Poster Session C
Rheumatoid arthritis (RA)
Session: (2095–2140) RA – Diagnosis, Manifestations, and Outcomes Poster III
2103: Inflammation and Immunomodulatory Therapies Influence the Relationship Between ATP-binding Cassette Transporter A1 (ABCA1)-mediated Cholesterol Efflux and Coronary Atherosclerosis in Rheumatoid Arthritis
Tuesday, November 14, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall

George Karpouzas, MD (he/him/his)
Harbor-UCLA Medical Center
Torrance, CA, United StatesDisclosure information not submitted.
Abstract Poster Presenter(s)
George Karpouzas1, Bianca Papotti2, Sarah Ormseth3, Marcella Palumbo2, Elizabeth Hernandez3, Maria Pia Adorni4, Francesca Zimetti2, Matthew Budoff1 and Nicoletta Ronda2, 1Harbor-UCLA Medical Center, Torrance, CA, 2University of Parma, Department of Food and Drug, Parma, Italy, 3The Lundquist Institute, Torrance, CA, 4Department of Medicine and Surgery, Unit of Neuroscience, University of Parma, Parma, Italy
Background/Purpose: High-density lipoprotein (HDL) eliminates cholesterol from atherosclerotic lesions, a function known as cholesterol efflux capacity (CEC). ATP-binding-cassette A1 (ABCA1) membrane transporter initiates cholesterol transfer from plaque macrophages to pre-b HDL particles. Methotrexate and biologic disease modifying drugs (bDMARDs) are atheroprotective whereas corticosteroids are proatherogenic. We here evaluated the influence of inflammation and these treatments on ABCA1-CEC and its relationship with coronary atherosclerosis burden, progression and cardiovascular risk in patients with rheumatoid arthritis (RA).
