Poster Session B
Rheumatoid arthritis (RA)
Session: (1308–1344) RA – Treatment Poster II
1310: Comparison of Two Methotrexate Initiation Strategies in Rheumatoid Arthritis in Current Practice
Monday, November 13, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- PV
Paola Vidal Montal, -None-
Bellvitge University Hospital
Barcelona, SpainDisclosure information not submitted.
Abstract Poster Presenter(s)
Paola Vidal-Montal1, Alice Combier2, Alexia STEELANDT2, Marion THOMAS2, Javier Narvaez3, Joan Miquel Nolla1, Yannick ALLANORE4 and Jerome AVOUAC5, 1Department of Rheumatology, Hospital Universitario de Bellvitge, Barcelona, Spain, 2HOPITAL COCHIN AP-HP, Service de Rhumatologie, Paris, France, 3Hospital Universitario de Bellvitge, Barcelona, Spain, 4Université Paris Cité, Paris, France, 5Rheumatology A Department, Hôpital Cochin, AP-HP Centre - Université Paris Cité, Paris, France
Background/Purpose: To compare the efficacy and tolerance at 3 and 6 months of two methotrexate (MTX) initiation strategies in rheumatoid arthritis (RA).
Methods: Retrospective,monocentric,cross-sectionalstudy including patients with RA who initiated MTX as first-line therapy during the last 2 years according to one of the following 2 strategies: a "conventional" strategy (CS) defined by an initiation of oral MTX at a dose of 10-15 mg/week or an "aggressive" strategy (AS), defined by an initiation of subcutaneous (SC) MTX at a dose of 15 mg/week SC or >15 mg/week either orally or SC. Each strategy allowed the possibility to increase the doses and/or switch to the SC route at 3 months. Efficacy was assessed at 3 and 6 months using the DAS28-CRP. The tolerance of each strategy was also assessed at month 3 and 6.
Results: We included 101 patients (85 women) with a mean age of 55±12 years and disease duration of 5±6 months. The frequency of rheumatoid factors, anti-CCP antibodies and erosions was 83%, 81% and 38% respectively. 61 patients initiated MTX according to the CS, with an increase of dose and/or a switch to the SC route at 3 months for 31 patients, and 40 patients started treatment according to the AS, with an increase of dose and/or switch to the SC route at 3 months for 14 patients. There was no difference between these 2 groups in terms of age, gender, disease duration, antibody status, frequency of bone erosions, body mass index, comorbidities and disease activity at baseline. Efficacy at 3 months was significantly higher with the AS (reduction of the DAS28-CRP from 4.34±0.91 to 2.39±0.75, mean difference of 1.95±1.21, p< 0.001) compared to the CS (reduction of the DAS28-CRP from 4.09±0.62 to 2.88±0.73, mean difference of 1.21±0.90, p=0.12) (Figure 1). The improvement of tender/swollen joint counts, patient global assessment and CRP levels was also significantly more important at 3 months with the AS (Table 1). At 6 months, although the DAS28-CRP was similar in the 2 groups (Figure 1), less patients from the AS subgroup required an escalation to a targeted biologic/synthetic therapy compared to the CS (12/40, 30% vs. 29/61, 48%, p=0.073). The frequency of digestive side effects at 3 months was significantly lower in the AS (3/40, 7,5% vs. 16/61, 26%, p=0.021). The frequency of hepatic cytolysis at 3 month was higher in the AS (4/40, 10% vs. 1/61,1,6%, p=0.057). The frequency of asthenia at 3 months was similar in both groups (7/4, 18% vs. 6/61, 10%, p=0.25). Only one infection was observed in the CS and no hematological side effect was recorded. At 6 months, the cumulative incidence of side effects was 23% with the AS compared to 46% with the CS (p=0.015). Only one treatment discontinuation was noted in the AS subgroup vs. 9 in the CS subgroup (p=0.042).
Conclusion: This study suggests that it is possible to use a more aggressive initiation strategy of MTX in RA in routine clinical practice. This strategy allows to obtain an earlier clinical response and it is associated with a better tolerance than the conventional strategy. These results need to be confirmed in prospective studies.
