Poster Session C
Epidemiology, health policy and outcomes
Session: (1840–1861) Health Services Research Poster III
1849: Impact of the First Three Waves of the Covid-19 Pandemic on Everyday Restrictions and Clinical Care of Patients with Spondyloarthritis Across Europe - Results from the EuroSpA Collaboration
Tuesday, November 14, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- BM
Brigitte Michelsen, MD, PhD
Copenhagen Center for Arthritis Research (COPECARE), Copenhagen University Hospital Rigshospitalet
Copenhagen, DenmarkDisclosure information not submitted.
Abstract Poster Presenter(s)
Brigitte Michelsen1, Bente Glintborg2, Kim Lauper3, Bjorn Gudbjornsson4, Lykke Ørnbjerg5, Gerdur Maria Gröndal6, Karin Laas7, Sigrid Vorobjov8, Dan Nordstrom9, Heikki Relas9, Adrian Ciurea10, Burkhard Moeller11, Isabel Castrejon12, Lucia Otero-Valera13, Ziga Rotar14, Katja Perdan Pirkmajer15, Anne Gitte Loft16, Jakub Zavada17, Karel Pavelka18, Eirik Kristianslund19, Tore Kvien20, Marleen van de Sande21, Pasoon Hellamand22, Florenzo Iannone23, Roberto F. Caporali24, Ana Maria Rodrigues25, Maria Jose Santos26, Catalin Codreanu27, Corina Mogosan28, Merete Hetland5 and Mikkel Østergaard29, 1Rigshospitalet Glostrup; Diakonhjemmet Hospital; Sørlandet Hospital, Copenhagen, Denmark, 2Rigshospitalet Glostrup, University of Copenhagen, Virum, Denmark, 3Geneva University Hospitals, Genève, Switzerland, 4Centre for Rheumatology Research, University Hospital, Reykjavik, Iceland, 5Copenhagen Center for Arthritis Research, Rigshospitalet, Copenhagen, Denmark, 6Landspitali University Hospital; University of Iceland, Reykjavik, Iceland, 7Department of Rheumatology, East-Tallinn Central Hospital, Tallinn, Estonia, 8National Institute for Health Development, Tallinn, Estonia, 9Helsinki University Hospital, Helsinki, Finland, 10University Hospital Zurich, Zürich, Switzerland, 11Inselspital - University Hospital Bern, Bern, Switzerland, 12Hospital Universitario Gregorio Marañón, Madrid, Spain, 13Spanish Society of Rheumatology, Madrid, Spain, 14University Medical Centre Ljubljana, Ljubljana, Slovenia, 15University Medical Centre Ljubljana; University of Ljubljana, Ljubljana, Slovenia, 16Aarhus University, Horsens, Denmark, 17Institute of Rheumatology; Charles University, Prague, Czech Republic, 18Institut of Rheumatology and Department of Rheumatology, First Faculty of Medicine, Charles University, Praha, Czech Republic, 19Diakonhjemmet Hospital, Oslo, Norway, 20Center for Treatment of Rheumatic and Musculoskeletal Diseases (REMEDY), Diakonhjemmet Hospital, Oslo, Norway, 21Amsterdam UMC, University of Amsterdam, Department of Rheumatology & Clinical Immunology and Department of Experimental Immunology, Amsterdam Infection & Immunity Institute; Amsterdam Rheumatology & Immunology Center (ARC), Academic Medical Center, Amsterdam, Netherlands, 22Amsterdam University Medical Centers, University of Amsterdam, Amsterdam, Netherlands, 23Rheumatology Unit, Department of Precision and Regenerative Medicine and Ionian Area, University of Bari "Aldo Moro", Bari, Italy, 24Department of Clinical Sciences and Community Health, University of Milan, and Department of Rheumatology and Medical Sciences, ASST Gaetano Pini-CTO, Milano, Italy, 25Sociedade Portuguesa de Reumatologia; Nova Medical School; Hospital dos Lusíadas, Lisbon, Portugal, 26Hospital Garcia de Orta, Almada, Lisboa, Portugal, 27Center for Rheumatic Diseases, Bucharest, Romania, 28University of Medicine and Pharmacy, Bucharest, Romania, 29Copenhagen Center for Arthritis Research, Center for Rheumatology and Spine Diseases, Centre for Head and Orthopaedics, Rigshospitalet; University of Copenhagen, Copenhagen, Denmark
Background/Purpose: The Covid-19 pandemic constituted major challenges for health-care services worldwide. We aimed to compare Covid-19 restrictions across Europe during the first three waves of the pandemic, as well as the consultation and follow-up practices of patients with spondyloarthritis before and during the pandemic.
