Poster Session C
Epidemiology, health policy and outcomes
Session: (2019–2038) Patient Outcomes, Preferences, & Attitudes Poster III
2026: Developing a Guided Joint Self-Exam for Rheumatoid Arthritis Patients to Use in Telehealth-Delivered Care
Tuesday, November 14, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
.png)
Swamy Venuturupalli, MD
Attune Health
Beverly Hills, CA, United StatesDisclosure information not submitted.
Abstract Poster Presenter(s)
Kelly Gavigan1, David Curtis2, Jeffrey Curtis3, W. Benjamin Nowell4, Danielle Ali5, Neelkamal Soares6, John Cush7, Rebecca Grainger8, Manas Jinka9, Sandeep Sodhi10, Natalie Fortune9 and Swamy Venuturupalli9, 1Global Healthy Living Foundation, Upper Nyack, NY, 2Global Healthy Living Foundation, San Francisco, CA, 3University of Alabama at Birmingham, Birmingham, AL, 4Global Healthy Living Foundation, Nyack, NY, 5Global Healthy Living Foundation, Upper Nayack, NY, 6Western Mich Univ Homer Stryker MD Sch of Medicine, Kalamazoo, MI, 7University of Texas Southwestern Medical School, Dallas, TX, 8University of Otago, Wellington, New Zealand, 9Attune Health, Beverly Hills, CA, 10Illumination Health, Hoover, AL
Background/Purpose: Telehealth services and, increasingly, remote therapeutic monitoring, can be used to enable the continuum of clinical care in out-of-office settings. This remotely provided care can increase personalization of healthcare by using patient-relevant data, but lack of opportunity for in-person examination may remain a barrier. Our objective was to develop and pilot a smartphone application (app)-based program to teach patients to perform guided self-examination to measure the disease activity of their rheumatoid arthritis (RA), to facilitate remote care.
Methods: An in-app tool to instruct patients with RA to conduct a self-guided 28 tender and swollen joint count (28-TJC, 28-SJC) was developed for the ArthritisPower research registry app based on previously developed videos1. An instructional script of on-screen messaging and animations was iteratively refined by the study team of physicians, researchers, patients, and patient advocates. A storyboard of the final script was user tested with ArthritisPower members living with RA. The tool was then piloted in the ArthritisPower app by RA patients in a community rheumatology practice to gain feedback on the user interface and user experience. Participation in the pilot consisted of registering for ArthritisPower, completing six physical and social health patient reported outcomes (PROs) (PROMIS measures for Pain Interference, Physical Function, Fatigue, and Satisfaction with Participation in Discretionary Activities; RADAI-5; and OMERACT RA Flare), and completing the guided, self-conducted 28-TJC, 28-SJC. The in-app assessment was compared to in-person assessment by a rheumatologist within 6 days of a scheduled office visit.
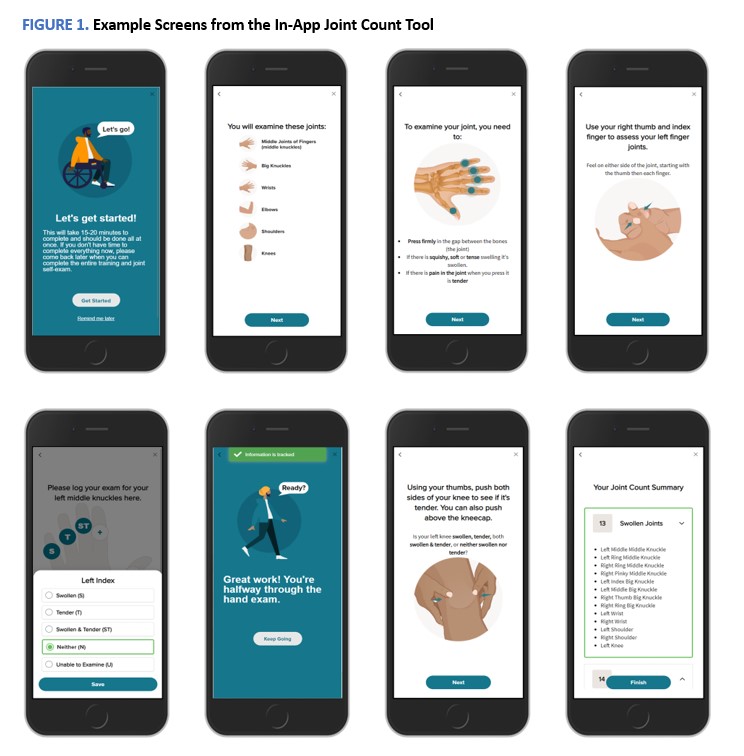
Results: Five RA patients participated in the pilot testing. The mean (SD) time to completion for the PROs and joint self-assessment tool was 18.3 (3.5) minutes, with the PROs taking 5.4 (0.9) minutes on average and the joint self-assessment taking 12.9 (3.5) minutes on average to complete. Based on this pilot, edits were made to the tool. These included simplifying the language in the registration process, clarifying what was required to successfully complete participation and the expected time commitment, clarifying concepts of tender and swollen joints, adding inspirational progress messaging, and refining the instructional tool. Example screens from the in-app module can be found in Figure 1.
