Poster Session C
Spondyloarthritis (SpA) including psoriatic arthritis (PsA)
Session: (2195–2226) Spondyloarthritis Including Psoriatic Arthritis – Diagnosis, Manifestations, & Outcomes Poster III: SpA
2199: Four-year Secukinumab Treatment Outcomes in Axial Spondyloarthritis and Psoriatic Arthritis Patients Treated in Routine Care: Results from the EuroSpA Collaboration
Tuesday, November 14, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- MP
Marion Pons, MD
Rigshospitalet, Valdemar Hansens Vej 17, 2600 Glostrup, Denmark.
Glostrup, DenmarkDisclosure information not submitted.
Abstract Poster Presenter(s)
Marion Pons1, Stylianos Georgiadis1, Zohra Faizy Ahmadzay2, Mikkel Østergaard3, Bente Glintborg1, Jette Heberg1, Sara Nysom Christiansen1, Simon Horskjær Rasmussen1, Isabel Castrejon4, Fernando Alonso5, Karin Laas6, Sigrid Vorobjov7, Nikita Khmelinskii8, Jakub Zavada9, Adrian Ciurea10, Michael Nissen11, Brigitte Michelsen12, Pawel Mielnik13, Anne Gitte Loft14, Ziga Rotar15, Katja Perdan Pirkmajer16, Gary Macfarlane17, Gareth T. Jones17, Dan Nordstrom18, Anna-Mari Hokkanen19, Irene van der Horst-Bruinsma20, Johan Karlsson Wallman21, Bjorn Gudbjornsson22, olafur Palsson23, Merete Hetland1 and Lykke Ørnbjerg1, 1Copenhagen Center for Arthritis Research, Rigshospitalet, Copenhagen, Denmark, 2Rigshospitalet Glostrup, University of Copenhagen, Glostrup, Denmark, 3Copenhagen Center for Arthritis Research, Center for Rheumatology and Spine Diseases, Centre for Head and Orthopaedics, Rigshospitalet; University of Copenhagen, Copenhagen, Denmark, 4Hospital Universitario Gregorio Marañón, Madrid, Spain, 5Spanish Society of Rheumatology, Madrid, Spain, 6Department of Rheumatology, East-Tallinn Central Hospital, Tallinn, Estonia, 7National Institute for Health Development, Tallinn, Estonia, 8Centro Hospitalar Universitário Lisboa Norte, Lisboa, Portugal, 9Institute of Rheumatology; Charles University, Prague, Czech Republic, 10University Hospital Zurich, Zürich, Switzerland, 11Geneva University Hospitals, Geneva, Switzerland, 12Rigshospitalet Glostrup; Diakonhjemmet Hospital; Sørlandet Hospital, Copenhagen, Denmark, 13Section for Rheumatology, Helse Førde, Førde, Norway, 14Aarhus University, Horsens, Denmark, 15University Medical Centre Ljubljana, Ljubljana, Slovenia, 16University Medical Centre Ljubljana; University of Ljubljana, Ljubljana, Slovenia, 17Aberdeen Centre for Arthritis and Musculoskeletal Health (Epidemiology Group), University of Aberdeen, Aberdeen, United Kingdom, 18Helsinki University Hospital, Helsinki, Finland, 19Departments of Medicine and Rheumatology, Helsinki University Hospital, Helsinki, Finland, 20Rheumatology, Radboud University Medical Center, Nijmegen, Netherlands, 21Lund University and Skåne University Hospital, Lund, Sweden, 22Centre for Rheumatology Research, University Hospital, Reykjavik, Iceland, 23Faculty of Medicine, University of Iceland, Reykjavik, Iceland
Background/Purpose: Axial spondyloarthritis (axSpA) and psoriatic arthritis (PsA) are part of the spondylarthritis spectrum that can be treated with secukinumab, a fully human IgG1 monoclonal antibody targeting interleukin-17A. Real-word data on long-term secukinumab effectiveness in these two diseases are limited. In separate cohorts of axSpA and PsA patients treated with secukinumab in routine care, we aimed to assess a) 24-, and 48-month retention rates; and b) proportions of patients achieving 24-, and 48-month composite scores of remission and response.
