Poster Session C
Pain syndromes, fibromyalgia and regional musculoskeletal disorders
Session: (1827–1839) Fibromyalgia & Other Clinical Pain Syndromes Poster
1834: Evaluation of Fibromyalgia, Clinical/Serologic Activity, and Patient Reported Outcomes in a Racially/Ethnically Diverse SLE Patient Cohort
Tuesday, November 14, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- KC
Kelly Corbitt, DO
University of Miami Miller School of Medicine
El Portal, FL, United StatesDisclosure information not submitted.
Abstract Poster Presenter(s)
Kelly Corbitt1, Philip Carlucci2, Brooke Cohen3, Mala Masson3, H Michael Belmont4, Amit Saxena5, Chung-E Tseng3, Jing Wang3, Kamil Barbour6, Jill Buyon7, Peter Izmirly2 and Heather Gold3, 1New York University, El Portal, FL, 2New York University School of Medicine, New York, NY, 3New York University, New York, NY, 4NYU School of Medicine, New York, NY, 5New York University Grossman School of Medicine, Rheumatology, New York, NY, 6Centers for Disease Control and Prevention, Atlanta, GA, 7NYU Grossman School of Medicine, New York, NY
Background/Purpose: SLE patients have a higher prevalence of fibromyalgia (FM) than the general population. FM symptoms, such as nociplastic pain and fatigue, are known to cause discordance in how patients and physicians view well-controlled SLE, and these variables impact quality of life in ways not captured by traditional disease activity scores. There is a paucity of objective data surrounding the clinical interplay of these two diseases. Given this, and emerging evidence suggesting the contribution of autoimmunity to FM, this study evaluated FM clinically, serologically, and via patient reported outcomes in a multi-racial/ethnic SLE patient cohort.
Methods: Patients from an established lupus cohort were screened for FM using the 2016 FM classification criteria during an in-person rheumatologist visit. Hybrid SELENA-SLEDAI scores, SLE classification criteria, medications, and SLICC damage index were evaluated. A portion of the patients who reported any chronic pain also completed 8 PROMIS measures. Clinical and serologic activity was compared in patients with and without FM, as well as T-score means of PROMIS measures between FM and non-FM chronic pain patients. For T-score means significantly different between groups, linear regression models adjusting for age, race/ethnicity, SELENA-SLEDAI score, and steroid use were estimated.
