Poster Session C
Rheumatoid arthritis (RA)
Session: (1713–1733) RA – Animal Models Poster
1719: Exploring the Link Between Osteitis and Bone Microstructure Changes in Rheumatoid Arthritis: Role of JAK-STAT Signaling Pathway
Tuesday, November 14, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
Tsuneyasu Yoshida, MD
Graduate School of Medicine, Kyoto University
KYOTO, JapanDisclosure information not submitted.
Abstract Poster Presenter(s)
Tsuneyasu Yoshida1, Yoichi Nakayama1, Masao Katsushima2, Yuri Nishida3, Mirei Shirakashi1, Ran Nakashima1, Ryu Watanabe2, Kosaku Murakami4, Hajime Yoshifuji1, Akio Morinobu1 and Motomu Hashimoto2, 1Department of Rheumatology and Clinical Immunology, Graduate School of Medicine, Kyoto University, Kyoto, Japan, 2Department of Clinical Immunology, Graduate School of Medicine, Osaka Metropolitan University, Osaka, Japan, 3Department of Endocrinology and Rheumatology, Kurashiki Central Hospital, Kurashiki, Japan, 4Center for Cancer Immunotherapy and Immunobiology, Graduate School of Medicine, Kyoto University, Kyoto, Japan
Background/Purpose: Osteitis on MRI and bone microstructure changes (BMC) on high-resolution peripheral quantitative CT are the earliest signs of arthritis, preceding the development of bone erosion in rheumatoid arthritis (RA). Recently, a JAK inhibitor, Baricitinib (BAR), was shown to inhibit these early changes. This study aimed to elucidate the underlying molecular mechanism of osteitis and BMC, including the role of JAK-STAT cytokines, using SKG mice, an animal model of RA with a ZAP70 gene point mutation.
Methods: Osteitis was assessed by an increase in the femur/gastrocnemius muscle (F/G) ratio in fat-suppressed T2-weighted images on MRI. BMC was evaluated by a decrease in bone volume/tissue volume (BV/TV) ratio on micro CT. Arthritis was induced by intraperitoneal injection of zymosan. BAR or a vehicle was orally administered in the early or late phases during arthritis development. Flow cytometry was used to analyze the bone marrow (BM) cells, including granulocyte-monocyte (GM) progenitor cells, granulocytes, and monocytes. Immunohistochemistry (IHC) was performed to identify the distribution of tartrate-resistant acid phosphatase (TRAP)-positive cells in the BM. The role of JAK-STAT cytokines was investigated by examining the expression of JAK-STAT cytokines in the BM or by supplementing recombinant (r-) GM-CSF or IL-6 to in vivo arthritis model or in vitro RANKL-mediated osteoclast differentiation assay.
Results: Osteitis developed earlier (at 2 weeks) than BMC (at 5 weeks) during arthritis development. Osteitis scores on MRI well correlated with BMC scores on micro CT (ρ=-0.58, p=0.03). Following zymosan injection, GM progenitor cells, granulocytes, and monocytes were increased in the BM. The number of monocytes significantly correlated with osteitis scores (ρ=0.77, p=0.0008). Monocytes were further subdivided into three populations (Ly6C high CD11b low, Ly6C high CD11b high, and Ly6C mid CD11b+), and TRAP-positive osteoclasts were preferentially differentiated from the Ly6C high CD11b low population (osteoclast precursor cells) in vitro. IHC revealed increased TRAP-positive cells at the edges of bone surfaces in the BM where BMC was observed. BAR effectively suppressed osteitis (F/G ratio: 1.44 in vehicle group vs. 1.17 in BAR group, p=0.0379) and BMC (BV/TV ratio: 37.3 % in vehicle group vs. 46.3 % in BAR group, p=0.0052). BAR reduced the number of GM progenitor cells, monocytes, and osteoclast precursor cells. Among JAK-STAT cytokines, GM-CSF and IL-6 were highly expressed in the BM after zymosan injection. In vivo administration of rGM-CSF to SKG mice promoted osteitis, especially when administered in the late phase of arthritis. In vitro addition of rGM-CSF to osteoclast differentiation assay markedly increased the number and size of osteoclasts, especially when added in the late phase of osteoclast differentiation, which was inhibited by further addition of BAR.
Conclusion: During arthritis progression, osteitis precedes BMC. The increase of GM-lineage cells in the BM induces osteitis, while proliferation and subsequent differentiation of osteoclast precursors contribute to BMC. JAK-STAT cytokines, especially GM-CSF, play a crucial role in these processes.
