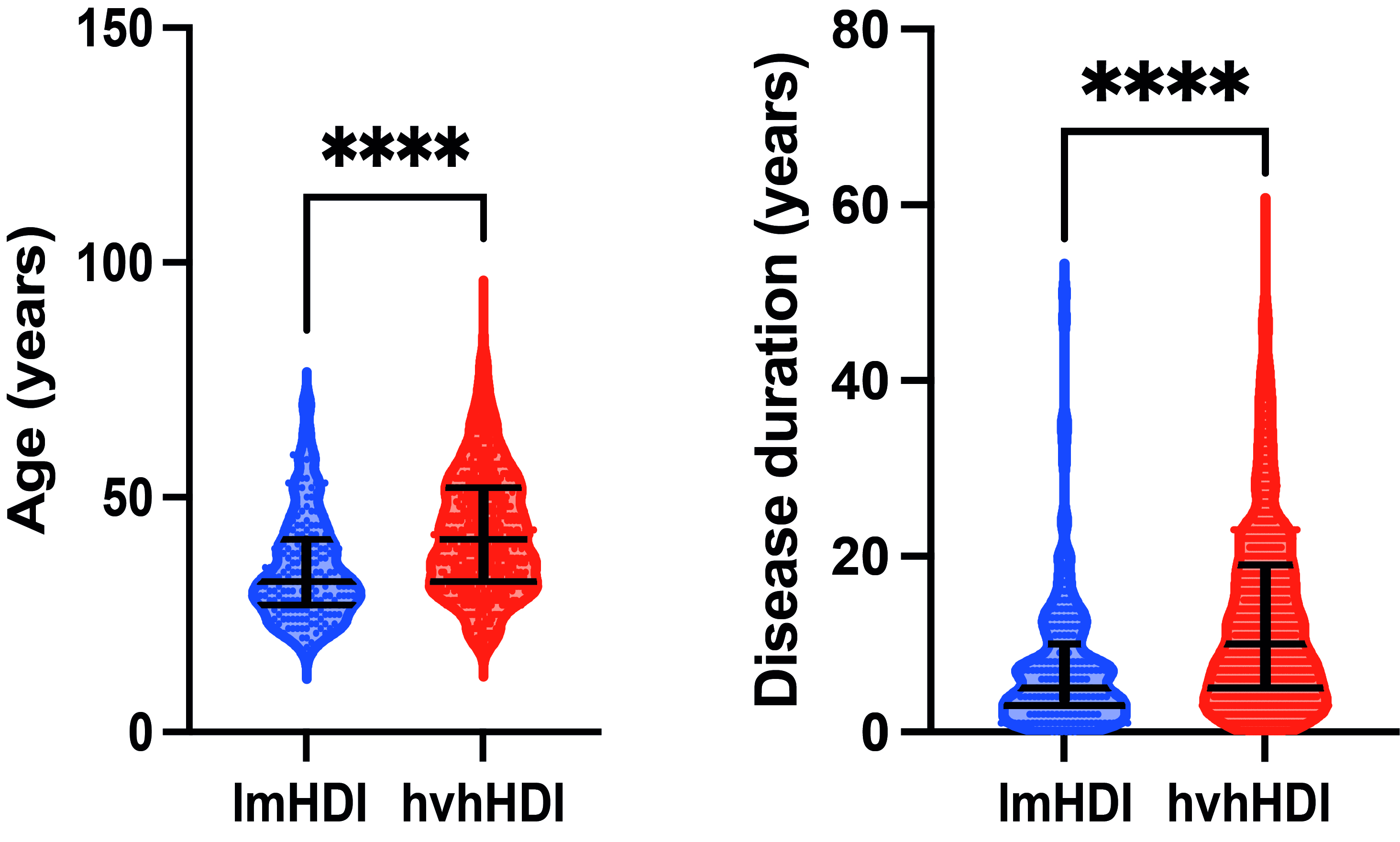
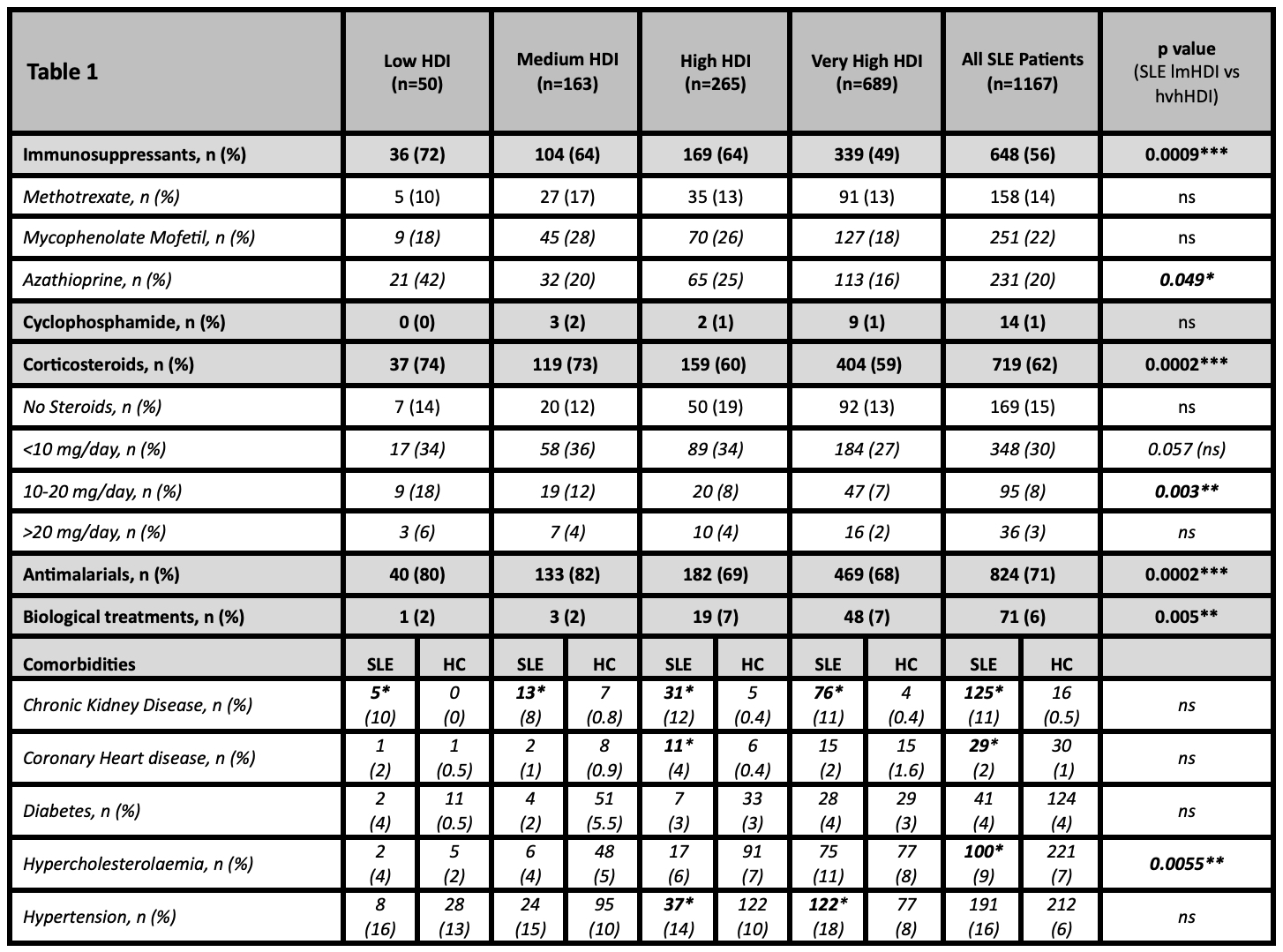
Results: A total of 1167 patients with SLE were included in analysis. As shown in Figure 1, patients from low/medium HDI (lmHDI) countries were significantly younger than those from high/very high HDI (hvhHDI) countries (median age 32, IQR 27-41 vs 41, IQR 32-52 years, p< 0.0001). In addition, disease duration was shorter in lmHDI countries (median 5, IQR 3-10 vs 10, IQR 5-19 years, p< 0.0001). As shown in Table 1, a higher proportion of SLE patients from lmHDI countries were on CS (73% vs 59%, p=0.0002), antimalarials (81% vs 68%, p=0.0002) and IS (66% vs 53%, p=0.0009) compared with patients from hvhHDI countries. Biologic use was more common in hvhHDI countries (7% vs 2%, p=0.0055). Comorbidity prevalence was similar between groups, however when adjusted for age, these were observed in younger patients from lmHDI countries than hvhHDI countries (with shorter disease durations), thus suggesting earlier steroid related complications. Patients with chronic kidney disease were significantly younger patients in lmHDI countries than hvhHDI countries (36.67 vs 44.64 years, p=0.015). Those with coronary artery disease (35.7 vs. 44.6 years, p=0.015) and hypertension (41.5 vs 49.8 years, p=0.003) were also younger in lmHDI countries.