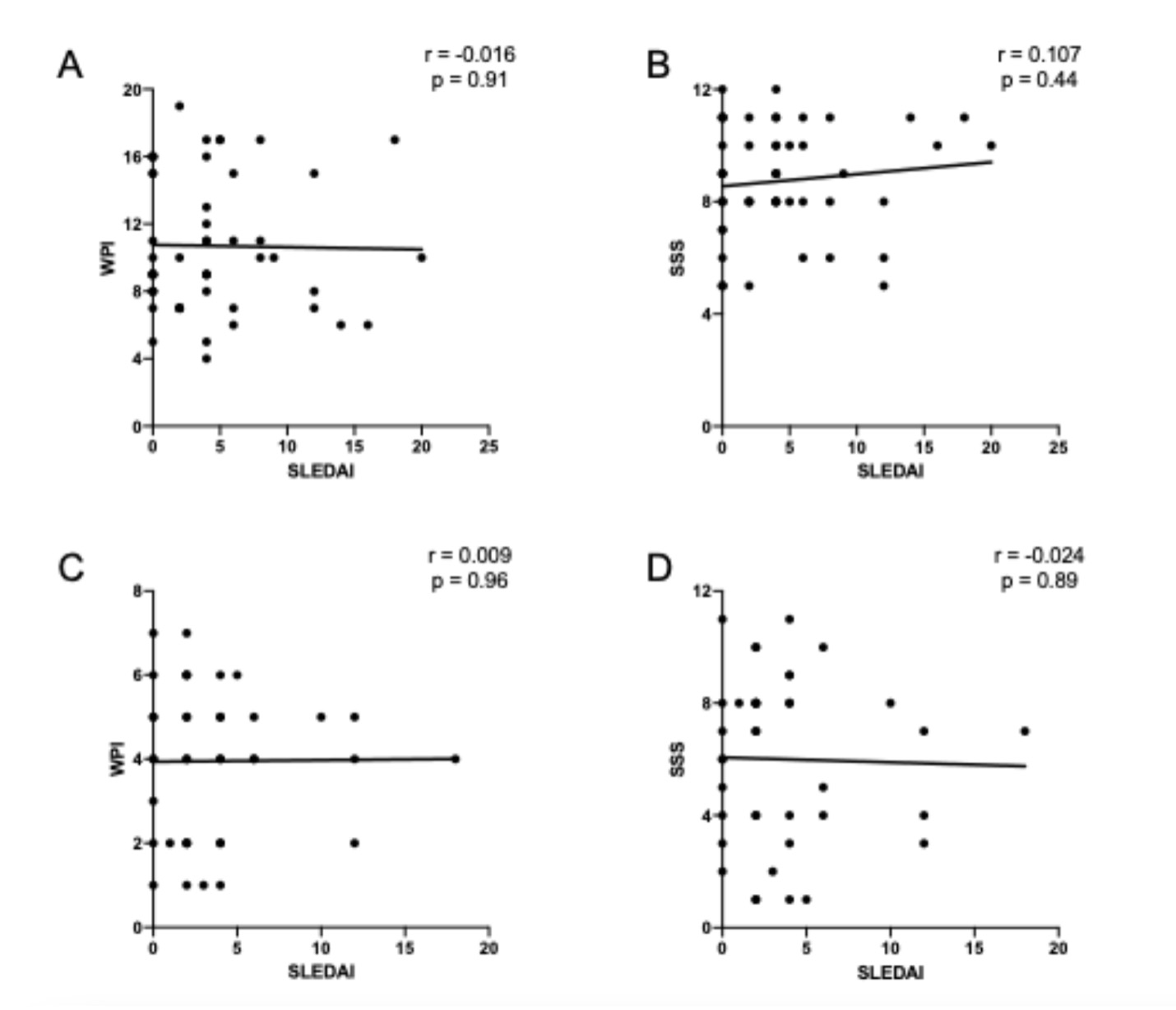
Results: Of 316 SLE patients completing the FM questionnaire, 55 (17%) met criteria for FM, 57 (18%) reported chronic pain without FM, and 204 (65%) had no chronic pain. The racial/ethnic composition of the FM patients was 35% White, 27% Black, 6% Asian, and 31% Hispanic, which differed from those without FM (p=0.023), primarily due to a smaller proportion of Asian patients with FM. There was no significant difference in overall SELENA-SLEDAI score. However, there was more active arthritis in the FM group versus the non-FM group (Table 1). The widespread pain index (WPI) and symptom severity score (SSS) were not correlated with current degree of SLE activity in FM or in non-FM chronic pain patients (Figure 1). Of the SLE criteria, FM patients were less likely to have ever had lupus nephritis and more likely to have malar rash. The SLICC damage index did not differ between groups. Of the 112 patients with chronic pain (plus/minus FM), 70 completed at least one PROMIS measure. Fatigue, sleep disturbance, and cognitive function were significantly worse in FM patients, whereas pain measures, depression, anxiety, and physical function did not differ between groups (Table 2). After adjusting for age, race (White/non-Hispanic), SELENA-SLEDAI score, and current steroid use, FM was associated with a 5-point increase in mean sleep disturbance and fatigue scores. Cognitive function, however, was no longer significantly associated with FM.
Conclusion: These findings underscore that symptoms associated with FM in SLE patients can be independent of chronic pain, SLE disease activity and damage, and overall suggests the need for better understanding of the biology of this group.
.jpg)
.jpg)
K. Corbitt: None; P. Carlucci: None; B. Cohen: None; M. Masson: None; H. Belmont: Alexion, 6, Aurinia, 6; A. Saxena: AbbVie, 1, AstraZeneca, 1, GSK, 1; C. Tseng: None; J. Wang: None; K. Barbour: None; J. Buyon: Bristol-Myers Squibb(BMS), 2, GlaxoSmithKlein(GSK), 2, Related Sciences, 1; P. Izmirly: None; H. Gold: None.