.jpg)
Table 1: Evaluation of efficacy parameter at 3 months according to the methotrexate initiation strategy
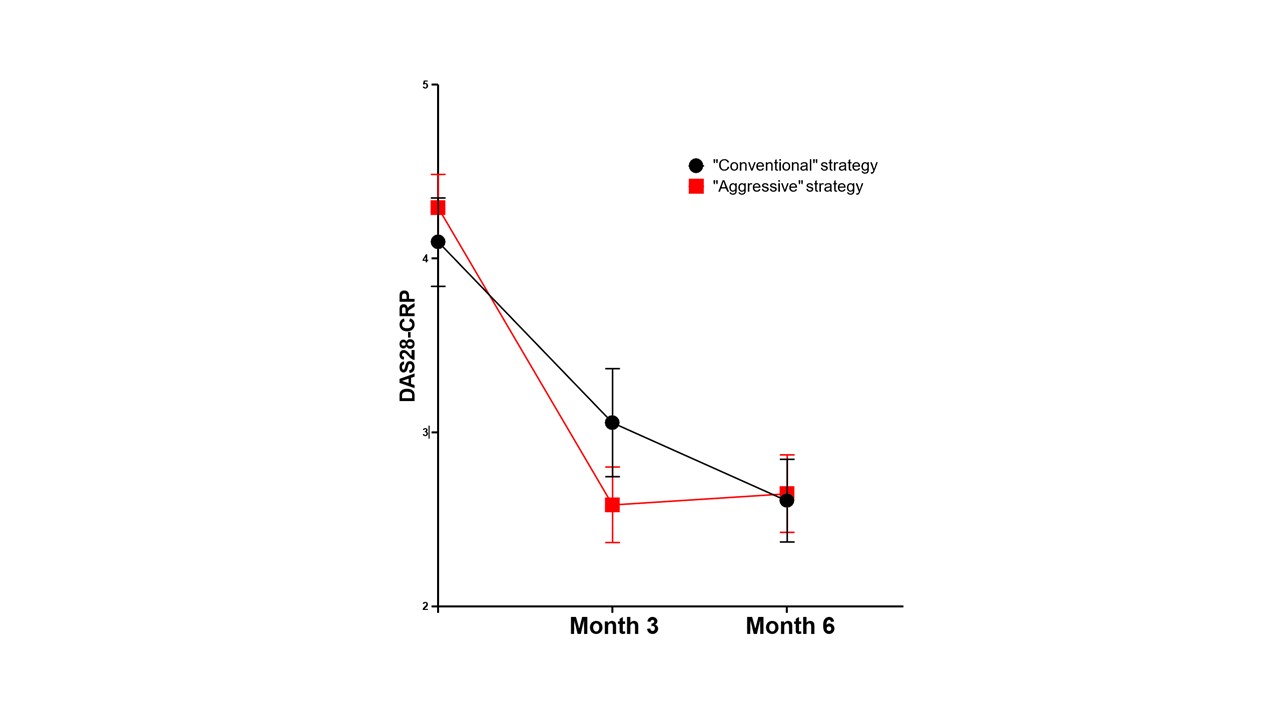
Figure 1: Evolution at 3 and 6 months of DAS28-CRP index according to the methotrexate initiation strategy
P. Vidal-Montal: None; A. Combier: None; A. STEELANDT: None; M. THOMAS: None; J. Narvaez: None; J. Nolla: None; Y. ALLANORE: AbbVie/Abbott, 2, Alpine Immunoscience, 5, AstraZeneca, 2, Bayer, 2, Boehringer-Ingelheim, 2, Janssen, 2, Medsenic, 2, 5, Mylan, 2, OSE Immunotherapeutics, 5, Prometeus, 2, Roche, 2, Sanofi, 2; J. AVOUAC: AbbVie, 1, 2, 4, 6, BMS, 4, 5, 6, Fresenius Kabi, 4, 5, Galapagos, 1, 2, 4, 6, Lilly, 6, Novartis, 5, 6, Pfizer, 5, 6, Sanofi, 4, 6.