Methods: Patients with axSpA and PsA who initiated secukinumab were included from 13 registriesparticipating in the European Spondyloarthritis (EuroSpA) Research Collaboration Network [1]. Kaplan-Meier plots with log-rank tests and Cox regression analyses were performed to assess 24-, and 48-month secukinumab retention rates overall, and compared by prior b/tsDMARD status (0/1/≥2). Comparisons of remission and response rates according to b/tsDMARDs status, were performed by logistic regression adjusted for age, gender, register, time since diagnosis and disease activity at treatment start (baseline). Missing baseline data were imputed by multivariate imputation by chained equations.
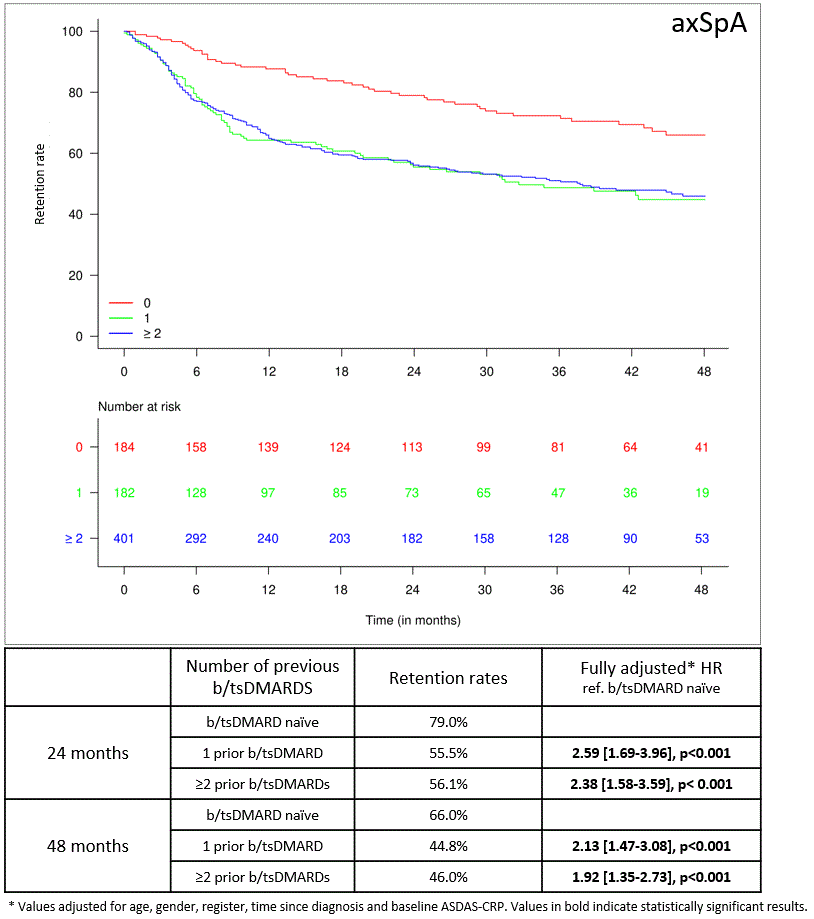
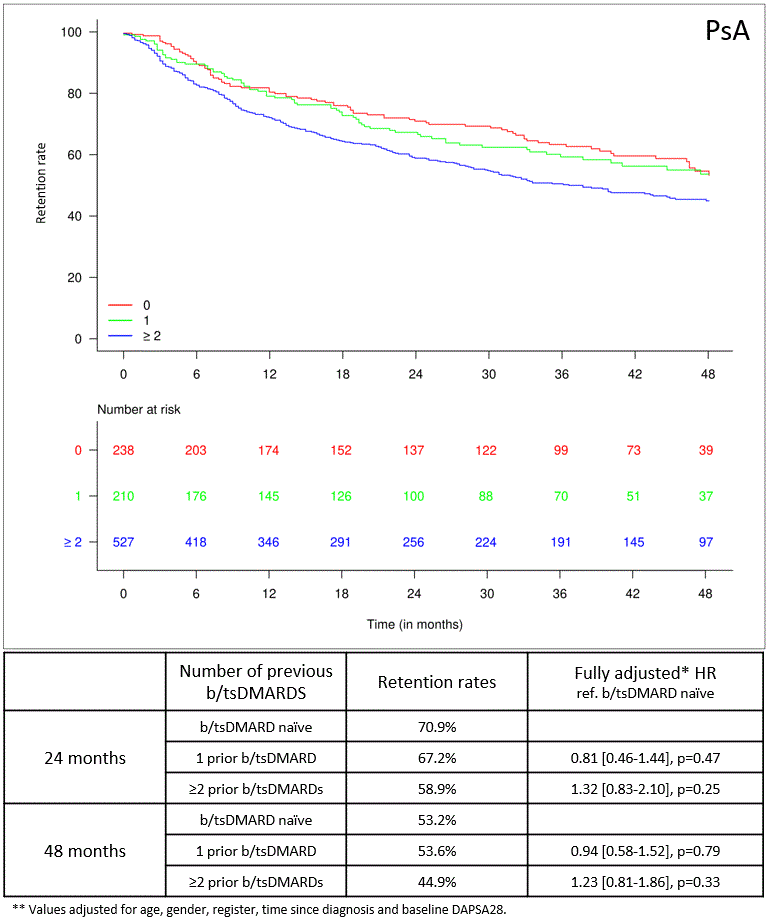
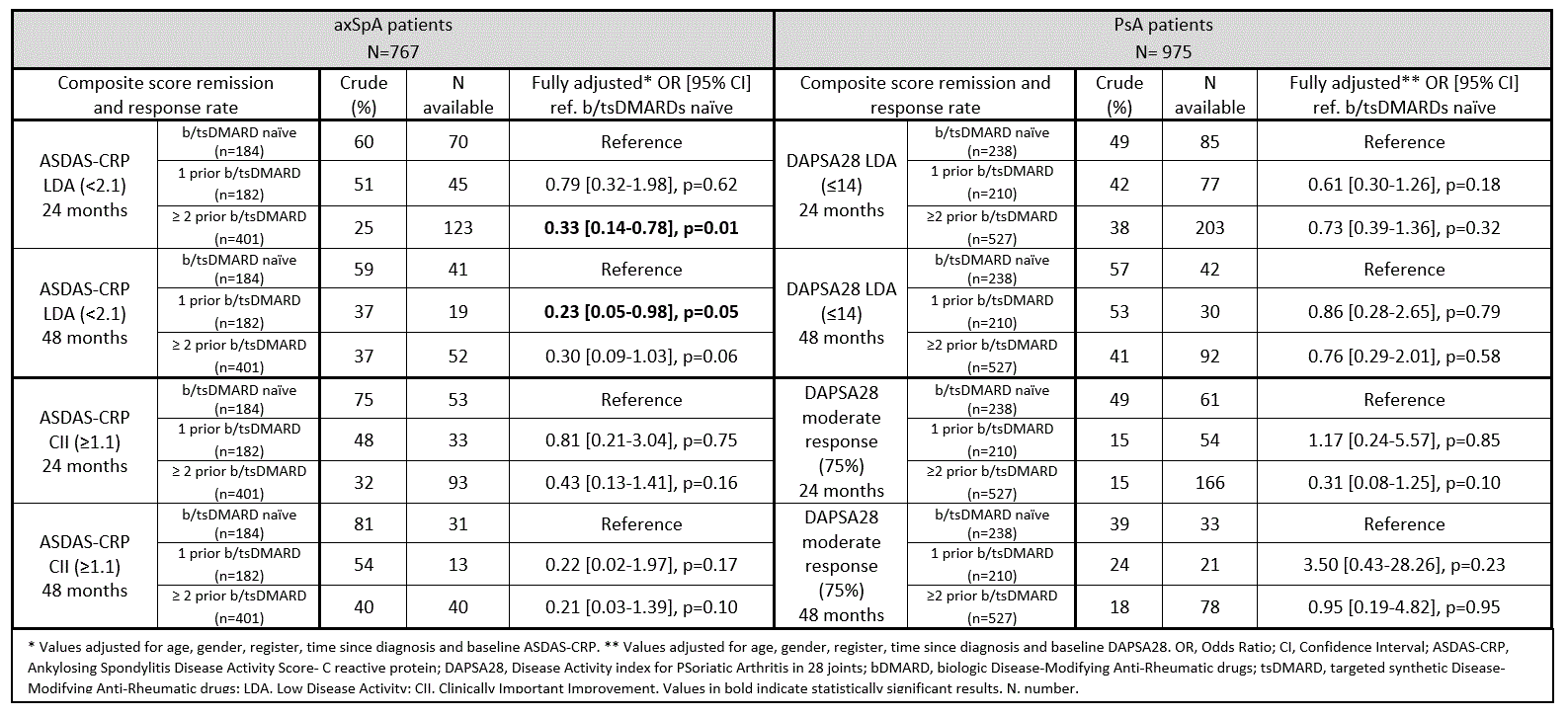
Results: A total of 767 axSpA and 975 PsA patients from 13 and 12 countries, respectively, were included. At baseline, axSpA patients had a median (IQR) age of 47 (38-55) years, were predominantly male (60%), and had high disease activity (median (IQR)Ankylosing Spondylitis Disease Activity Score (ASDAS)-CRP 3.6 (2.9-4.2)). PsA patients had a median (IQR) age of 52 (44-59) years, were predominantly female (56%), and had moderate disease activity (median (IQR) Disease Activity index for PSoriatic Arthritis in 28 joints (DAPSA28) 25.1 (16.7-37.3)). The overall 24-/48-month secukinumab retention rates were 61%/51% in axSpA, and 63%/49% in PsA patients, respectively. The 24-/48-month secukinumab retention rates in axSpA were significantly higher in bio-naïve patients than in patients treated with 1 or ≥2 prior b/tsDMARDs (Figure 1). In PsA patients, both 24-/48-month secukinumab retention rates were similar between bio-naïve patients and patients who previously received 1 prior b/tsDMARD, and numerically lower in patients having