Conclusion: Piloting in real-world settings highlighted areas for change that are hoped to increase successful participation, such as simplifying language, clarifying requirements, and adding motivational messaging. The refined tool is currently being deployed in a larger study to evaluate the accuracy and utility of patient-conducted TJC and SJC for guiding RA management decisions. 1Grainger, et al. ACR Open Rheum. 2020;2(12):705-709.
K. Gavigan: Global Healthy Living Foundation, 3; D. Curtis: Global Healthy Living Foundation, 3; J. Curtis: AbbVie, 2, 5, Amgen, 2, 5, Bristol-Myers Squibb, 2, 5, CorEvitas, 2, 5, Eli Lilly and Company, 2, 5, Janssen, 2, 5, Myriad, 2, 5, Novartis, 2, 5, Pfizer, 2, 5, Sanofi, 2, 5, UCB, 2, 5; W. Nowell: AbbVie/Abbott, 2, 5, Amgen, 5, Janssen, 2, 5, Scipher Medicine, 5; D. Ali: Global Healthy Living Foundation, 3; N. Soares: None; J. Cush: None; R. Grainger: AbbVie, 2, 6, Cornerstones, 6, Janssen, 6, Novartis, 2, Pfizer, 6; M. Jinka: None; S. Sodhi: None; N. Fortune: None; S. Venuturupalli: None.
Background/Purpose: Telehealth services and, increasingly, remote therapeutic monitoring, can be used to enable the continuum of clinical care in out-of-office settings. This remotely provided care can increase personalization of healthcare by using patient-relevant data, but lack of opportunity for in-person examination may remain a barrier. Our objective was to develop and pilot a smartphone application (app)-based program to teach patients to perform guided self-examination to measure the disease activity of their rheumatoid arthritis (RA), to facilitate remote care.
Methods: An in-app tool to instruct patients with RA to conduct a self-guided 28 tender and swollen joint count (28-TJC, 28-SJC) was developed for the ArthritisPower research registry app based on previously developed videos1. An instructional script of on-screen messaging and animations was iteratively refined by the study team of physicians, researchers, patients, and patient advocates. A storyboard of the final script was user tested with ArthritisPower members living with RA. The tool was then piloted in the ArthritisPower app by RA patients in a community rheumatology practice to gain feedback on the user interface and user experience. Participation in the pilot consisted of registering for ArthritisPower, completing six physical and social health patient reported outcomes (PROs) (PROMIS measures for Pain Interference, Physical Function, Fatigue, and Satisfaction with Participation in Discretionary Activities; RADAI-5; and OMERACT RA Flare), and completing the guided, self-conducted 28-TJC, 28-SJC. The in-app assessment was compared to in-person assessment by a rheumatologist within 6 days of a scheduled office visit.
Results: Five RA patients participated in the pilot testing. The mean (SD) time to completion for the PROs and joint self-assessment tool was 18.3 (3.5) minutes, with the PROs taking 5.4 (0.9) minutes on average and the joint self-assessment taking 12.9 (3.5) minutes on average to complete. Based on this pilot, edits were made to the tool. These included simplifying the language in the registration process, clarifying what was required to successfully complete participation and the expected time commitment, clarifying concepts of tender and swollen joints, adding inspirational progress messaging, and refining the instructional tool. Example screens from the in-app module can be found in Figure 1.
Conclusion: Piloting in real-world settings highlighted areas for change that are hoped to increase successful participation, such as simplifying language, clarifying requirements, and adding motivational messaging. The refined tool is currently being deployed in a larger study to evaluate the accuracy and utility of patient-conducted TJC and SJC for guiding RA management decisions. 1Grainger, et al. ACR Open Rheum. 2020;2(12):705-709.

FIGURE 1. Example Screens from the In-App Joint Count Tool
K. Gavigan: Global Healthy Living Foundation, 3; D. Curtis: Global Healthy Living Foundation, 3; J. Curtis: AbbVie, 2, 5, Amgen, 2, 5, Bristol-Myers Squibb, 2, 5, CorEvitas, 2, 5, Eli Lilly and Company, 2, 5, Janssen, 2, 5, Myriad, 2, 5, Novartis, 2, 5, Pfizer, 2, 5, Sanofi, 2, 5, UCB, 2, 5; W. Nowell: AbbVie/Abbott, 2, 5, Amgen, 5, Janssen, 2, 5, Scipher Medicine, 5; D. Ali: Global Healthy Living Foundation, 3; N. Soares: None; J. Cush: None; R. Grainger: AbbVie, 2, 6, Cornerstones, 6, Janssen, 6, Novartis, 2, Pfizer, 6; M. Jinka: None; S. Sodhi: None; N. Fortune: None; S. Venuturupalli: None.



