Poster Session C
Vasculitis
Session: (2370–2386) Vasculitis – ANCA-Associated Poster III: Biomarkers & Renal Outcomes
2374: Torque Teno Virus as a Potential Biomarker for Assessment of Immunocompetence in Patients with ANCA-associated Vasculitis
Tuesday, November 14, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- BO
Balazs Odler, MD, PhD (he/him/his)
Medical University of Graz
Graz, AustriaDisclosure information not submitted.
Abstract Poster Presenter(s)
Balazs Odler1, Regina Riedl1, Jacinta Lee2, Laura Cooney3, Peter Merkel4, William St.Clair5, Duvuru Geetha6, Paul Monach7, David Jayne2, Rona Smith2, Paul A. Lyons2, Mark Little8, Salem Almaani9, Alexander Rosenkranz1, Ulrich Specks10, John Stone11 and Andreas Kronbichler12, 1Medical University of Graz, Graz, Austria, 2University of Cambridge, Cambridge, United Kingdom, 3Immune Tolerance Network, Seattle, WA, 4University of Pennsylvania, Philadelphia, PA, 5Duke University Medical Center, Durham, NC, 6Johns Hopkins University, Baltimore, MD, 7VA Boston Healthcare System, Boston, MA, 8Trinity College Dublin, Dublin, Ireland, 9Ohio State University Medical Center, Columbus, OH, 10Mayo Clinic, Rochester, MN, 11Massachusetts General Hospital Rheumatology Unit, Harvard Medical School, Concord, MA, 12Department of Internal Medicine IV (Nephrology and Hypertension), Innsbruck, Austria
Background/Purpose: ANCA-associated vasculitis (AAV) is a life-threatening disorder characterized by a relapsing-remitting disease course necessitating immunosuppression to control disease activity. Balancing the risks of infection against maintaining good disease control is crucial and yet often challenging. There is a lack of validated biomarkers to tailor immunosuppression to minimize therapy-related complications while maintaining a relapse-free period. The monitoring of Torque teno virus (TTV) has been successfully used in transplant medicine to predict the risk of organ rejection and infectious complications. However, the presence and role of TTV in association with clinical outcomes in AAV has yet to be reported.
Methods: Consecutive plasma samples (N=915) of 81 patients with AAV from the RAVE trial were used for assessment of the TTV load. Participants received either rituximab (RTX, N=40) or cyclophosphamide (CYC, N=41) followed by azathioprine (AZA). The TTV DNA was quantified using a real-time polymerase chain reaction method (TTV R-GENE®, BioMérieux) at multiple study visits. Change in TTV load and differences between non-relapsing and relapsing patients were investigated during a follow-up period of 1080 days.
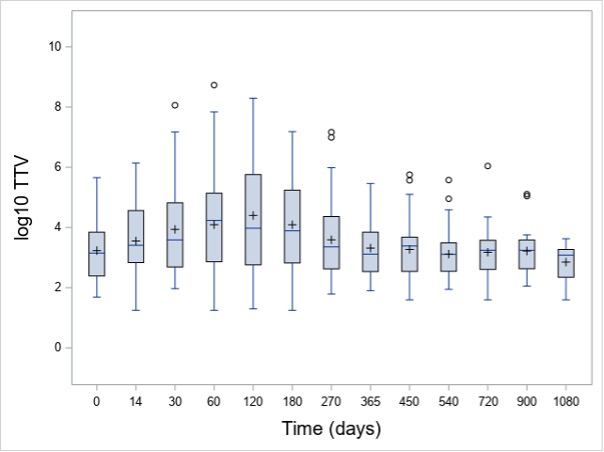
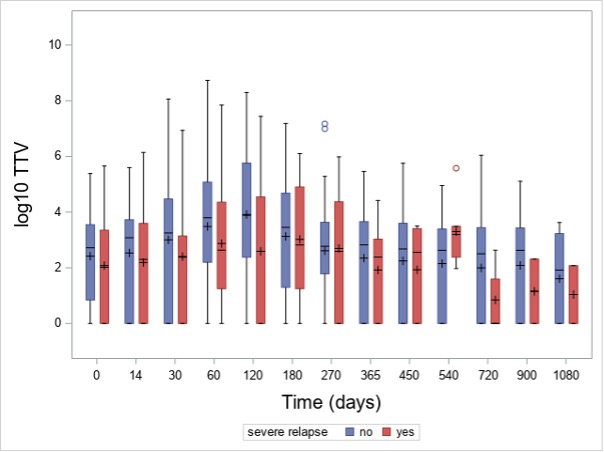
Results: In total, 915 TTV quantifications were performed. At baseline, 72% (N=58) of patients had detectable TTV in the peripheral blood. The baseline median TTV load of all patients was 3 x 102 c/mL (interquartile range [IQR]: 0 – 3 x 103 c/mL). TTV load increased after initiation of immunosuppression and peaked at day 120 (median 2 x 103 c/mL, IQR: 2 x 102 – 5 x 105; Figure 1) and was detectable in 80% of the patients. Patients receiving RTX as a remission induction therapy showed a non-significant higher TTV load during the first 180 days in comparison to CYC/AZA-treated individuals at any visits. Patients with disease relapse had a lower TTV load at day 120 as compared to those without relapse (median 4 x 102 [IQR: 0 – 3 x 104] c/mL vs median 8 x 103 [IQR: 2 x 102 – 6 x 105] c/mL, p=0.036, respectively; Figure 2). For the 25 severe relapses, the median time to relapse was 339 (IQR: 232-516) days. Six relapses occured before 120 day, while 19 after day 120.