Methods: Rheumatologists completed a Research Electronic Data Capture (REDCap) survey in 13 observational registries in the European Spondyloarthritis Research Collaboration Network (EuroSpA) between July 1st and October 27,th 2022: ATTRA (Czech Republic), DANBIO (Denmark), ESRBTR (Estonia), ROB-FIN (Finland), ICEBIO (Iceland), GISEA (Italy), ARC (Netherlands), NOR-DMARD (Norway), Reuma.pt (Portugal), RRBR (Romania), biorx.si (Slovenia), BIOBADASER (Spain), and SCQM (Switzerland), answering questions on Covid-19 restrictions in their individual countries and the impact of Covid-19 on consultation and follow-up of routine care patients with spondyloarthritis in their registries. The survey covered the period from the start of the pandemic in the different countries, until 1st of March, 2022.
Results: The first case of Covid-19 in each country was reported between 31st of January 2020 in Spain and 4th of March 2020 in Slovenia. Use of facemask in public was mandatory in all countries during the peak of the pandemic, but use of filtering facemask (type P2 or P3) only in four countries (Table 1). All countries except for Iceland had a nationwide total lockdown. Most countries had nationwide curfews, which occasionally applied to specific periods throughout the day. All countries had social distancing varying between one and two meters from others in public. This was mandatory at some time-point during the pandemic in all countries, except in Finland and Portugal, where it was recommended.
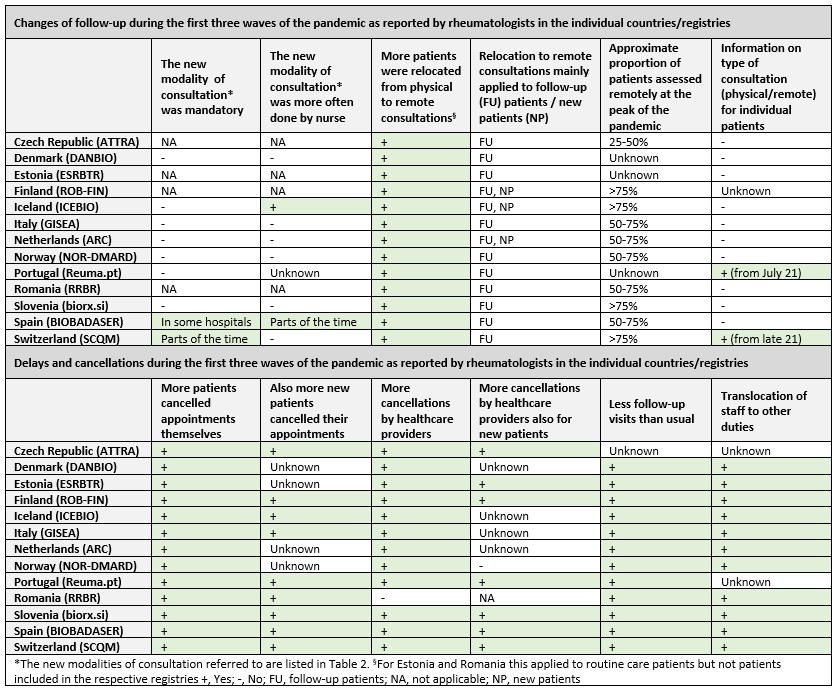
Before the pandemic, only four countries/registries had alternative modes of consultation in addition to the usual routine care physical consultations (Table 2). However, during the pandemic, nine registries initiated other modes of routine care consultations, the most common being phone consultations. The new modalities of consultations were not mandatory in any registry, except for mandatory non-urgent remote consultations in Switzerland during the first wave of the pandemic (phone or video, Table 3). In addition, in Spain, phone consultations and consultations between primary care physicians and rheumatologists were mandatory at some hospitals.
