Poster Session C
Epidemiology, health policy and outcomes
Session: (1796–1826) Epidemiology & Public Health Poster III
1801: Tixagevimab/Cilgavimab for the Prevention of COVID-19 in Vaccine-refractory Patients with Autoimmune Diseases: A Prospective Cohort Study
Tuesday, November 14, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- FF
Filippo Fagni, MD (he/him/his)
University clinic of Erlangen and Friedrich Alexander Universität Erlangen-Nürnberg
Erlangen, GermanyDisclosure information not submitted.
Abstract Poster Presenter(s)
Ioanna Minopoulou1, Koray Tascilar2, Giulia Corte1, Melek Yalcin Mutlu1, Katja Schmidt2, Daniela Bohr1, Fabian Hartmann2, Bernhard Manger2, Arnd Kleyer3, David Simon1, Thomas Harrer2, Georg Schett4 and Filippo Fagni1, 1Friedrich-Alexander-Universität (FAU) Erlangen-Nürnberg & Universitätsklinikum Erlangen, Department of Internal Medicine 3 - Rheumatology and Immunology / Deutsches Zentrum Immuntherapie, Erlangen, Germany, 2Department of Internal Medicine 3, Friedrich-Alexander University (FAU) Erlangen-Nuremberg and Universitätsklinikum Erlangen, Erlangen, Germany, 3University Hospital Erlangen, Erlangen, Germany, 4Friedrich-Alexander-Universität Erlangen-Nürnberg, Erlangen, Germany
Background/Purpose: Patients with immune-mediated inflammatory diseases (IMIDs) and particularly those treated with B-cell-depleting agents frequently fail to develop humoral responses to SARS-CoV-2 vaccinations and have an increased risk of breakthrough infections and severe COVID-19 (Simon D, Arthritis Rheum (2022); Avouac J, Lancet Rheum (2022)). Passive immunization with monoclonal antibodies such as the FDA- and EMA-approved combination tixagevimab/cilgavimab could be an alternative option for this frail population. However, despite promising results from trials on healthy individuals (Levin MJ, NEJM (2022)), efficacy data on IMIDs are scarce. With our study, we aimed to investigate the effect of a pre-exposure prophylaxis (PrEP) with tixagevimab/cilgavimab on the humoral responses and on the prevention of symptomatic COVID-19 in vaccine-refractory IMID patients.
Methods: A prospective interventional cohort study was performed on a cohort of high-risk vaccine-refractory IMID patients undergoing B-cell depletion and/or with primary immunodeficiencies that received PrEP with a single dose of subcutaneous tixagevimab/cilgavimab (150mg/150mg). COVID-19 outcomes and serum anti-SARS-CoV-2 IgG were assessed at baseline, day 1, day 14, day 28, month 3, and after at least 6 months. Standardised incidence ratios (SIR) of COVID-19 were compared to (i) a control group of other vaccine-refractory IMID and primary immunodeficiency patients who did not receive prophylactic treatment with tixagevimab/cilgavimab and (ii) the local general population to assess the risk of symptomatic COVID-19. All included patients were attending the outpatient clinic of the Department of Medicine 3 (Rheumatology and Immunology) of the University Clinic Erlangen between March and September 2022. Ethical approval (#157_20 B) to conduct this analysis was granted by the institutional review board of the University Clinic of Erlangen.