Conclusion: In this large international study evaluating treatment and steroid related comorbidity incidence in SLE populations based on country HDI we identified several differences in pharmacological management globally. Whilst there was no significant difference in the incidence of comorbidities, these occurred in younger patients and earlier in the disease course in those from lmHDI countries. CS use was higher in lmHDI countries and may be a key contributor to the increased incidence in these comorbidities. Interestingly, Hydroxychloroquine, which has frequently been shown to reduce damage and cardiovascular comorbidities was more commonly used in lmHDI countries but these comorbidities persisted. Further studies are required to identify other factors contributing to this earlier development of comorbidities.
A. Holloway: None; S. Lee: None; E. Nikiphorou: AbbVie/Abbott, 6, Celltrion, 6, Eli Lilly, 6, fresenius, 6, Galapagos, 6, Gilead, 1, 6, Pfizer, 6, Sanofi, 6; I. Parodis: Amgen, 5, 6, AstraZeneca, 5, 6, Aurinia Pharmaceuticals, 5, 6, Bristol-Myers Squibb(BMS), 5, 6, Elli Lilly and Company, 5, 6, F. Hoffmann-La Roche AG, 5, 6, Gilead Sciences, 5, 6, GSK, 5, 6, Janssen Pharmaceuticals, 5, 6, Novartis, 5, 6, Otsuka Pharmaceutical, 5, 6; S. Appenzeller: None; N. R: None; J. Day: CSL limited, 5; M. Joshi: None; S. Saha: None; S. Shaharir: None; W. Katchamart: None; P. Akarawatcharangura Goo: None; L. Traboco: None; Y. Chen: None; P. Sen: None; J. Lilleker: None; A. Nune: None; J. Pauling: AstraZeneca, 2, Boehringer-Ingelheim, 2, IsoMab, 2, Janssen, 2, 6, Permeatus, 2, Sojournix Pharma, 2; A. Tan: Abbvie, 1, 6, Gilead, 6, Janssen, 6, Lilly, 6, Novartis, 6, Pfizer, 6, UCB, 6; N. Ziade: Abbvie, 6, Boehringer-Ingelheim, 6, Eli Lilly, 6, Janssen, 6, Newbridge, 6, Novartis, 6, Pfizer, 6, Pierre Fabre, 6, Roche, 6, sanofi, 6; M. Milchert: None; A. Gracia-Ramos: None; C. Caballero: None; C. Study Group: None; V. Agarwal: None; R. Aggarwal: Actigraph, 2, Alexion, 2, ANI Pharmaceuticals, 2, Argenx, 2, AstraZeneca, 2, Boehringer-Ingelheim, 2, 5, Bristol-Myers Squibb(BMS), 2, 5, CabalettaBio, 2, Capella Bioscience, 2, Corbus, 2, CSL Behring, 2, EMD Serono, 2, 5, Galapagos, 2, Horizon Therapeutics, 2, I-Cell, 2, Janssen, 2, 5, Kezar, 2, Kyverna, 2, Mallinckrodt, 5, Merck, 2, Octapharma, 2, Pfizer, 2, 5, Q32, 5, Roivant, 2, Sanofi, 2, Teva, 2; L. Gupta: None; C. Wincup: None.
Background/Purpose: Regional disparities in the management of SLE are frequently described. Governance, funding, logistic barriers, and physician choice may be important determinants though data is scare from many underrepresented regions thus limiting our understanding and ability to appraise this. Steroids are a key contributor to damage and the use of steroid sparing medication has an important role in reducing these complications. In this study, we sought to evaluate global patterns in the treatment of SLE and identify the prevalence of steroid related comorbidities.
Methods: We identified SLE patients from the COVAD 2 database, an international study of over 20,000 respondents. Data collection included, demographics, comorbidities, treatment including; corticosteroids (CS), antimalarials, immunosuppressants (IS), cyclophosphamide and biologics. Country Human Development Index (HDI) classification, a composite index formulated by the United Nations to rank countries into tiers of development, was utilised. Statistical analysis comprised of Chi square for comparison of categorical values and t-test for comparison between groups. Significance was defined as p< 0.05.