In every registry, rheumatologists reported an increased patient relocation from physical to remote consultations during the pandemic. At the peak of the pandemic, most of the patients were assessed remotely; however, this mainly applied to follow-up patients. More patients and healthcare providers canceled appointments during the pandemic than before. Furthermore, in all registries, staff was translocated to other duties and patients had fewer follow-up consultations than usual.
Conclusion: The Covid-19 pandemic had far-reaching consequences across Europe, not only due to nationwide restrictions, but also as a result of its negative impact on the accessibility of rheumatological care and follow-up.
.jpg)
.jpg)
B. Michelsen: Novartis, 5; B. Glintborg: AbbVie/Abbott, 5, Bristol-Myers Squibb(BMS), 5, Sandoz, 5; K. Lauper: Eli Lilly, 5, Pfizer, 2; B. Gudbjornsson: Nordic-Pharma, 6, Novartis, 2, 6; L. Ørnbjerg: Novartis, 5; G. Gröndal: None; K. Laas: None; S. Vorobjov: None; D. Nordstrom: AbbVie/Abbott, 2, BMS, 2, Lilly, 2, MSD, 2, Novartis, 2, Pfizer, 2, UCB, 2; H. Relas: None; A. Ciurea: None; B. Moeller: None; I. Castrejon: Bristol Myers Squibb, 1, 6, Galapagos, 2, GlaxoSmithKline, 1, 6, Lilly, 1, 6, Merck Sharp & Dohme, 6, Pfizer, 1, 2, 6; L. Otero-Valera: None; Z. Rotar: None; K. Perdan Pirkmajer: None; A. Loft: Ucb, 1, 6, 12, Congress participation; J. Zavada: None; K. Pavelka: Abbvie, 2, 6, Amgen, 2, 6, Bristol-Myers Squibb(BMS), 2, 6, Egis, 2, 6, MSD, 2, 6, Pfizer, 2, 6, Roche, 2, 6, UCB, 2, 6; E. Kristianslund: None; T. Kvien: AbbVie/Abbott, 1, 2, 6, Bristol-Myers Squibb(BMS), 5, Galapagos, 2, 5, Gilead, 2, grunenthal, 6, Janssen, 2, 6, Novartis, 5, Pfizer, 2, 5, sandoz, 2, 6, UCB, 2, 5, 6; M. van de Sande: AbbVie, 2, Eli Lilly, 5, Janssen, 6, Novartis, 2, 5, 6, UCB Pharma, 2, 5, 6; P. Hellamand: Novartis, 12, Research grant to employer (not to me); F. Iannone: Abbvie, 2, 5, BMS, 2, 5, Janssen, 2, 5, Lilly, 2, 5, MSD, 2, 5, Novartis, 2, 5, Pfizer, 2, 5, Roche, 2, 5, UCB, 2, 5; R. Caporali: AbbVie, 2, 6, Amgen, 2, 6, BMS, 2, 6, Celltrion, 2, 6, Fresenius Kabi, 2, Galapagos, 2, 6, Janssen, 2, 6, Lilly, 2, 6, MSD, 2, 6, Novartis, 2, 6, Pfizer, 2, 6, Roche, 2, Sandoz, 2, 6, UCB, 2, 6; A. Rodrigues: AbbVie/Abbott, 5, Amgen, 5, 6, Novartis, 5, Pfizer, 5; M. Santos: None; C. Codreanu: AbbVie/Abbott, 2, 6, Amgen, 1, 6, Boehringer-Ingelheim, 1, 6, Eli Lilly, 1, 6, Novartis, 1, 6, Pfizer, 1, 6; C. Mogosan: None; M. Hetland: AbbVie/Abbott, 1, 5, Bristol-Myers Squibb(BMS), 5, Danbio, 12, Chari of Danbio registry, Eli Lilly, 5, MEDAC, 6, Novartis, 5, Pfizer, 5, 6, Sandoz, 5, 6; M. Østergaard: AbbVie, 2, 5, 6, Amgen, 5, Boehringer-Ingelheim, 2, 6, Bristol-Myers Squibb(BMS), 2, 5, 6, Celgene, 2, 5, 6, Eli Lilly, 2, 6, Galapagos, 2, 6, Gilead, 2, 6, Hospira, 2, 6, Janssen, 2, 6, MEDAC, 6, Merck, 2, 5, 6, Novartis, 2, 5, 6, Novo Nordisk, 2, 6, Orion, 2, 6, Pfizer, 2, 6, Regeneron, 2, 6, Roche, 2, 6, Sandoz, 2, 6, Sanofi, 2, 6, UCB, 2, 6.