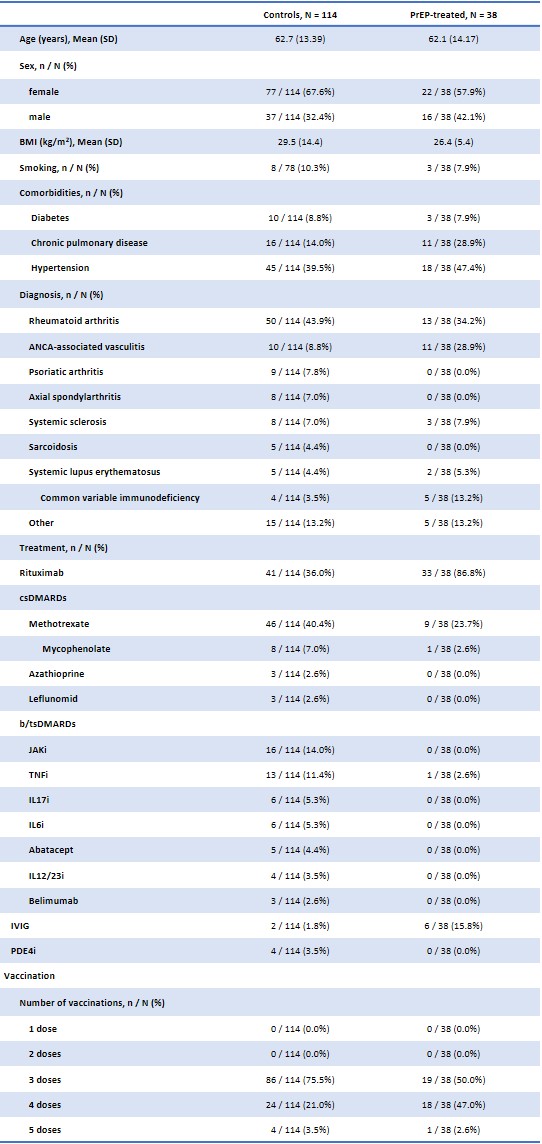
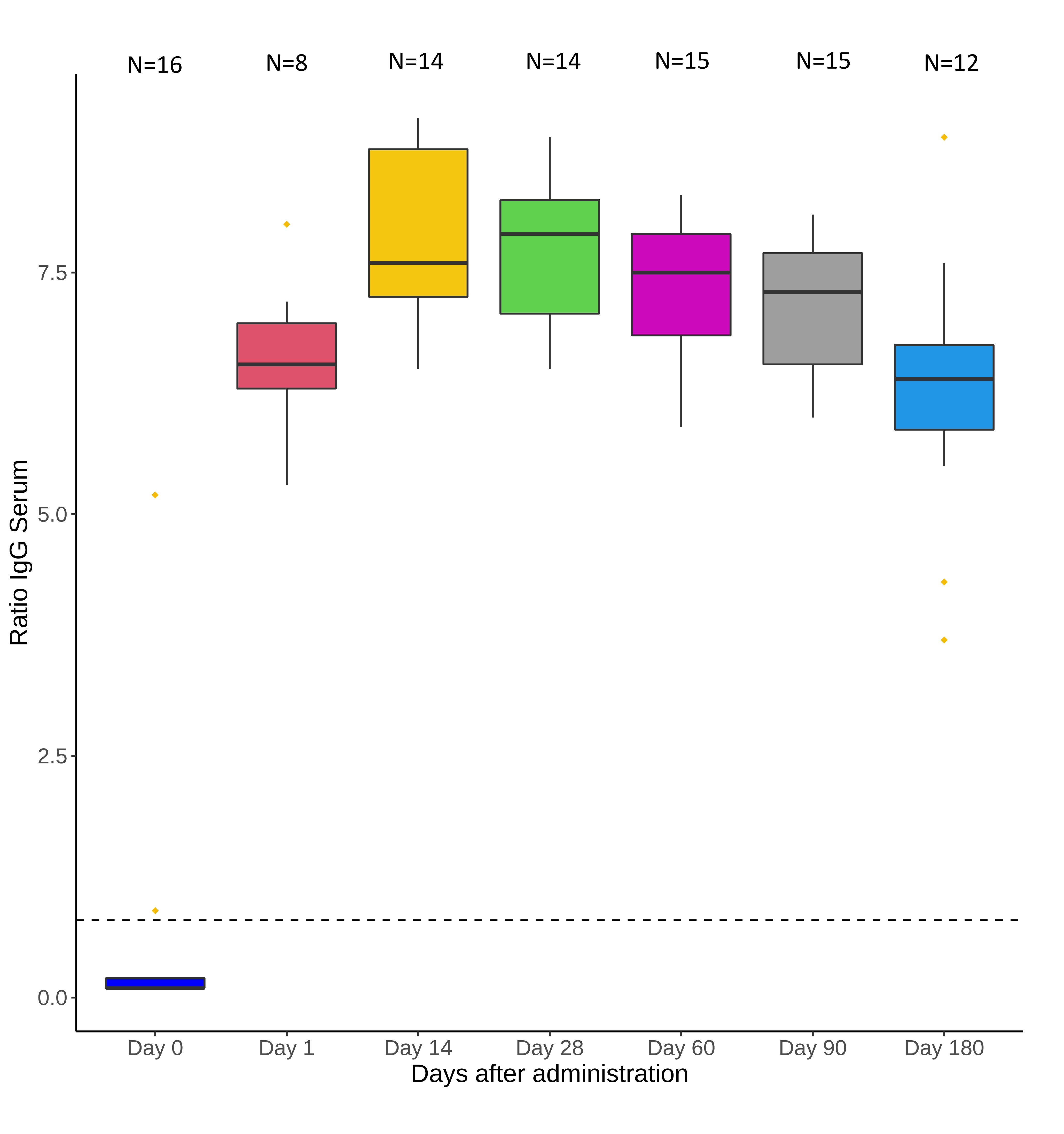
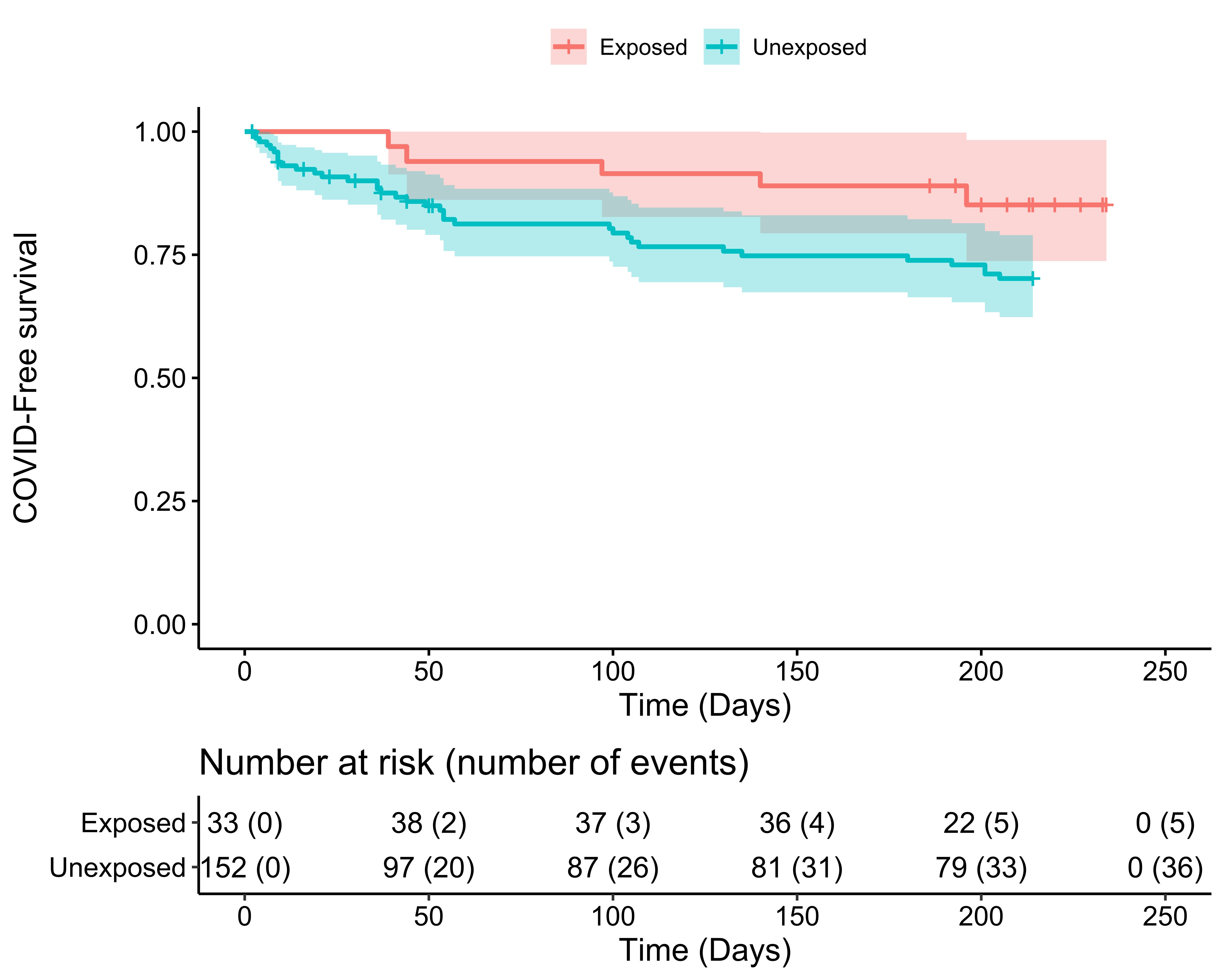
Results: A total of 38 high-risk IMID patients received tixagevimab/cilgavimab and were compared with 114 untreated high-risk IMID controls. Clinical and demographic characteristics are shown in Table 1. Serum anti-Spike IgG increased to 6.6 OD (SD: ±0.8) at day one and remained positive up to month 6 (6.3±1.4 OD) (Figure 1). Compared to the general population, the SIR of COVID-19 in treated patients was 0.76 (95% CI: 0.24-1.58) despite the increased risk profile and more frequent use of B-cell-depleting therapies. The SIR of the untreated control group was 1.51 (1.07-2.02), corresponding to a significantly increased incidence of COVID-19. A Kaplan-Meyer curve of infection-free survival throughout the 6-month follow-up period is shown in figure 2.