Methods: Coronary atherosclerosis (noncalcified, partially or fully calcified plaque) was evaluated with computed tomography angiography in 140 patients without cardiovascular disease and reassessed in 99 after 6.9±0.4 years. Incident cardiovascular events were recorded. ABCA1-CEC was measured in J774 macrophages. Multivariable negative binomial and robust logistic regression evaluated associations of ABCA1-CEC with baseline plaque numbers and their progression at follow-up respectively. The moderating role of baseline inflammation, prednisone, methotrexate and bDMARD use on the relationship between ABCA1-CEC and cardiovascular risk was examined in multivariable Cox models including the respective moderators and their interaction term with ABCA1-CEC.
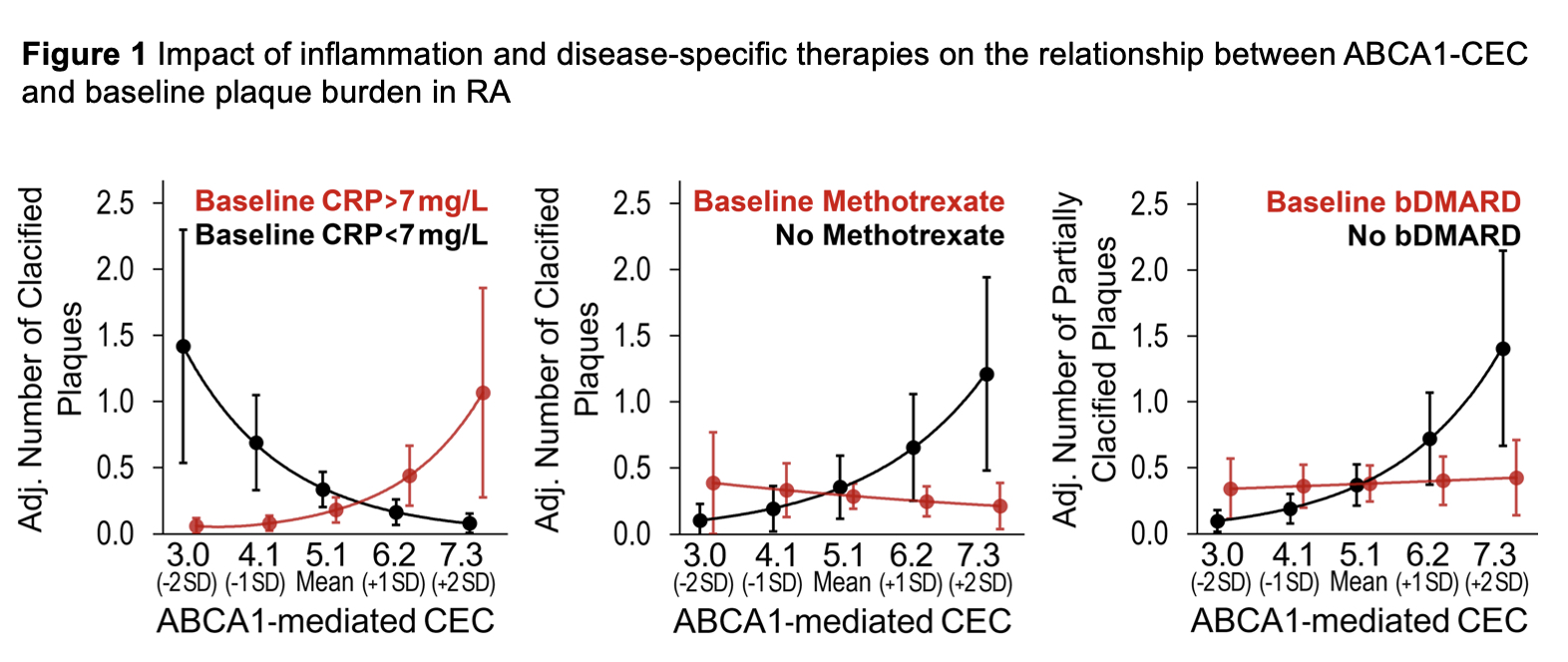
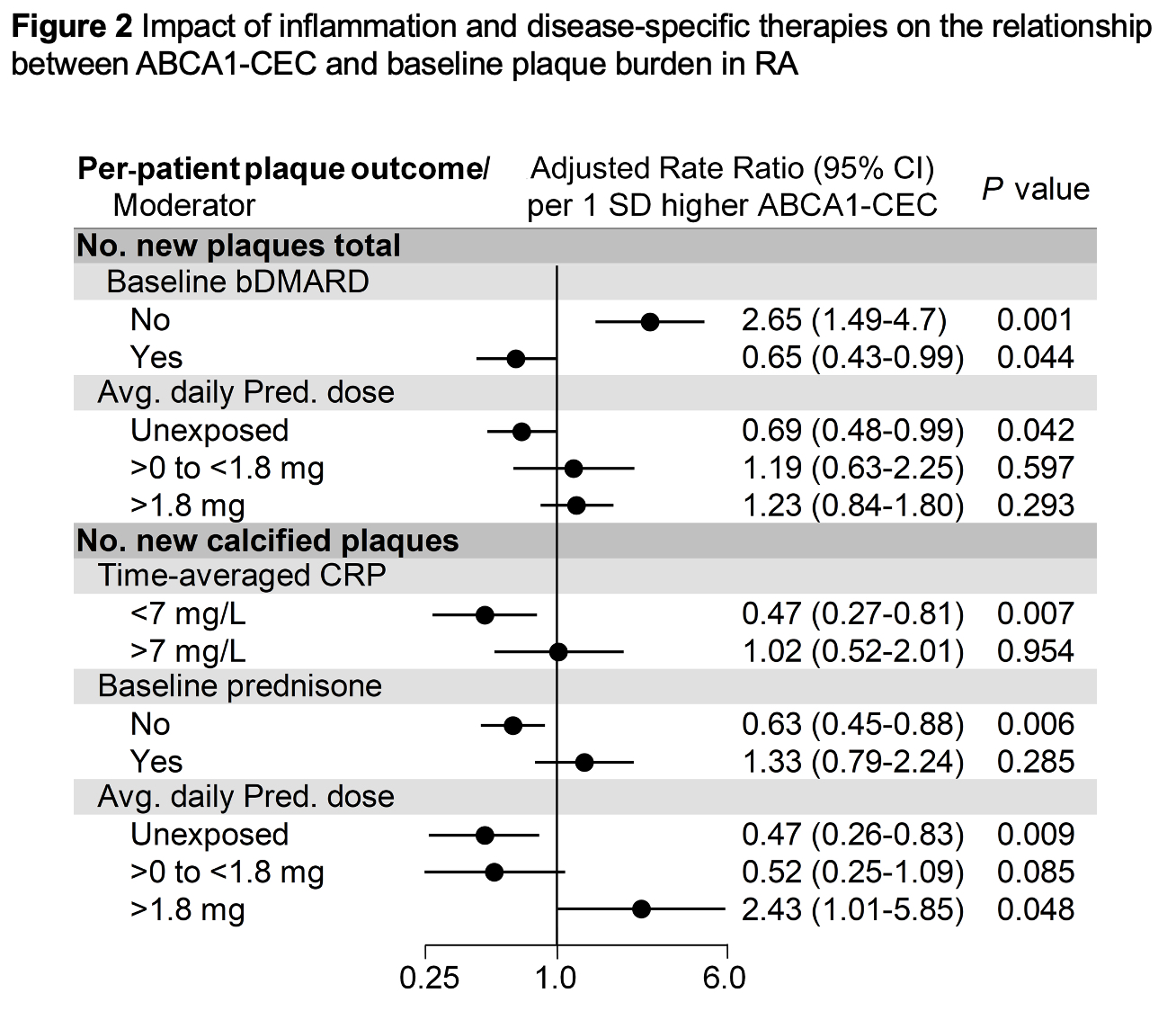
Results: ABCA1-CEC had no main effect on baseline atherosclerosis; prednisone use did not influence this relationship. Higher ABCA1-CEC (per standard deviation increment) associated with (i) more calcified plaques at baseline only in patients with CRP > 7mg/L (median) (p for interaction = 0.001), more calcified plaques only in methotrexate nonusers (p for interaction = 0.037), and more partially-calcified plaques only in bDMARD nonusers (p for interaction = 0.029, Figure 1); (ii) fewer new calcified plaques in patients with time-averaged CRP < 7mg/L (median) (p for interaction = 0.028, Figure 2); (iii) fewer new calcified plaques in baseline prednisone nonusers but not users (p for interaction = 0.021); (iv) fewer new total plaques in prednisone unexposed but not those exposed to prednisone during follow-up (p for interaction = 0.034), and fewer new calcified plaques in prednisone unexposed and more in those with high time-weighted average dose (p for interaction = 0.004); (v) more new plaques in baseline bDMARD nonusers and fewer in bDMARD users (p for interaction≤0.001); (vi) greater cardiovascular risk in baseline prednisone users but not nonusers (p for interaction = 0.027).