Background/Purpose: SLE patients have a higher prevalence of fibromyalgia (FM) than the general population. FM symptoms, such as nociplastic pain and fatigue, are known to cause discordance in how patients and physicians view well-controlled SLE, and these variables impact quality of life in ways not captured by traditional disease activity scores. There is a paucity of objective data surrounding the clinical interplay of these two diseases. Given this, and emerging evidence suggesting the contribution of autoimmunity to FM, this study evaluated FM clinically, serologically, and via patient reported outcomes in a multi-racial/ethnic SLE patient cohort.
Methods: Patients from an established lupus cohort were screened for FM using the 2016 FM classification criteria during an in-person rheumatologist visit. Hybrid SELENA-SLEDAI scores, SLE classification criteria, medications, and SLICC damage index were evaluated. A portion of the patients who reported any chronic pain also completed 8 PROMIS measures. Clinical and serologic activity was compared in patients with and without FM, as well as T-score means of PROMIS measures between FM and non-FM chronic pain patients. For T-score means significantly different between groups, linear regression models adjusting for age, race/ethnicity, SELENA-SLEDAI score, and steroid use were estimated.
Results: Of 316 SLE patients completing the FM questionnaire, 55 (17%) met criteria for FM, 57 (18%) reported chronic pain without FM, and 204 (65%) had no chronic pain. The racial/ethnic composition of the FM patients was 35% White, 27% Black, 6% Asian, and 31% Hispanic, which differed from those without FM (p=0.023), primarily due to a smaller proportion of Asian patients with FM. There was no significant difference in overall SELENA-SLEDAI score. However, there was more active arthritis in the FM group versus the non-FM group (Table 1). The widespread pain index (WPI) and symptom severity score (SSS) were not correlated with current degree of SLE activity in FM or in non-FM chronic pain patients (Figure 1). Of the SLE criteria, FM patients were less likely to have ever had lupus nephritis and more likely to have malar rash. The SLICC damage index did not differ between groups. Of the 112 patients with chronic pain (plus/minus FM), 70 completed at least one PROMIS measure. Fatigue, sleep disturbance, and cognitive function were significantly worse in FM patients, whereas pain measures, depression, anxiety, and physical function did not differ between groups (Table 2). After adjusting for age, race (White/non-Hispanic), SELENA-SLEDAI score, and current steroid use, FM was associated with a 5-point increase in mean sleep disturbance and fatigue scores. Cognitive function, however, was no longer significantly associated with FM.
Conclusion: These findings underscore that symptoms associated with FM in SLE patients can be independent of chronic pain, SLE disease activity and damage, and overall suggests the need for better understanding of the biology of this group.

Figure 1. WPI and SSS do not correlate with SLEDAI in FM or non-FM chronic pain patients. A–B show the correlation between WPI (A) or SSS (B) and SLEDAI in FM patients and C–D show the correlation between WPI (C) or SSS (D) and SLEDAI in non-FM chronic pain patients.
.jpg)
Table 1. Clinical and serologic profiling of SLE patients with FM compared to those without FM, n=316. Demographics, current SLE manifestations via SELENA-SLEDAI, SLE classification criteria and associated antibodies, and SLICC damage index are all shown. *N is specified where it differs from overall sample size.
.jpg)
Table 2. PROMIS measures assessed between groups, with “worse” quality of life represented by higher scores for symptoms and lower scores for function. PROMIS T score mean for the general United States population=50 (SD=10). Minimally important difference=2–3 for PROMIS measures in patients with lupus. Non-FM chronic pain patients had n=35 in the top 4 measures of the table and n=33 for the remaining 4 measures.
K. Corbitt: None; P. Carlucci: None; B. Cohen: None; M. Masson: None; H. Belmont: Alexion, 6, Aurinia, 6; A. Saxena: AbbVie, 1, AstraZeneca, 1, GSK, 1; C. Tseng: None; J. Wang: None; K. Barbour: None; J. Buyon: Bristol-Myers Squibb(BMS), 2, GlaxoSmithKlein(GSK), 2, Related Sciences, 1; P. Izmirly: None; H. Gold: None.