Conclusion: Passive immunization with tixagevimab/cilgavimab is safe and effective in quicklly inducing anti-SARS-CoV-2 humoral immunity and potentially in preventing COVID-19 in high-risk vaccine-refractory IMID patients. These data provide a proof of concept for the use of monoclonal antibodies as a preventative strategy against SARS-CoV-2 in vulnerable populations such as those with inadequate vaccine responses. Thus, the development of new neutralizing antibodies to protect this frail population remains critical.
I. Minopoulou: AbbVie/Abbott, 6; K. Tascilar: None; G. Corte: None; M. Yalcin Mutlu: None; K. Schmidt: None; D. Bohr: None; F. Hartmann: None; B. Manger: AbbVie/Abbott, 6, Alexion, 6, Celgene, 6, Eli Lilly, 6, EUSA, 6, Gilead, 6, Janssen, 6, Merck/MSD, 6, Pfizer, 6, Roche, 6, Sanofi, 6; A. Kleyer: None; D. Simon: Janssen, 5; T. Harrer: None; G. Schett: None; F. Fagni: Eli Lilly, 6, Galapagos, 6, Novartis, 6.
Background/Purpose: Patients with immune-mediated inflammatory diseases (IMIDs) and particularly those treated with B-cell-depleting agents frequently fail to develop humoral responses to SARS-CoV-2 vaccinations and have an increased risk of breakthrough infections and severe COVID-19 (Simon D, Arthritis Rheum (2022); Avouac J, Lancet Rheum (2022)). Passive immunization with monoclonal antibodies such as the FDA- and EMA-approved combination tixagevimab/cilgavimab could be an alternative option for this frail population. However, despite promising results from trials on healthy individuals (Levin MJ, NEJM (2022)), efficacy data on IMIDs are scarce. With our study, we aimed to investigate the effect of a pre-exposure prophylaxis (PrEP) with tixagevimab/cilgavimab on the humoral responses and on the prevention of symptomatic COVID-19 in vaccine-refractory IMID patients.