received ≥2 b/tsDMARDs (Figure 2). In axSpA patients, ASDAS-CRP low disease activity (LDA) and ASDAS-CRP clinically important improvement (CII) crude rates were numerically higher in the bio-naïve group at 24 and 48 months. After confounder adjustment, bio-naïve patients had higher rates of ASDAS-CRP LDA than patients who had previously received one or more b/tsDMARDs (Table 1). Similarly, the crude proportions of PsA patients achieving DAPSA28 LDA or moderate response were numerically (but not statistically significantly) higher in the bio-naïve group (Table 1).
Conclusion: This large real-life study showed that secukinumab retention rates after four years wereapproximately 50% in patients with axSpA and PsA. Bio-naïve patients had higher retention, remission and response rates than patients with prior b/tsDMARD exposure, particularly in axSpA.
References 1 https://eurospa.eu/.
M. Pons: Novartis, 5; S. Georgiadis: Novartis, 5; Z. Faizy Ahmadzay: Novartis, 5; M. Østergaard: AbbVie, 2, 5, 6, Amgen, 5, Boehringer-Ingelheim, 2, 6, Bristol-Myers Squibb(BMS), 2, 5, 6, Celgene, 2, 5, 6, Eli Lilly, 2, 6, Galapagos, 2, 6, Gilead, 2, 6, Hospira, 2, 6, Janssen, 2, 6, MEDAC, 6, Merck, 2, 5, 6, Novartis, 2, 5, 6, Novo Nordisk, 2, 6, Orion, 2, 6, Pfizer, 2, 6, Regeneron, 2, 6, Roche, 2, 6, Sandoz, 2, 6, Sanofi, 2, 6, UCB, 2, 6; B. Glintborg: AbbVie/Abbott, 5, Pfizer, 5, Sandoz, 5; J. Heberg: Novartis, 5; S. Nysom Christiansen: Novartis, 5, 6; S. Horskjær Rasmussen: Novartis, 5; I. Castrejon: Bristol Myers Squibb, 1, 6, Galapagos, 2, GlaxoSmithKline, 1, 6, Lilly, 1, 6, Merck Sharp & Dohme, 6, Pfizer, 1, 2, 6; F. Alonso: None; K. Laas: None; S. Vorobjov: None; N. Khmelinskii: None; J. Zavada: None; A. Ciurea: None; M. Nissen: AbbVie/Abbott, 2, Eli Lilly, 2, 12, Involved in Clinical Trial, Janssen, 2, Novartis, 6, 12, research funding paid