T. Yoshida: None; Y. Nakayama: None; M. Katsushima: None; Y. Nishida: None; M. Shirakashi: None; R. Nakashima: None; R. Watanabe: AbbVie, 5, Asahi Kasei, 6, Chugai, 6, Eli Lilly, 6, GlaxoSmithKlein(GSK), 6, Sanofi, 6; K. Murakami: None; H. Yoshifuji: None; A. Morinobu: None; M. Hashimoto: Abbvie, 5, 6, Asahi Kasei, 5, 6, Astellas, 5, 6, Ayumi, 5, 6, Brystol Meyers, 5, 6, Chugai, 5, 6, Daiichi Sankyo, 5, 6, EA Pharma, 5, 6, Eisai, 5, 6, Eli Lilly, 5, 6, Novartis Pharma, 5, 6, Taisho Toyama, 5, 6, Tanabe Mitsubishi, 5, 6.
Background/Purpose: Osteitis on MRI and bone microstructure changes (BMC) on high-resolution peripheral quantitative CT are the earliest signs of arthritis, preceding the development of bone erosion in rheumatoid arthritis (RA). Recently, a JAK inhibitor, Baricitinib (BAR), was shown to inhibit these early changes. This study aimed to elucidate the underlying molecular mechanism of osteitis and BMC, including the role of JAK-STAT cytokines, using SKG mice, an animal model of RA with a ZAP70 gene point mutation.
Methods: Osteitis was assessed by an increase in the femur/gastrocnemius muscle (F/G) ratio in fat-suppressed T2-weighted images on MRI. BMC was evaluated by a decrease in bone volume/tissue volume (BV/TV) ratio on micro CT. Arthritis was induced by intraperitoneal injection of zymosan. BAR or a vehicle was orally administered in the early or late phases during arthritis development. Flow cytometry was used to analyze the bone marrow (BM) cells, including granulocyte-monocyte (GM) progenitor cells, granulocytes, and monocytes. Immunohistochemistry (IHC) was performed to identify the distribution of tartrate-resistant acid phosphatase (TRAP)-positive cells in the BM. The role of JAK-STAT cytokines was investigated by examining the expression of JAK-STAT cytokines in the BM or by supplementing recombinant (r-) GM-CSF or IL-6 to in vivo arthritis model or in vitro RANKL-mediated osteoclast differentiation assay.
Results: Osteitis developed earlier (at 2 weeks) than BMC (at 5 weeks) during arthritis development. Osteitis scores on MRI well correlated with BMC scores on micro CT (ρ=-0.58, p=0.03). Following zymosan injection, GM progenitor cells, granulocytes, and monocytes were increased in the BM. The number of monocytes significantly correlated with osteitis scores (ρ=0.77, p=0.0008). Monocytes were further subdivided into three populations (Ly6C high CD11b low, Ly6C high CD11b high, and Ly6C mid CD11b+), and TRAP-positive osteoclasts were preferentially differentiated from the Ly6C high CD11b low population (osteoclast precursor cells) in vitro. IHC revealed increased TRAP-positive cells at the edges of bone surfaces in the BM where BMC was observed. BAR effectively suppressed osteitis (F/G ratio: 1.44 in vehicle group vs. 1.17 in BAR group, p=0.0379) and BMC (BV/TV ratio: 37.3 % in vehicle group vs. 46.3 % in BAR group, p=0.0052). BAR reduced the number of GM progenitor cells, monocytes, and osteoclast precursor cells. Among JAK-STAT cytokines, GM-CSF and IL-6 were highly expressed in the BM after zymosan injection. In vivo administration of rGM-CSF to SKG mice promoted osteitis, especially when administered in the late phase of arthritis. In vitro addition of rGM-CSF to osteoclast differentiation assay markedly increased the number and size of osteoclasts, especially when added in the late phase of osteoclast differentiation, which was inhibited by further addition of BAR.
Conclusion: During arthritis progression, osteitis precedes BMC. The increase of GM-lineage cells in the BM induces osteitis, while proliferation and subsequent differentiation of osteoclast precursors contribute to BMC. JAK-STAT cytokines, especially GM-CSF, play a crucial role in these processes.

