Poster Session C
Epidemiology, health policy and outcomes
Session: (1895–1912) Measures & Measurement of Healthcare Quality Poster II
1905: Implementation of the Clinical Disease Activity Index to Optimize Treat-to-Target Management of Rheumatoid Arthritis at the University of North Carolina Hospitals Rheumatology Specialty Clinic
Tuesday, November 14, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- RI
Rumey Ishizawar, MD, PhD
University of North Carolina at Chapel Hill
Chapel Hill, NC, United StatesDisclosure(s): No financial relationships with ineligible companies to disclose
Abstract Poster Presenter(s)
Rumey Ishizawar1, Thuyvan Phan2 and Steven Takacs3, 1University of North Carolina at Chapel Hill, School of Medicine, Division of Rheumatology, Allergy, and Immunology, Thurston Arthritis Research Center, Chapel Hill, NC, 2University of North Carolina Hospitals Department of Pharmacy, Apex, NC, 3University of North Carolina Faculty Physicians, Chapel Hill, NC
Background/Purpose: The 2021 American College of Rheumatology (ACR) guidelines for the treatment of rheumatoid arthritis (RA) recommend a treat-to-target approach to optimize clinical outcomes. This approach requires frequent monitoring of disease activity using validated instruments and modification of treatment with the goal of reaching low disease activity or remission. The Centers for Medicare and Medicaid Services (CMS) and Rheumatology Informatics System for Effectiveness (RISE) registry have defined that patients with a diagnosis of RA should have an assessment using an ACR-preferred RA disease activity tool at ≥50% of encounters annually for the traditional Merit-based Incentive Payment System (MIPS). The University of North Carolina (UNC) Hospitals Rheumatology Specialty Clinic utilizes an electronic health record (EHR) with a flowsheet built to measure Clinical Disease Activity Index (CDAI); one such validated instrument. The purpose of this study was to understand use, limitations, and intervene to improve utilization of the CDAI among patients with RA.
Methods: Qualtrics™ surveys were distributed pre- and post-intervention to clinical care providers. Surveys queried knowledge of the ACR guidelines, CDAI implementation, and perceived barriers. Responses were anonymous and there was no identification if providers were trainees, faculty, or advance care partners. CDAI utilization was measured for the 6 months preceding the intervention and then 6 months post-intervention through the EHR. Interventions included 1) education to providers and care partners by a clinical pharmacist, 2) EHR daily reports for providers to capture relevant scheduled visits, 3) creation of a physical patient global assessment form, and 4) involvement of care partners in distributing and capturing the patient global assessment during patient triage. This project received IRB waiver as non-human subject research (UNC IRB 22-1967).
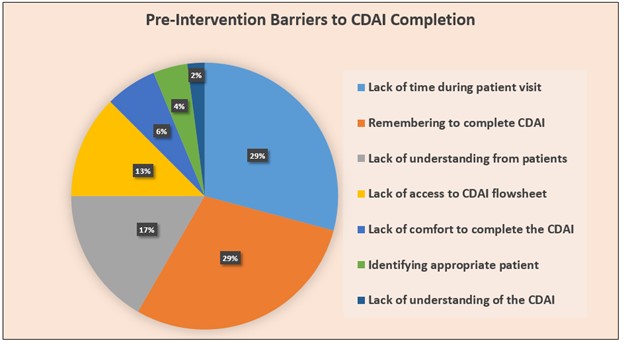
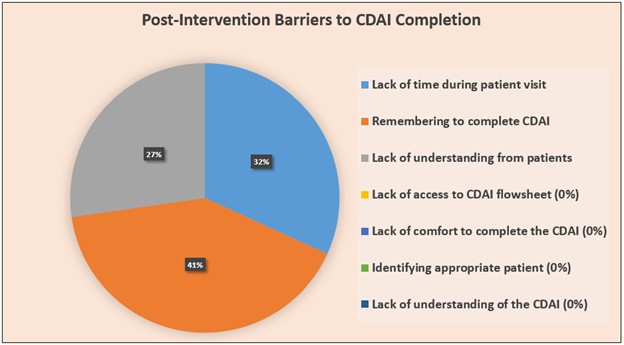
Results: On average, CDAI use in the 6 months prior to the intervention was 33%, improving to 58% post-intervention. We observed a slight lag for initial uptake as well as a plateau following intervention. Pre-intervention (n=17 providers) and post-intervention surveys (n=11 providers) indicated similar barriers, including: 1) lack of time during patient visit, 2) remembering to complete the CDAI, and 3) lack of understanding from patients to input CDAI global assessment. Following the intervention, provider barriers that were removed include: 1) lack of understanding of the CDAI, 2) lack of comfort in completing the CDAI, 3) access to the CDAI flowsheet in the EHR, and 4) identifying appropriate patients to complete the CDAI.