Conclusion: Among patients with AAV, TTV load reflects the intensity of immunosuppression and is associated with disease relapses. These results suggest that TTV might be a biomarker for the assessment of immunocompetence and identifying patients at risk of relapse in AAV.
B. Odler: CSL Vifor, 6, Delta4, 1, Otsuka Pharmaceuticals, 5, 6; R. Riedl: None; J. Lee: None; L. Cooney: None; P. Merkel: AbbVie/Abbott, 5, Amgen, 2, 5, ArGenx, 2, AstraZeneca, 2, 5, Boehringer-Ingelheim, 2, 5, Bristol-Myers Squibb(BMS), 2, 5, Cabaletta, 2, CSL Behring, 2, Eicos, 5, Electra, 5, Genentech, 5, GlaxoSmithKlein(GSK), 2, 5, HiBio, 2, InflaRx, 2, 5, Janssen, 2, Jubilant, 2, Kyverna, 2, 11, MiroBio, 2, Neutrolis, 5, Novartis, 2, NS Pharma, 2, Q32, 2, Regeneron, 2, Sanofi, 2, Sparrow, 2, Takeda, 2, 5, UpToDate, 9, Visterra, 2; W. St.Clair: CSL Behring, 2, Horizon Therapeutics, 2, 5, Related Sciences, 2, Resolve Therapeutics, 2, Sonoma biotherapeutics, 2, Up To Date, 9; D. Geetha: Amgen, 2, Aurinia, 2, calliditas, 2, chemocentryx, 2, GlaxoSmithKlein(GSK), 2; P. Monach: Genentech, 12, Lecture with honorarium, HI-Bio, 2; D. Jayne: AstraZeneca, 2, Aurinia, 4, Boehringer Ingelheim, 2, Chinook, 2, CSL Vifor, 2, Roche, 2; R. Smith: GlaxoSmithKlein(GSK), 5, Union Therapeutics, 5; P. Lyons: None; M. Little: Vifor, 5; S. Almaani: Amgen, 2, Aurinia, 2, Chemocentryx, 2, Kezar, 2, Otsuka, 2; A. Rosenkranz: None; U. Specks: Amgen, 2, AstraZeneca, 2, 5, Boehringer-Ingelheim, 1, Bristol-Myers Squibb(BMS), 5, ChemoCentryx, 2, Genentech, 5, GlaxoSmithKlein(GSK), 5; J. Stone: Abvie, 2, Amgen, 1, 2, Argenx, 2, Aztrazeneca, 2, Bristol Myers Squibb, 2, 5, Celgene, 2, Chemocentryx, 2, Chugai, 2, GSK, 2, Horizon Therapeutics, 1, 2, 5, InflaRx, 2, IQVIA, 1, 2, Kyverna, 2, Mirabio, 2, NIH, 5, Novartis, 2, PPD, 2, Prometheus, 2, Q32, 2, Regeneron, 2, Roche-Genentech, 2, Roivant, 2, Sanofi, 2, 5, Spruce Biosciences, 2, Star Therapeutics, 2, Steritas, 12, Chair, Scientific Advisory Board (no fiduciary responsibilities), ZenasBio, 2; A. Kronbichler: Catalyst Biosciences, 2, CSL Vifor, 2, Delta4, 2, GlaxoSmithKlein(GSK), 2, Otsuka, 2, Waiden Biosciences, 2.
Background/Purpose: ANCA-associated vasculitis (AAV) is a life-threatening disorder characterized by a relapsing-remitting disease course necessitating immunosuppression to control disease activity. Balancing the risks of infection against maintaining good disease control is crucial and yet often challenging. There is a lack of validated biomarkers to tailor immunosuppression to minimize therapy-related complications while maintaining a relapse-free period. The monitoring of Torque teno virus (TTV) has been successfully used in transplant medicine to predict the risk of organ rejection and infectious complications. However, the presence and role of TTV in association with clinical outcomes in AAV has yet to be reported.