Background/Purpose: The Covid-19 pandemic constituted major challenges for health-care services worldwide. We aimed to compare Covid-19 restrictions across Europe during the first three waves of the pandemic, as well as the consultation and follow-up practices of patients with spondyloarthritis before and during the pandemic.
Methods: Rheumatologists completed a Research Electronic Data Capture (REDCap) survey in 13 observational registries in the European Spondyloarthritis Research Collaboration Network (EuroSpA) between July 1st and October 27,th 2022: ATTRA (Czech Republic), DANBIO (Denmark), ESRBTR (Estonia), ROB-FIN (Finland), ICEBIO (Iceland), GISEA (Italy), ARC (Netherlands), NOR-DMARD (Norway), Reuma.pt (Portugal), RRBR (Romania), biorx.si (Slovenia), BIOBADASER (Spain), and SCQM (Switzerland), answering questions on Covid-19 restrictions in their individual countries and the impact of Covid-19 on consultation and follow-up of routine care patients with spondyloarthritis in their registries. The survey covered the period from the start of the pandemic in the different countries, until 1st of March, 2022.
Results: The first case of Covid-19 in each country was reported between 31st of January 2020 in Spain and 4th of March 2020 in Slovenia. Use of facemask in public was mandatory in all countries during the peak of the pandemic, but use of filtering facemask (type P2 or P3) only in four countries (Table 1). All countries except for Iceland had a nationwide total lockdown. Most countries had nationwide curfews, which occasionally applied to specific periods throughout the day. All countries had social distancing varying between one and two meters from others in public. This was mandatory at some time-point during the pandemic in all countries, except in Finland and Portugal, where it was recommended.
Before the pandemic, only four countries/registries had alternative modes of consultation in addition to the usual routine care physical consultations (Table 2). However, during the pandemic, nine registries initiated other modes of routine care consultations, the most common being phone consultations. The new modalities of consultations were not mandatory in any registry, except for mandatory non-urgent remote consultations in Switzerland during the first wave of the pandemic (phone or video, Table 3). In addition, in Spain, phone consultations and consultations between primary care physicians and rheumatologists were mandatory at some hospitals.
In every registry, rheumatologists reported an increased patient relocation from physical to remote consultations during the pandemic. At the peak of the pandemic, most of the patients were assessed remotely; however, this mainly applied to follow-up patients. More patients and healthcare providers canceled appointments during the pandemic than before. Furthermore, in all registries, staff was translocated to other duties and patients had fewer follow-up consultations than usual.
Conclusion: The Covid-19 pandemic had far-reaching consequences across Europe, not only due to nationwide restrictions, but also as a result of its negative impact on the accessibility of rheumatological care and follow-up.