to institution, Pfizer, 6, UCB, 2, 12, funding support to attend EULAR 2023, paid to institution; B. Michelsen: Novartis, 5; P. Mielnik: GALAPAGOS, 2; A. Loft: Ucb, 1, 6, 12, Congress participation; Z. Rotar: None; K. Perdan Pirkmajer: None; G. Macfarlane: None; G. Jones: Amgen, 5; D. Nordstrom: AbbVie/Abbott, 2, BMS, 2, Lilly, 2, MSD, 2, Novartis, 2, Pfizer, 2, UCB, 2; A. Hokkanen: MSD, 5; I. van der Horst-Bruinsma: Abbvie, 2, 5, 5, Lilly, 2, MSD, 2, 5, Novartis, 2, Pfizer, 5, UCB, 2, 5, 6; J. Karlsson Wallman: AbbVie, 5, 6, Amgen, 5, 6, Eli Lilly, 5, Novartis, 5, Pfizer, 5; B. Gudbjornsson: Nordic-Pharma, 6, Novartis, 2, 6; o. Palsson: None; M. Hetland: AbbVie/Abbott, 1, 5, Bristol-Myers Squibb(BMS), 5, Danbio, 12, Chari of Danbio registry, Eli Lilly, 5, MEDAC, 6, Novartis, 5, Pfizer, 5, 6, Sandoz, 5, 6; L. Ørnbjerg: Novartis, 5.
Background/Purpose: Axial spondyloarthritis (axSpA) and psoriatic arthritis (PsA) are part of the spondylarthritis spectrum that can be treated with secukinumab, a fully human IgG1 monoclonal antibody targeting interleukin-17A. Real-word data on long-term secukinumab effectiveness in these two diseases are limited. In separate cohorts of axSpA and PsA patients treated with secukinumab in routine care, we aimed to assess a) 24-, and 48-month retention rates; and b) proportions of patients achieving 24-, and 48-month composite scores of remission and response.
Methods: Patients with axSpA and PsA who initiated secukinumab were included from 13 registriesparticipating in the European Spondyloarthritis (EuroSpA) Research Collaboration Network [1]. Kaplan-Meier plots with log-rank tests and Cox regression analyses were performed to assess 24-, and 48-month secukinumab retention rates overall, and compared by prior b/tsDMARD status (0/1/≥2). Comparisons of remission and response rates according to b/tsDMARDs status, were performed by logistic regression adjusted for age, gender, register, time since diagnosis and disease activity at treatment start (baseline). Missing baseline data were imputed by multivariate imputation by chained equations.
