Poster Session C
Fibrosing rheumatic diseases (scleroderma, MCTD, IgG4-related disease, scleroderma mimics)
Session: (2352–2369) Systemic Sclerosis & Related Disorders – Clinical Poster III: Translational Science
2358: Neutrophil Extracellular Traps Induce Endothelial-to-Mesenchymal Transition and Associate with Vascular Complications in Scleroderma
Tuesday, November 14, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- RA
Ramadan Ali, PhD
University of Michigan
Ann Arobr, MI, United StatesDisclosure information not submitted.
Abstract Poster Presenter(s)
Ramadan Ali1, Wenying Liang1, Claire Shiple1, Rosemary Gedert2, Suiyuan Huang1, Cyrus Sarosh1, Eliza Pei-Suen Tsou1, Dinesh Khanna1 and Jason Knight1, 1University of Michigan, Ann Arbor, MI, 2University of Michigan, Onsted, MI
Background/Purpose: Systemic sclerosis (SSc, also known as scleroderma) is a disease characterized by fibrosis, autoimmunity, and vasculopathy. Many devastating complications such as digital ulcerations, scleroderma renal crisis, and pulmonary hypertension are the result of deranged vasculature. Neutrophils and neutrophil extracellular traps (NETs) have recently been revealed as drivers of diverse vasculopathies. Here, we investigated the potential role of NETs in SSc vascular complications.
Methods: Plasma from SSc patients (cohort 1: n=29, cohort 2: n=37) and healthy controls (n= 21) were evaluated for NET remnants (MPO-DNA complexes) and another marker of neutrophil activation (calprotectin) via ELISAs. For cohort 2, we compared diffuse SSc patients with known vascular complications (digital ulcers, pulmonary hypertension, and scleroderma renal crisis) to a cohort matched for age, sex, and disease duration but without vascular complications. To assess the effect of NETs on endothelial-to-mesenchymal transition (EndoMT), healthy human umbilical vein endothelial cells (HUVECs) or human dermal microvascular endothelial cells (MVECs) were treated with NETs (0.1-1 µg DNA/ml), TGF-β (10-100 ng/ml), or NETs + TGF-β. Gene expression of mesenchymal markers ACTA2 and S100A4 was measured by quantitative PCR.