Conclusion: A simple educational intervention for both providers and care partners, as well as implementation of new workflow increased CDAI utilization in our clinic. Continuing assessment and repeated interventions are likely needed to optimize use. Identified barriers such as lack of time, remembering to complete CDAI, and patient understanding to complete global assessment are future targets for productive change.
.jpg)
R. Ishizawar: None; T. Phan: None; S. Takacs: None.
Background/Purpose: The 2021 American College of Rheumatology (ACR) guidelines for the treatment of rheumatoid arthritis (RA) recommend a treat-to-target approach to optimize clinical outcomes. This approach requires frequent monitoring of disease activity using validated instruments and modification of treatment with the goal of reaching low disease activity or remission. The Centers for Medicare and Medicaid Services (CMS) and Rheumatology Informatics System for Effectiveness (RISE) registry have defined that patients with a diagnosis of RA should have an assessment using an ACR-preferred RA disease activity tool at ≥50% of encounters annually for the traditional Merit-based Incentive Payment System (MIPS). The University of North Carolina (UNC) Hospitals Rheumatology Specialty Clinic utilizes an electronic health record (EHR) with a flowsheet built to measure Clinical Disease Activity Index (CDAI); one such validated instrument. The purpose of this study was to understand use, limitations, and intervene to improve utilization of the CDAI among patients with RA.
Methods: Qualtrics™ surveys were distributed pre- and post-intervention to clinical care providers. Surveys queried knowledge of the ACR guidelines, CDAI implementation, and perceived barriers. Responses were anonymous and there was no identification if providers were trainees, faculty, or advance care partners. CDAI utilization was measured for the 6 months preceding the intervention and then 6 months post-intervention through the EHR. Interventions included 1) education to providers and care partners by a clinical pharmacist, 2) EHR daily reports for providers to capture relevant scheduled visits, 3) creation of a physical patient global assessment form, and 4) involvement of care partners in distributing and capturing the patient global assessment during patient triage. This project received IRB waiver as non-human subject research (UNC IRB 22-1967).
Results: On average, CDAI use in the 6 months prior to the intervention was 33%, improving to 58% post-intervention. We observed a slight lag for initial uptake as well as a plateau following intervention. Pre-intervention (n=17 providers) and post-intervention surveys (n=11 providers) indicated similar barriers, including: 1) lack of time during patient visit, 2) remembering to complete the CDAI, and 3) lack of understanding from patients to input CDAI global assessment. Following the intervention, provider barriers that were removed include: 1) lack of understanding of the CDAI, 2) lack of comfort in completing the CDAI, 3) access to the CDAI flowsheet in the EHR, and 4) identifying appropriate patients to complete the CDAI.
Conclusion: A simple educational intervention for both providers and care partners, as well as implementation of new workflow increased CDAI utilization in our clinic. Continuing assessment and repeated interventions are likely needed to optimize use. Identified barriers such as lack of time, remembering to complete CDAI, and patient understanding to complete global assessment are future targets for productive change.
.jpg)
Figure 1. CDAI Completion Rate Over Time

Figure 2a. Pre-Intervention Barriers to CDAI Completion

Figure 2b. Post-Intervention Barriers to CDAI Completion
R. Ishizawar: None; T. Phan: None; S. Takacs: None.