Background/Purpose: To compare the efficacy and tolerance at 3 and 6 months of two methotrexate (MTX) initiation strategies in rheumatoid arthritis (RA).
Methods: Retrospective,monocentric,cross-sectionalstudy including patients with RA who initiated MTX as first-line therapy during the last 2 years according to one of the following 2 strategies: a "conventional" strategy (CS) defined by an initiation of oral MTX at a dose of 10-15 mg/week or an "aggressive" strategy (AS), defined by an initiation of subcutaneous (SC) MTX at a dose of 15 mg/week SC or >15 mg/week either orally or SC. Each strategy allowed the possibility to increase the doses and/or switch to the SC route at 3 months. Efficacy was assessed at 3 and 6 months using the DAS28-CRP. The tolerance of each strategy was also assessed at month 3 and 6.
Results: We included 101 patients (85 women) with a mean age of 55±12 years and disease duration of 5±6 months. The frequency of rheumatoid factors, anti-CCP antibodies and erosions was 83%, 81% and 38% respectively. 61 patients initiated MTX according to the CS, with an increase of dose and/or a switch to the SC route at 3 months for 31 patients, and 40 patients started treatment according to the AS, with an increase of dose and/or switch to the SC route at 3 months for 14 patients. There was no difference between these 2 groups in terms of age, gender, disease duration, antibody status, frequency of bone erosions, body mass index, comorbidities and disease activity at baseline. Efficacy at 3 months was significantly higher with the AS (reduction of the DAS28-CRP from 4.34±0.91 to 2.39±0.75, mean difference of 1.95±1.21, p< 0.001) compared to the CS (reduction of the DAS28-CRP from 4.09±0.62 to 2.88±0.73, mean difference of 1.21±0.90, p=0.12) (Figure 1). The improvement of tender/swollen joint counts, patient global assessment and CRP levels was also significantly more important at 3 months with the AS (Table 1). At 6 months, although the DAS28-CRP was similar in the 2 groups (Figure 1), less patients from the AS subgroup required an escalation to a targeted biologic/synthetic therapy compared to the CS (12/40, 30% vs. 29/61, 48%, p=0.073). The frequency of digestive side effects at 3 months was significantly lower in the AS (3/40, 7,5% vs. 16/61, 26%, p=0.021). The frequency of hepatic cytolysis at 3 month was higher in the AS (4/40, 10% vs. 1/61,1,6%, p=0.057). The frequency of asthenia at 3 months was similar in both groups (7/4, 18% vs. 6/61, 10%, p=0.25). Only one infection was observed in the CS and no hematological side effect was recorded. At 6 months, the cumulative incidence of side effects was 23% with the AS compared to 46% with the CS (p=0.015). Only one treatment discontinuation was noted in the AS subgroup vs. 9 in the CS subgroup (p=0.042).
Conclusion: This study suggests that it is possible to use a more aggressive initiation strategy of MTX in RA in routine clinical practice. This strategy allows to obtain an earlier clinical response and it is associated with a better tolerance than the conventional strategy. These results need to be confirmed in prospective studies.
.jpg)
Table 1: Evaluation of efficacy parameter at 3 months according to the methotrexate initiation strategy

Figure 1: Evolution at 3 and 6 months of DAS28-CRP index according to the methotrexate initiation strategy
P. Vidal-Montal: None; A. Combier: None; A. STEELANDT: None; M. THOMAS: None; J. Narvaez: None; J. Nolla: None; Y. ALLANORE: AbbVie/Abbott, 2, Alpine Immunoscience, 5, AstraZeneca, 2, Bayer, 2, Boehringer-Ingelheim, 2, Janssen, 2, Medsenic, 2, 5, Mylan, 2, OSE Immunotherapeutics, 5, Prometeus, 2, Roche, 2, Sanofi, 2; J. AVOUAC: AbbVie, 1, 2, 4, 6, BMS, 4, 5, 6, Fresenius Kabi, 4, 5, Galapagos, 1, 2, 4, 6, Lilly, 6, Novartis, 5, 6, Pfizer, 5, 6, Sanofi, 4, 6.