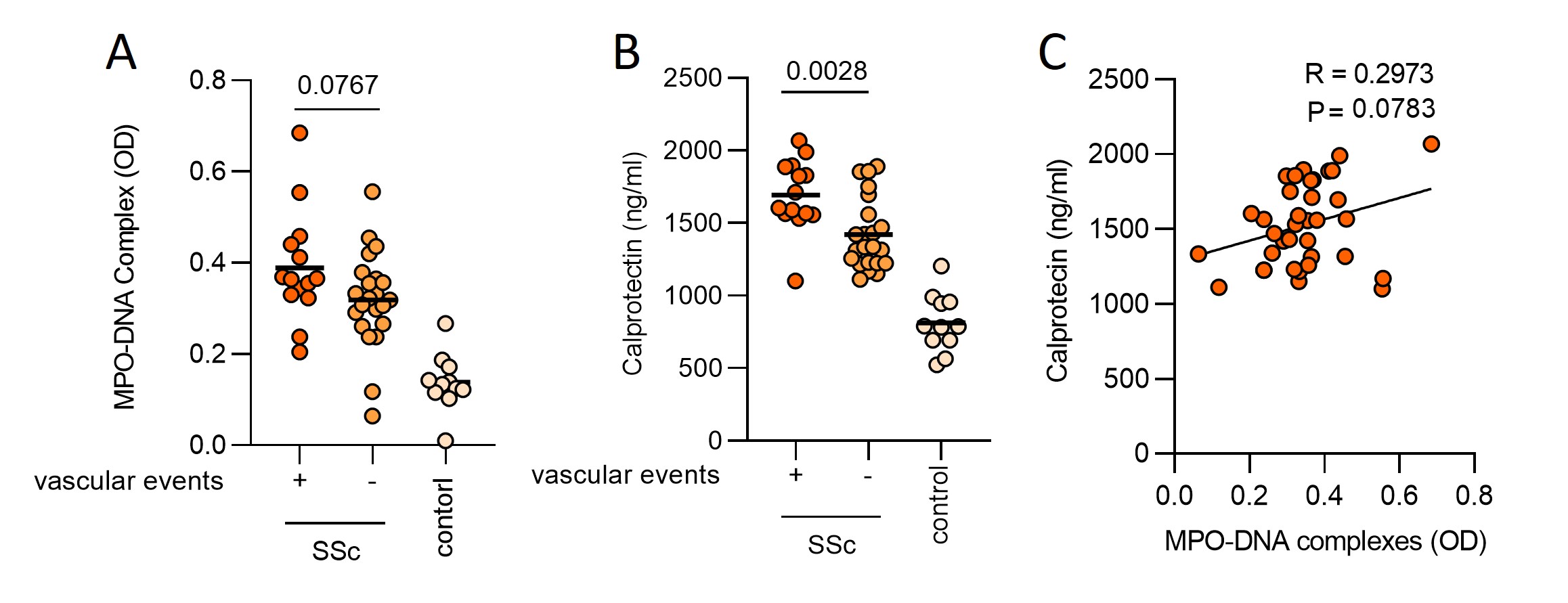
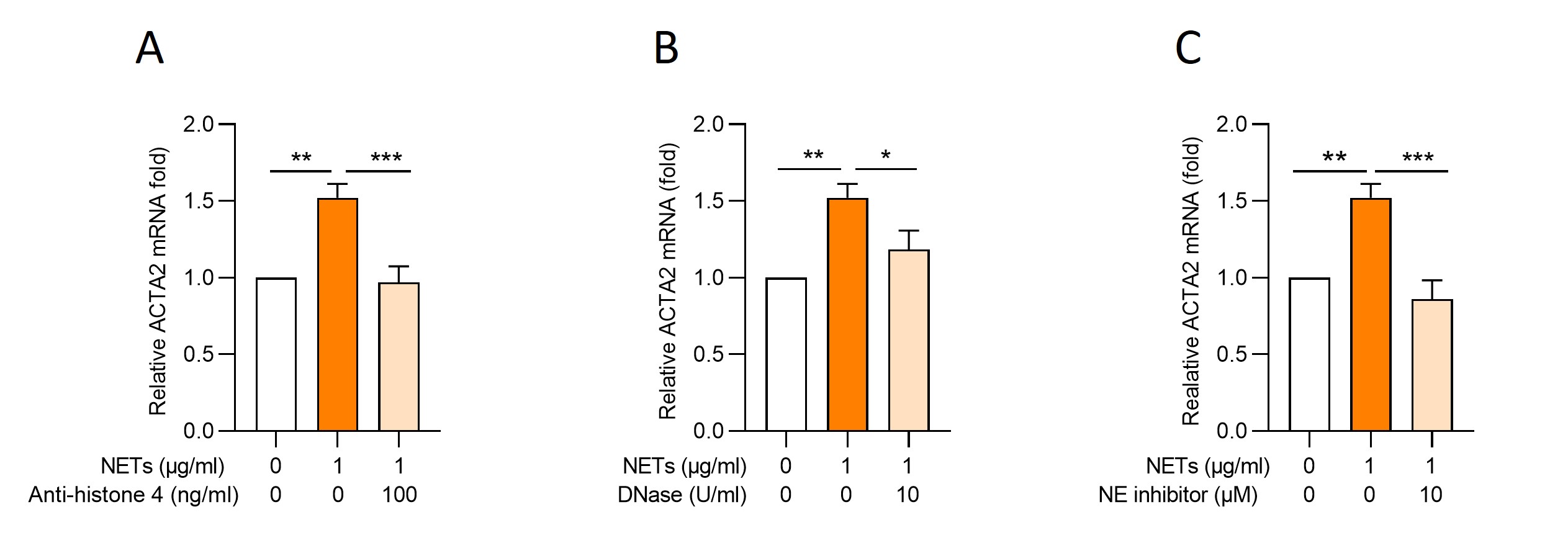
Results: We tested cohort 1 (mixed SSc subtypes) and found higher levels of NET remnants in the plasma of patients with SSc compared to healthy controls (MPO-DNA complexes, mean 2.1 vs. 0.89 µg/ml, P=0.0018). At the same time, we found more plasma calprotectin in patients with diffuse SSc as compared with limited SSc (P=0.033). We then focused on patients with diffuse SSc (cohort 2), some with a history of vascular complications, and some without (Figure 1). We found more plasma calprotectin in patients with a history of vascular complications (P=0.0028); there was also a trend toward higher levels of NET remnants in the vascular patients although it missed statistical significance (P=0.0767). Interestingly, we found a positive correlation between circulating NETs and TGF-β (R=0.405, P=0.032), the latter a molecular determinant of fibrosis in SSc. We next assessed the potential role of NETs as inducers of EndoMT and the extent to which NETs might synergize with TGF-β in this induction. The combination of NETs and TGF-β (both at suboptimal concentrations) increased the expression of mesenchymal markers ACTA2 (P=0.035) and S100A4 (near significance at P=0.051) in HUVECs as compared with NETs or TGF-β alone. At higher concentrations (1 µg DNA/ml), NETs alone were also able to induce EndoMT (P=0.001). Notably, the addition of anti-NET therapies including anti-histone 4 (P=0.0007), DNase (P=0.034), and a neutrophil elastase inhibitor (P=0.0003) significantly reduced NET-mediated EndoMT (Figure 2).
Conclusion: Our data suggest that SSc patients with vascular complications have higher circulating markers of neutrophil activation than a matched cohort without vascular complications. Our data also suggest that NETs, similar to TGF-β, can contribute to EndoMT, and that anti-NET therapies might have potential for combatting EndoMT and vascular disease in SSc.