Results: A total of 767 axSpA and 975 PsA patients from 13 and 12 countries, respectively, were included. At baseline, axSpA patients had a median (IQR) age of 47 (38-55) years, were predominantly male (60%), and had high disease activity (median (IQR)Ankylosing Spondylitis Disease Activity Score (ASDAS)-CRP 3.6 (2.9-4.2)). PsA patients had a median (IQR) age of 52 (44-59) years, were predominantly female (56%), and had moderate disease activity (median (IQR) Disease Activity index for PSoriatic Arthritis in 28 joints (DAPSA28) 25.1 (16.7-37.3)). The overall 24-/48-month secukinumab retention rates were 61%/51% in axSpA, and 63%/49% in PsA patients, respectively. The 24-/48-month secukinumab retention rates in axSpA were significantly higher in bio-naïve patients than in patients treated with 1 or ≥2 prior b/tsDMARDs (Figure 1). In PsA patients, both 24-/48-month secukinumab retention rates were similar between bio-naïve patients and patients who previously received 1 prior b/tsDMARD, and numerically lower in patients having received ≥2 b/tsDMARDs (Figure 2). In axSpA patients, ASDAS-CRP low disease activity (LDA) and ASDAS-CRP clinically important improvement (CII) crude rates were numerically higher in the bio-naïve group at 24 and 48 months. After confounder adjustment, bio-naïve patients had higher rates of ASDAS-CRP LDA than patients who had previously received one or more b/tsDMARDs (Table 1). Similarly, the crude proportions of PsA patients achieving DAPSA28 LDA or moderate response were numerically (but not statistically significantly) higher in the bio-naïve group (Table 1).