Methods: A prospective interventional cohort study was performed on a cohort of high-risk vaccine-refractory IMID patients undergoing B-cell depletion and/or with primary immunodeficiencies that received PrEP with a single dose of subcutaneous tixagevimab/cilgavimab (150mg/150mg). COVID-19 outcomes and serum anti-SARS-CoV-2 IgG were assessed at baseline, day 1, day 14, day 28, month 3, and after at least 6 months. Standardised incidence ratios (SIR) of COVID-19 were compared to (i) a control group of other vaccine-refractory IMID and primary immunodeficiency patients who did not receive prophylactic treatment with tixagevimab/cilgavimab and (ii) the local general population to assess the risk of symptomatic COVID-19. All included patients were attending the outpatient clinic of the Department of Medicine 3 (Rheumatology and Immunology) of the University Clinic Erlangen between March and September 2022. Ethical approval (#157_20 B) to conduct this analysis was granted by the institutional review board of the University Clinic of Erlangen.
Results: A total of 38 high-risk IMID patients received tixagevimab/cilgavimab and were compared with 114 untreated high-risk IMID controls. Clinical and demographic characteristics are shown in Table 1. Serum anti-Spike IgG increased to 6.6 OD (SD: ±0.8) at day one and remained positive up to month 6 (6.3±1.4 OD) (Figure 1). Compared to the general population, the SIR of COVID-19 in treated patients was 0.76 (95% CI: 0.24-1.58) despite the increased risk profile and more frequent use of B-cell-depleting therapies. The SIR of the untreated control group was 1.51 (1.07-2.02), corresponding to a significantly increased incidence of COVID-19. A Kaplan-Meyer curve of infection-free survival throughout the 6-month follow-up period is shown in figure 2.
Conclusion: Passive immunization with tixagevimab/cilgavimab is safe and effective in quicklly inducing anti-SARS-CoV-2 humoral immunity and potentially in preventing COVID-19 in high-risk vaccine-refractory IMID patients. These data provide a proof of concept for the use of monoclonal antibodies as a preventative strategy against SARS-CoV-2 in vulnerable populations such as those with inadequate vaccine responses. Thus, the development of new neutralizing antibodies to protect this frail population remains critical.

Table 1. Demographic and clinical characteristics of the treated cohort and control group.

Figure 1. Box and whiskers plot of the levels of serum anti-SARS-CoV-2 spike IgG antibodies before and after 1 day, 14 days, 28 days, 60 days, and 90 days from tixagevimab/cilgavimab treatment. The dashed horizontal line represents the cut-off value of 0.8 optical density ratio (anti-SARS-CoV-2 IgG negative/positive).

Figure 2. Kaplan-Meier curve of COVID-free survival for the treated group (red line) and the untreated vaccine-refractory controls (blue line).
I. Minopoulou: AbbVie/Abbott, 6; K. Tascilar: None; G. Corte: None; M. Yalcin Mutlu: None; K. Schmidt: None; D. Bohr: None; F. Hartmann: None; B. Manger: AbbVie/Abbott, 6, Alexion, 6, Celgene, 6, Eli Lilly, 6, EUSA, 6, Gilead, 6, Janssen, 6, Merck/MSD, 6, Pfizer, 6, Roche, 6, Sanofi, 6; A. Kleyer: None; D. Simon: Janssen, 5; T. Harrer: None; G. Schett: None; F. Fagni: Eli Lilly, 6, Galapagos, 6, Novartis, 6.