.jpg)
Table 1 Restrictions during the Covid-19 pandemic across European countries/registries
.jpg)
Table 2 Routine care consultations in the registries before and during the Covid-19 pandemic

Table 3 Changes of follow-up and delays in the registries during the Covid-19 pandemic
B. Michelsen: Novartis, 5; B. Glintborg: AbbVie/Abbott, 5, Bristol-Myers Squibb(BMS), 5, Sandoz, 5; K. Lauper: Eli Lilly, 5, Pfizer, 2; B. Gudbjornsson: Nordic-Pharma, 6, Novartis, 2, 6; L. Ørnbjerg: Novartis, 5; G. Gröndal: None; K. Laas: None; S. Vorobjov: None; D. Nordstrom: AbbVie/Abbott, 2, BMS, 2, Lilly, 2, MSD, 2, Novartis, 2, Pfizer, 2, UCB, 2; H. Relas: None; A. Ciurea: None; B. Moeller: None; I. Castrejon: Bristol Myers Squibb, 1, 6, Galapagos, 2, GlaxoSmithKline, 1, 6, Lilly, 1, 6, Merck Sharp & Dohme, 6, Pfizer, 1, 2, 6; L. Otero-Valera: None; Z. Rotar: None; K. Perdan Pirkmajer: None; A. Loft: Ucb, 1, 6, 12, Congress participation; J. Zavada: None; K. Pavelka: Abbvie, 2, 6, Amgen, 2, 6, Bristol-Myers Squibb(BMS), 2, 6, Egis, 2, 6, MSD, 2, 6, Pfizer, 2, 6, Roche, 2, 6, UCB, 2, 6; E. Kristianslund: None; T. Kvien: AbbVie/Abbott, 1, 2, 6, Bristol-Myers Squibb(BMS), 5, Galapagos, 2, 5, Gilead, 2, grunenthal, 6, Janssen, 2, 6, Novartis, 5, Pfizer, 2, 5, sandoz, 2, 6, UCB, 2, 5, 6; M. van de Sande: AbbVie, 2, Eli Lilly, 5, Janssen, 6, Novartis, 2, 5, 6, UCB Pharma, 2, 5, 6; P. Hellamand: Novartis, 12, Research grant to employer (not to me); F. Iannone: Abbvie, 2, 5, BMS, 2, 5, Janssen, 2, 5, Lilly, 2, 5, MSD, 2, 5, Novartis, 2, 5, Pfizer, 2, 5, Roche, 2, 5, UCB, 2, 5; R. Caporali: AbbVie, 2, 6, Amgen, 2, 6, BMS, 2, 6, Celltrion, 2, 6, Fresenius Kabi, 2, Galapagos, 2, 6, Janssen, 2, 6, Lilly, 2, 6, MSD, 2, 6, Novartis, 2, 6, Pfizer, 2, 6, Roche, 2, Sandoz, 2, 6, UCB, 2, 6; A. Rodrigues: AbbVie/Abbott, 5, Amgen, 5, 6, Novartis, 5, Pfizer, 5; M. Santos: None; C. Codreanu: AbbVie/Abbott, 2, 6, Amgen, 1, 6, Boehringer-Ingelheim, 1, 6, Eli Lilly, 1, 6, Novartis, 1, 6, Pfizer, 1, 6; C. Mogosan: None; M. Hetland: AbbVie/Abbott, 1, 5, Bristol-Myers Squibb(BMS), 5, Danbio, 12, Chari of Danbio registry, Eli Lilly, 5, MEDAC, 6, Novartis, 5, Pfizer, 5, 6, Sandoz, 5, 6; M. Østergaard: AbbVie, 2, 5, 6, Amgen, 5, Boehringer-Ingelheim, 2, 6, Bristol-Myers Squibb(BMS), 2, 5, 6, Celgene, 2, 5, 6, Eli Lilly, 2, 6, Galapagos, 2, 6, Gilead, 2, 6, Hospira, 2, 6, Janssen, 2, 6, MEDAC, 6, Merck, 2, 5, 6, Novartis, 2, 5, 6, Novo Nordisk, 2, 6, Orion, 2, 6, Pfizer, 2, 6, Regeneron, 2, 6, Roche, 2, 6, Sandoz, 2, 6, Sanofi, 2, 6, UCB, 2, 6.



