Poster Session B
Spondyloarthritis (SpA) including psoriatic arthritis (PsA)
Session: (1383–1411) Spondyloarthritis Including Psoriatic Arthritis – Diagnosis, Manifestations, & Outcomes Poster II: Imaging & AS
1386: Opioid Prescription Rates in Patients with Ankylosing Spondylitis in the UK Between 2001-2020: An Electronic Health Records (EHR) Analysis
Monday, November 13, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- BD
Berk Degirmenci, MD
Boston University Medical Center
Boston, MA, United StatesDisclosure information not submitted.
Abstract Poster Presenter(s)
Huseyin Berk Degirmenci1, Christine E. Peloquin2, Sara Lodi2, Pedro Machado3, S. Reza Jafarzadeh4, Tuhina Neogi5, Lianne Gensler6, Maureen Dubreuil7 and Jean Liew2, 1St. Elizabeth's Medical Center, Boston, MA, 2Boston University, Boston, MA, 3Centre for Rheumatology & Department of Neuromuscular Diseases, University College London, London, UK. Department of Rheumatology, Northwick Park Hospital, London North West University Healthcare NHS trust, London, UK., London, United Kingdom, 4Boston University Chobanian & Avedisian School of Medicine, Boston, MA, 5Boston University School of Medicine, Boston, MA, 6University of California San Francisco, Department of Medicine, Division of Rheumatology, San Francisco, CA, 7Department of Rheumatology, Boston University School of Medicine, Milton, MA
Background/Purpose: Although current axial spondyloarthritis (axSpA) management guidelines do not address opioid use for chronic pain, available data show that opioid use is common among this population despite widespread uptake of effective biologic therapies, including TNF inhibitors and IL-17 inhibitors. We examined temporal trends in opioid prescription for individuals with ankylosing spondylitis (AS) in the United Kingdom (UK) before and after effective immunomodulatory agents have become available in the mid-2000s.
Methods: We used the IQVIA Medical Research Database (IMRD), incorporating data from THIN, a Cegedim database, an electronic health record database collected by general practitioners throughout the UK. We included adults aged 18-89 from 2000-2020 with AS, defined by ≥2 diagnostic Read codes (coded thesaurus of clinical terms used in the National Health Service since 1985) ≥7 days apart, and ≥1 year data prior to AS diagnosis. Baseline was defined as the date of the second AS diagnosis code. We excluded individuals with ≥2 rheumatoid arthritis Read codes prior to baseline. Individuals were followed until censoring at the date of codes for hospice or palliative care services, death, or end of the study (12/2020). Clinical characteristics included age, gender, baseline treatments for AS (defined within 1 year prior to the index date), and the presence of specific comorbidities (defined using Read codes any time prior to the index date). We calculated annual prescription rates from 2001-2020 for opioids and NSAIDs (comparator drug), as the number of prescriptions in a calendar year divided by person-time contributed by enrolled subjects meeting inclusion criteria for the year. Biologic prescription rates were not adequately captured in this general practitioner-entered database and thus not reported.