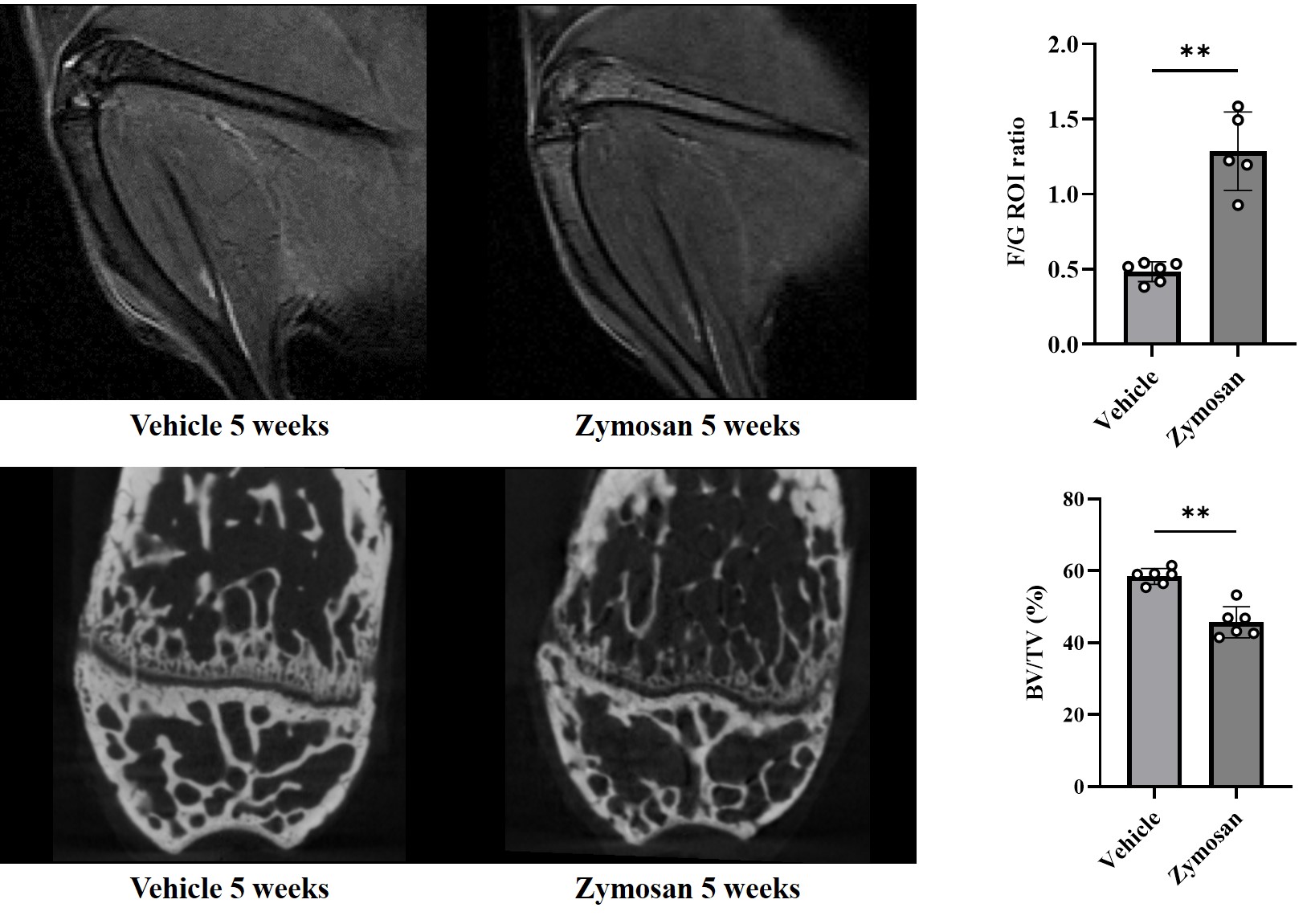
Figure: Osteitis and bone microstructure changes after arthritis induction in SKG mice
BV/TV: Bone volume/Tissue volume, F/G: Femur/Gastrocnemius muscle
Mann-Whitney test, ** p < 0.01
BV/TV: Bone volume/Tissue volume, F/G: Femur/Gastrocnemius muscle
Mann-Whitney test, ** p < 0.01
T. Yoshida: None; Y. Nakayama: None; M. Katsushima: None; Y. Nishida: None; M. Shirakashi: None; R. Nakashima: None; R. Watanabe: AbbVie, 5, Asahi Kasei, 6, Chugai, 6, Eli Lilly, 6, GlaxoSmithKlein(GSK), 6, Sanofi, 6; K. Murakami: None; H. Yoshifuji: None; A. Morinobu: None; M. Hashimoto: Abbvie, 5, 6, Asahi Kasei, 5, 6, Astellas, 5, 6, Ayumi, 5, 6, Brystol Meyers, 5, 6, Chugai, 5, 6, Daiichi Sankyo, 5, 6, EA Pharma, 5, 6, Eisai, 5, 6, Eli Lilly, 5, 6, Novartis Pharma, 5, 6, Taisho Toyama, 5, 6, Tanabe Mitsubishi, 5, 6.