Conclusion: This large real-life study showed that secukinumab retention rates after four years wereapproximately 50% in patients with axSpA and PsA. Bio-naïve patients had higher retention, remission and response rates than patients with prior b/tsDMARD exposure, particularly in axSpA.
References 1 https://eurospa.eu/.

Figure 1: The 48-month retention rate for secukinumab in axSpA patients, stratified according to the number of previous b/tsDMARDs

Figure 2: The 48-month retention rate for secukinumab in PsA patients, stratified according to the number of previous b/tsDMARDs

Table 1: Composite score remission and response rates in axSpA and PsA patients treated with secukinumab
M. Pons: Novartis, 5; S. Georgiadis: Novartis, 5; Z. Faizy Ahmadzay: Novartis, 5; M. Østergaard: AbbVie, 2, 5, 6, Amgen, 5, Boehringer-Ingelheim, 2, 6, Bristol-Myers Squibb(BMS), 2, 5, 6, Celgene, 2, 5, 6, Eli Lilly, 2, 6, Galapagos, 2, 6, Gilead, 2, 6, Hospira, 2, 6, Janssen, 2, 6, MEDAC, 6, Merck, 2, 5, 6, Novartis, 2, 5, 6, Novo Nordisk, 2, 6, Orion, 2, 6, Pfizer, 2, 6, Regeneron, 2, 6, Roche, 2, 6, Sandoz, 2, 6, Sanofi, 2, 6, UCB, 2, 6; B. Glintborg: AbbVie/Abbott, 5, Pfizer, 5, Sandoz, 5; J. Heberg: Novartis, 5; S. Nysom Christiansen: Novartis, 5, 6; S. Horskjær Rasmussen: Novartis, 5; I. Castrejon: Bristol Myers Squibb, 1, 6, Galapagos, 2, GlaxoSmithKline, 1, 6, Lilly, 1, 6, Merck Sharp & Dohme, 6, Pfizer, 1, 2, 6; F. Alonso: None; K. Laas: None; S. Vorobjov: None; N. Khmelinskii: None; J. Zavada: None; A. Ciurea: None; M. Nissen: AbbVie/Abbott, 2, Eli Lilly, 2, 12, Involved in Clinical Trial, Janssen, 2, Novartis, 6, 12, research funding paid to institution, Pfizer, 6, UCB, 2, 12, funding support to attend EULAR 2023, paid to institution; B. Michelsen: Novartis, 5; P. Mielnik: GALAPAGOS, 2; A. Loft: Ucb, 1, 6, 12, Congress participation; Z. Rotar: None; K. Perdan Pirkmajer: None; G. Macfarlane: None; G. Jones: Amgen, 5; D. Nordstrom: AbbVie/Abbott, 2, BMS, 2, Lilly, 2, MSD, 2, Novartis, 2, Pfizer, 2, UCB, 2; A. Hokkanen: MSD, 5; I. van der Horst-Bruinsma: Abbvie, 2, 5, 5, Lilly, 2, MSD, 2, 5, Novartis, 2, Pfizer, 5, UCB, 2, 5, 6; J. Karlsson Wallman: AbbVie, 5, 6, Amgen, 5, 6, Eli Lilly, 5, Novartis, 5, Pfizer, 5; B. Gudbjornsson: Nordic-Pharma, 6, Novartis, 2, 6; o. Palsson: None; M. Hetland: AbbVie/Abbott, 1, 5, Bristol-Myers Squibb(BMS), 5, Danbio, 12, Chari of Danbio registry, Eli Lilly, 5, MEDAC, 6, Novartis, 5, Pfizer, 5, 6, Sandoz, 5, 6; L. Ørnbjerg: Novartis, 5.