Conclusion: ABCA1-CEC attenuated coronary atherosclerosis burden and progression in patients with low baseline and cumulative inflammation and baseline methotrexate and bDMARD use. In contrast, ABCA1-CEC associated with plaque increase in corticosteroid users, methotrexate and bDMARD nonusers. While in well-treated, controlled disease ABCA1-CEC is atheroprotective, in uncontrolled RA its action may be masked or fails to counteract the proatherogenic state promoted by inflammation.
G. Karpouzas: Janssen, 1, Pfizer, 5, Scipher, 1; B. Papotti: None; S. Ormseth: None; M. Palumbo: None; E. Hernandez: None; M. Adorni: None; F. Zimetti: None; M. Budoff: None; N. Ronda: None.
Background/Purpose: High-density lipoprotein (HDL) eliminates cholesterol from atherosclerotic lesions, a function known as cholesterol efflux capacity (CEC). ATP-binding-cassette A1 (ABCA1) membrane transporter initiates cholesterol transfer from plaque macrophages to pre-b HDL particles. Methotrexate and biologic disease modifying drugs (bDMARDs) are atheroprotective whereas corticosteroids are proatherogenic. We here evaluated the influence of inflammation and these treatments on ABCA1-CEC and its relationship with coronary atherosclerosis burden, progression and cardiovascular risk in patients with rheumatoid arthritis (RA).
Methods: Coronary atherosclerosis (noncalcified, partially or fully calcified plaque) was evaluated with computed tomography angiography in 140 patients without cardiovascular disease and reassessed in 99 after 6.9±0.4 years. Incident cardiovascular events were recorded. ABCA1-CEC was measured in J774 macrophages. Multivariable negative binomial and robust logistic regression evaluated associations of ABCA1-CEC with baseline plaque numbers and their progression at follow-up respectively. The moderating role of baseline inflammation, prednisone, methotrexate and bDMARD use on the relationship between ABCA1-CEC and cardiovascular risk was examined in multivariable Cox models including the respective moderators and their interaction term with ABCA1-CEC.
Results: ABCA1-CEC had no main effect on baseline atherosclerosis; prednisone use did not influence this relationship. Higher ABCA1-CEC (per standard deviation increment) associated with (i) more calcified plaques at baseline only in patients with CRP > 7mg/L (median) (p for interaction = 0.001), more calcified plaques only in methotrexate nonusers (p for interaction = 0.037), and more partially-calcified plaques only in bDMARD nonusers (p for interaction = 0.029, Figure 1); (ii) fewer new calcified plaques in patients with time-averaged CRP < 7mg/L (median) (p for interaction = 0.028, Figure 2); (iii) fewer new calcified plaques in baseline prednisone nonusers but not users (p for interaction = 0.021); (iv) fewer new total plaques in prednisone unexposed but not those exposed to prednisone during follow-up (p for interaction = 0.034), and fewer new calcified plaques in prednisone unexposed and more in those with high time-weighted average dose (p for interaction = 0.004); (v) more new plaques in baseline bDMARD nonusers and fewer in bDMARD users (p for interaction≤0.001); (vi) greater cardiovascular risk in baseline prednisone users but not nonusers (p for interaction = 0.027).
Conclusion: ABCA1-CEC attenuated coronary atherosclerosis burden and progression in patients with low baseline and cumulative inflammation and baseline methotrexate and bDMARD use. In contrast, ABCA1-CEC associated with plaque increase in corticosteroid users, methotrexate and bDMARD nonusers. While in well-treated, controlled disease ABCA1-CEC is atheroprotective, in uncontrolled RA its action may be masked or fails to counteract the proatherogenic state promoted by inflammation.


G. Karpouzas: Janssen, 1, Pfizer, 5, Scipher, 1; B. Papotti: None; S. Ormseth: None; M. Palumbo: None; E. Hernandez: None; M. Adorni: None; F. Zimetti: None; M. Budoff: None; N. Ronda: None.