Results: A total of 1167 patients with SLE were included in analysis. As shown in Figure 1, patients from low/medium HDI (lmHDI) countries were significantly younger than those from high/very high HDI (hvhHDI) countries (median age 32, IQR 27-41 vs 41, IQR 32-52 years, p< 0.0001). In addition, disease duration was shorter in lmHDI countries (median 5, IQR 3-10 vs 10, IQR 5-19 years, p< 0.0001). As shown in Table 1, a higher proportion of SLE patients from lmHDI countries were on CS (73% vs 59%, p=0.0002), antimalarials (81% vs 68%, p=0.0002) and IS (66% vs 53%, p=0.0009) compared with patients from hvhHDI countries. Biologic use was more common in hvhHDI countries (7% vs 2%, p=0.0055). Comorbidity prevalence was similar between groups, however when adjusted for age, these were observed in younger patients from lmHDI countries than hvhHDI countries (with shorter disease durations), thus suggesting earlier steroid related complications. Patients with chronic kidney disease were significantly younger patients in lmHDI countries than hvhHDI countries (36.67 vs 44.64 years, p=0.015). Those with coronary artery disease (35.7 vs. 44.6 years, p=0.015) and hypertension (41.5 vs 49.8 years, p=0.003) were also younger in lmHDI countries.
Conclusion: In this large international study evaluating treatment and steroid related comorbidity incidence in SLE populations based on country HDI we identified several differences in pharmacological management globally. Whilst there was no significant difference in the incidence of comorbidities, these occurred in younger patients and earlier in the disease course in those from lmHDI countries. CS use was higher in lmHDI countries and may be a key contributor to the increased incidence in these comorbidities. Interestingly, Hydroxychloroquine, which has frequently been shown to reduce damage and cardiovascular comorbidities was more commonly used in lmHDI countries but these comorbidities persisted. Further studies are required to identify other factors contributing to this earlier development of comorbidities.

Figure 1. Patients from low/medium HDI (lmHDI) countries were significantly younger and had shorter disease duration than those from high/very high HDI (hvhHDI) countries (p<0.0001)

Table 1. Differences observed between patients from low/medium HDI (lmHDI) and high/very high HDI (hvhHDI) countries. Corticosteroid and anti-malarial treatment was more frequently used in the treatment of patients in low/medium HDI (lmHDI) countries, whilst biologic use was used more frequently in high/very high HDI (hvhHDI) countries.
A. Holloway: None; S. Lee: None; E. Nikiphorou: AbbVie/Abbott, 6, Celltrion, 6, Eli Lilly, 6, fresenius, 6, Galapagos, 6, Gilead, 1, 6, Pfizer, 6, Sanofi, 6; I. Parodis: Amgen, 5, 6, AstraZeneca, 5, 6, Aurinia Pharmaceuticals, 5, 6, Bristol-Myers Squibb(BMS), 5, 6, Elli Lilly and Company, 5, 6, F. Hoffmann-La Roche AG, 5, 6, Gilead Sciences, 5, 6, GSK, 5, 6, Janssen Pharmaceuticals, 5, 6, Novartis, 5, 6, Otsuka Pharmaceutical, 5, 6; S. Appenzeller: None; N. R: None; J. Day: CSL limited, 5; M. Joshi: None; S. Saha: None; S. Shaharir: None; W. Katchamart: None; P. Akarawatcharangura Goo: None; L. Traboco: None; Y. Chen: None; P. Sen: None; J. Lilleker: None; A. Nune: None; J. Pauling: AstraZeneca, 2, Boehringer-Ingelheim, 2, IsoMab, 2, Janssen, 2, 6, Permeatus, 2, Sojournix Pharma, 2; A. Tan: Abbvie, 1, 6, Gilead, 6, Janssen, 6, Lilly, 6, Novartis, 6, Pfizer, 6, UCB, 6; N. Ziade: Abbvie, 6, Boehringer-Ingelheim, 6, Eli Lilly, 6, Janssen, 6, Newbridge, 6, Novartis, 6, Pfizer, 6, Pierre Fabre, 6, Roche, 6, sanofi, 6; M. Milchert: None; A. Gracia-Ramos: None; C. Caballero: None; C. Study Group: None; V. Agarwal: None; R. Aggarwal: Actigraph, 2, Alexion, 2, ANI Pharmaceuticals, 2, Argenx, 2, AstraZeneca, 2, Boehringer-Ingelheim, 2, 5, Bristol-Myers Squibb(BMS), 2, 5, CabalettaBio, 2, Capella Bioscience, 2, Corbus, 2, CSL Behring, 2, EMD Serono, 2, 5, Galapagos, 2, Horizon Therapeutics, 2, I-Cell, 2, Janssen, 2, 5, Kezar, 2, Kyverna, 2, Mallinckrodt, 5, Merck, 2, Octapharma, 2, Pfizer, 2, 5, Q32, 5, Roivant, 2, Sanofi, 2, Teva, 2; L. Gupta: None; C. Wincup: None.