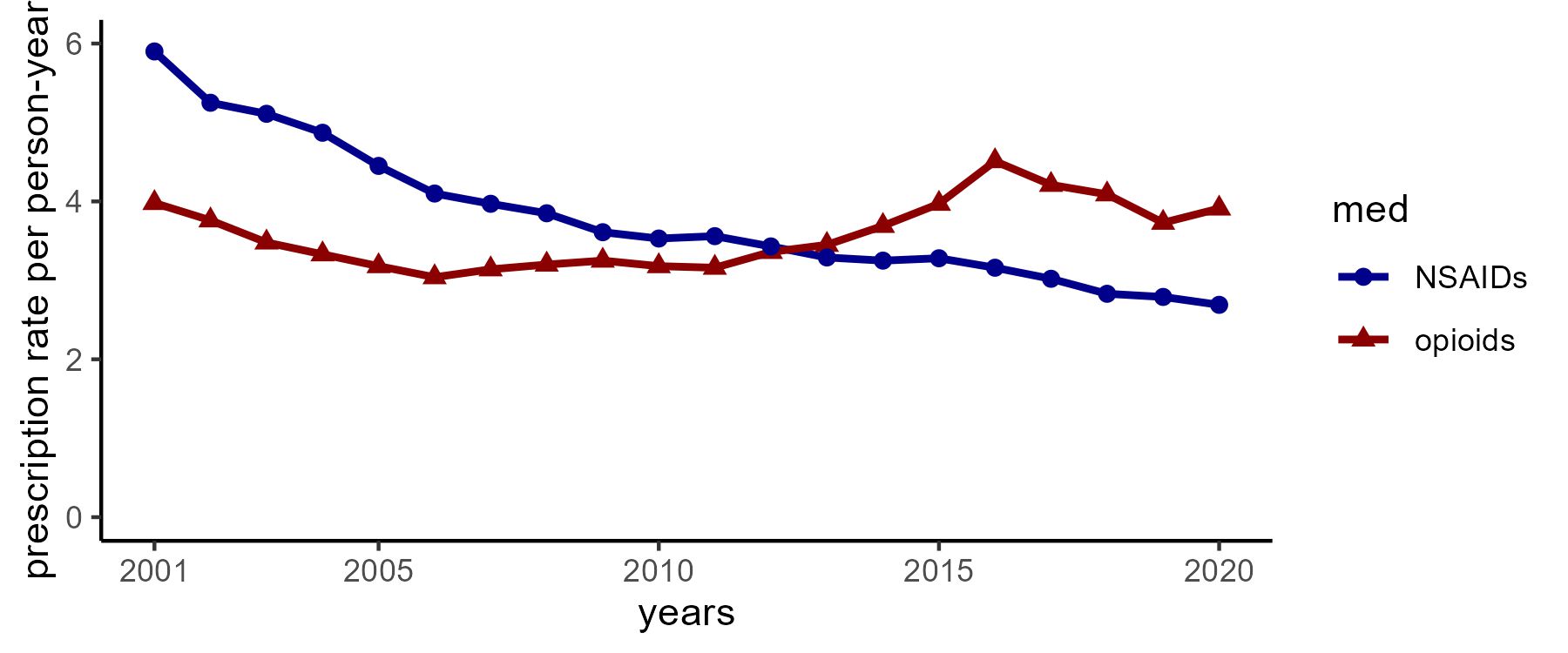
Results: We identified 2094 individuals with incident AS (mean age 47 years, 74.6% male, median follow-up time of 6.2 years with interquartile range 2.8-10.6 years). Of these, 395 (18.9%) had history of hypertension, 128 (6.1%) had history of cardiovascular disease, 72 (3.4%) had chronic kidney disease, and 70 (3.3%) had history of cancer. Baseline conventional synthetic DMARD use was present in 10.9% and glucocorticoid use in 10.4%. Opioid prescription rates were relatively unchanged over time with a range of 3.04-4.51 prescriptions per person-year (PY) from 2001-2020 whereas NSAID rates declined over time from 5.90 prescriptions per PY in 2001 to 2.69 per PY in 2020 (Figure).
Conclusion: In this UK-based AS cohort, opioid prescription rates remained relatively stable between 2001-2020 whereas NSAID use decreased over time consonant with the uptake of effective biologic therapies. Further, the rates of opioid prescriptions were similar in range to that of NSAID prescriptions. These findings may indicate inadequate pain control in AS despite the introduction and uptake of effective biologic agents.
H. Degirmenci: None; C. Peloquin: None; S. Lodi: None; P. Machado: AbbVie/Abbott, 2, 6, Bristol-Myers Squibb(BMS), 2, 6, Celgene, 2, 6, Eli Lilly, 2, 6, Galapagos, 2, 6, Janssen, 2, 6, Merck/MSD, 2, 6, Novartis, 2, 6, Orphazyme, 2, 6, Pfizer, 2, 6, Roche, 2, 6, UCB, 2, 6; S. Jafarzadeh: None; T. Neogi: None; L. Gensler: AbbVie, 2, Acelyrin, 2, Eli Lilly, 2, Fresenius Kabi, 2, Janssen, 2, Novartis, 2, 5, Pfizer, 2, UCB Pharma, 2, 5; M. Dubreuil: Amgen, 2, Pfizer, 5, UCB Pharma, 2; J. Liew: None.