R. Ali: None; W. Liang: None; C. Shiple: None; R. Gedert: None; S. Huang: None; C. Sarosh: None; E. Tsou: None; D. Khanna: AbbVie, 12, DSMB, AstraZeneca, 2, Boehringer-Ingelheim, 2, Bristol-Myers Squibb, 2, 5, CSL Behring, 2, Genentech, 2, Horizon Therapeutics, 2, 5, Janssen, 2, 6, Pfizer, 5, Prometheus, 2; J. Knight: Jazz Pharmaceuticals, 2.
Background/Purpose: Systemic sclerosis (SSc, also known as scleroderma) is a disease characterized by fibrosis, autoimmunity, and vasculopathy. Many devastating complications such as digital ulcerations, scleroderma renal crisis, and pulmonary hypertension are the result of deranged vasculature. Neutrophils and neutrophil extracellular traps (NETs) have recently been revealed as drivers of diverse vasculopathies. Here, we investigated the potential role of NETs in SSc vascular complications.
Methods: Plasma from SSc patients (cohort 1: n=29, cohort 2: n=37) and healthy controls (n= 21) were evaluated for NET remnants (MPO-DNA complexes) and another marker of neutrophil activation (calprotectin) via ELISAs. For cohort 2, we compared diffuse SSc patients with known vascular complications (digital ulcers, pulmonary hypertension, and scleroderma renal crisis) to a cohort matched for age, sex, and disease duration but without vascular complications. To assess the effect of NETs on endothelial-to-mesenchymal transition (EndoMT), healthy human umbilical vein endothelial cells (HUVECs) or human dermal microvascular endothelial cells (MVECs) were treated with NETs (0.1-1 µg DNA/ml), TGF-β (10-100 ng/ml), or NETs + TGF-β. Gene expression of mesenchymal markers ACTA2 and S100A4 was measured by quantitative PCR.
Results: We tested cohort 1 (mixed SSc subtypes) and found higher levels of NET remnants in the plasma of patients with SSc compared to healthy controls (MPO-DNA complexes, mean 2.1 vs. 0.89 µg/ml, P=0.0018). At the same time, we found more plasma calprotectin in patients with diffuse SSc as compared with limited SSc (P=0.033). We then focused on patients with diffuse SSc (cohort 2), some with a history of vascular complications, and some without (Figure 1). We found more plasma calprotectin in patients with a history of vascular complications (P=0.0028); there was also a trend toward higher levels of NET remnants in the vascular patients although it missed statistical significance (P=0.0767). Interestingly, we found a positive correlation between circulating NETs and TGF-β (R=0.405, P=0.032), the latter a molecular determinant of fibrosis in SSc. We next assessed the potential role of NETs as inducers of EndoMT and the extent to which NETs might synergize with TGF-β in this induction. The combination of NETs and TGF-β (both at suboptimal concentrations) increased the expression of mesenchymal markers ACTA2 (P=0.035) and S100A4 (near significance at P=0.051) in HUVECs as compared with NETs or TGF-β alone. At higher concentrations (1 µg DNA/ml), NETs alone were also able to induce EndoMT (P=0.001). Notably, the addition of anti-NET therapies including anti-histone 4 (P=0.0007), DNase (P=0.034), and a neutrophil elastase inhibitor (P=0.0003) significantly reduced NET-mediated EndoMT (Figure 2).
Conclusion: Our data suggest that SSc patients with vascular complications have higher circulating markers of neutrophil activation than a matched cohort without vascular complications. Our data also suggest that NETs, similar to TGF-β, can contribute to EndoMT, and that anti-NET therapies might have potential for combatting EndoMT and vascular disease in SSc.

Fig 1: Plasma from SSc patients with vascular complications (n = 14), SSc patients without vascular complications (n = 23), and healthy controls (n = 11) were assessed for (A) MPO-DNA complexes and (B) calprotectin. Groups in A and B were compared by unpaired t-test, and Pearson correlation was computed for C.

Fig 2: HUVECs were stimulated with NETs for 24 hours in the presence or absence of (A) anti-histone 4, (B) DNase, and (C) the NE inhibitor Sivelestat. The expression of mesenchymal markers was quantified by quantitative PCR. Groups were compared by unpaired ANOVA with correction for multiple comparisons.
R. Ali: None; W. Liang: None; C. Shiple: None; R. Gedert: None; S. Huang: None; C. Sarosh: None; E. Tsou: None; D. Khanna: AbbVie, 12, DSMB, AstraZeneca, 2, Boehringer-Ingelheim, 2, Bristol-Myers Squibb, 2, 5, CSL Behring, 2, Genentech, 2, Horizon Therapeutics, 2, 5, Janssen, 2, 6, Pfizer, 5, Prometheus, 2; J. Knight: Jazz Pharmaceuticals, 2.