Background/Purpose: Although current axial spondyloarthritis (axSpA) management guidelines do not address opioid use for chronic pain, available data show that opioid use is common among this population despite widespread uptake of effective biologic therapies, including TNF inhibitors and IL-17 inhibitors. We examined temporal trends in opioid prescription for individuals with ankylosing spondylitis (AS) in the United Kingdom (UK) before and after effective immunomodulatory agents have become available in the mid-2000s.
Methods: We used the IQVIA Medical Research Database (IMRD), incorporating data from THIN, a Cegedim database, an electronic health record database collected by general practitioners throughout the UK. We included adults aged 18-89 from 2000-2020 with AS, defined by ≥2 diagnostic Read codes (coded thesaurus of clinical terms used in the National Health Service since 1985) ≥7 days apart, and ≥1 year data prior to AS diagnosis. Baseline was defined as the date of the second AS diagnosis code. We excluded individuals with ≥2 rheumatoid arthritis Read codes prior to baseline. Individuals were followed until censoring at the date of codes for hospice or palliative care services, death, or end of the study (12/2020). Clinical characteristics included age, gender, baseline treatments for AS (defined within 1 year prior to the index date), and the presence of specific comorbidities (defined using Read codes any time prior to the index date). We calculated annual prescription rates from 2001-2020 for opioids and NSAIDs (comparator drug), as the number of prescriptions in a calendar year divided by person-time contributed by enrolled subjects meeting inclusion criteria for the year. Biologic prescription rates were not adequately captured in this general practitioner-entered database and thus not reported.
Results: We identified 2094 individuals with incident AS (mean age 47 years, 74.6% male, median follow-up time of 6.2 years with interquartile range 2.8-10.6 years). Of these, 395 (18.9%) had history of hypertension, 128 (6.1%) had history of cardiovascular disease, 72 (3.4%) had chronic kidney disease, and 70 (3.3%) had history of cancer. Baseline conventional synthetic DMARD use was present in 10.9% and glucocorticoid use in 10.4%. Opioid prescription rates were relatively unchanged over time with a range of 3.04-4.51 prescriptions per person-year (PY) from 2001-2020 whereas NSAID rates declined over time from 5.90 prescriptions per PY in 2001 to 2.69 per PY in 2020 (Figure).
Conclusion: In this UK-based AS cohort, opioid prescription rates remained relatively stable between 2001-2020 whereas NSAID use decreased over time consonant with the uptake of effective biologic therapies. Further, the rates of opioid prescriptions were similar in range to that of NSAID prescriptions. These findings may indicate inadequate pain control in AS despite the introduction and uptake of effective biologic agents.

FIGURE
Annual prescription rates of opioids and NSAIDs among people with ankylosing spondylitis in the IQVIA Medical Research Database 2001-2020
Annual prescription rates of opioids and NSAIDs among people with ankylosing spondylitis in the IQVIA Medical Research Database 2001-2020
H. Degirmenci: None; C. Peloquin: None; S. Lodi: None; P. Machado: AbbVie/Abbott, 2, 6, Bristol-Myers Squibb(BMS), 2, 6, Celgene, 2, 6, Eli Lilly, 2, 6, Galapagos, 2, 6, Janssen, 2, 6, Merck/MSD, 2, 6, Novartis, 2, 6, Orphazyme, 2, 6, Pfizer, 2, 6, Roche, 2, 6, UCB, 2, 6; S. Jafarzadeh: None; T. Neogi: None; L. Gensler: AbbVie, 2, Acelyrin, 2, Eli Lilly, 2, Fresenius Kabi, 2, Janssen, 2, Novartis, 2, 5, Pfizer, 2, UCB Pharma, 2, 5; M. Dubreuil: Amgen, 2, Pfizer, 5, UCB Pharma, 2; J. Liew: None.