Methods: Consecutive plasma samples (N=915) of 81 patients with AAV from the RAVE trial were used for assessment of the TTV load. Participants received either rituximab (RTX, N=40) or cyclophosphamide (CYC, N=41) followed by azathioprine (AZA). The TTV DNA was quantified using a real-time polymerase chain reaction method (TTV R-GENE®, BioMérieux) at multiple study visits. Change in TTV load and differences between non-relapsing and relapsing patients were investigated during a follow-up period of 1080 days.
Results: In total, 915 TTV quantifications were performed. At baseline, 72% (N=58) of patients had detectable TTV in the peripheral blood. The baseline median TTV load of all patients was 3 x 102 c/mL (interquartile range [IQR]: 0 – 3 x 103 c/mL). TTV load increased after initiation of immunosuppression and peaked at day 120 (median 2 x 103 c/mL, IQR: 2 x 102 – 5 x 105; Figure 1) and was detectable in 80% of the patients. Patients receiving RTX as a remission induction therapy showed a non-significant higher TTV load during the first 180 days in comparison to CYC/AZA-treated individuals at any visits. Patients with disease relapse had a lower TTV load at day 120 as compared to those without relapse (median 4 x 102 [IQR: 0 – 3 x 104] c/mL vs median 8 x 103 [IQR: 2 x 102 – 6 x 105] c/mL, p=0.036, respectively; Figure 2). For the 25 severe relapses, the median time to relapse was 339 (IQR: 232-516) days. Six relapses occured before 120 day, while 19 after day 120.
Conclusion: Among patients with AAV, TTV load reflects the intensity of immunosuppression and is associated with disease relapses. These results suggest that TTV might be a biomarker for the assessment of immunocompetence and identifying patients at risk of relapse in AAV.

Figure 1. Change in TTV load after randomization in the study cohort of patients with ANCA-associated vasculitis. Box plots represent the interquartile range, while plus signs indicate the mean value.

Figure 2. TTV load in the study cohort of ANCA-associated vasculitis patients with and without a disease relapse over time. Box plots represent the interquartile range, while plus signs indicate the mean value.
B. Odler: CSL Vifor, 6, Delta4, 1, Otsuka Pharmaceuticals, 5, 6; R. Riedl: None; J. Lee: None; L. Cooney: None; P. Merkel: AbbVie/Abbott, 5, Amgen, 2, 5, ArGenx, 2, AstraZeneca, 2, 5, Boehringer-Ingelheim, 2, 5, Bristol-Myers Squibb(BMS), 2, 5, Cabaletta, 2, CSL Behring, 2, Eicos, 5, Electra, 5, Genentech, 5, GlaxoSmithKlein(GSK), 2, 5, HiBio, 2, InflaRx, 2, 5, Janssen, 2, Jubilant, 2, Kyverna, 2, 11, MiroBio, 2, Neutrolis, 5, Novartis, 2, NS Pharma, 2, Q32, 2, Regeneron, 2, Sanofi, 2, Sparrow, 2, Takeda, 2, 5, UpToDate, 9, Visterra, 2; W. St.Clair: CSL Behring, 2, Horizon Therapeutics, 2, 5, Related Sciences, 2, Resolve Therapeutics, 2, Sonoma biotherapeutics, 2, Up To Date, 9; D. Geetha: Amgen, 2, Aurinia, 2, calliditas, 2, chemocentryx, 2, GlaxoSmithKlein(GSK), 2; P. Monach: Genentech, 12, Lecture with honorarium, HI-Bio, 2; D. Jayne: AstraZeneca, 2, Aurinia, 4, Boehringer Ingelheim, 2, Chinook, 2, CSL Vifor, 2, Roche, 2; R. Smith: GlaxoSmithKlein(GSK), 5, Union Therapeutics, 5; P. Lyons: None; M. Little: Vifor, 5; S. Almaani: Amgen, 2, Aurinia, 2, Chemocentryx, 2, Kezar, 2, Otsuka, 2; A. Rosenkranz: None; U. Specks: Amgen, 2, AstraZeneca, 2, 5, Boehringer-Ingelheim, 1, Bristol-Myers Squibb(BMS), 5, ChemoCentryx, 2, Genentech, 5, GlaxoSmithKlein(GSK), 5; J. Stone: Abvie, 2, Amgen, 1, 2, Argenx, 2, Aztrazeneca, 2, Bristol Myers Squibb, 2, 5, Celgene, 2, Chemocentryx, 2, Chugai, 2, GSK, 2, Horizon Therapeutics, 1, 2, 5, InflaRx, 2, IQVIA, 1, 2, Kyverna, 2, Mirabio, 2, NIH, 5, Novartis, 2, PPD, 2, Prometheus, 2, Q32, 2, Regeneron, 2, Roche-Genentech, 2, Roivant, 2, Sanofi, 2, 5, Spruce Biosciences, 2, Star Therapeutics, 2, Steritas, 12, Chair, Scientific Advisory Board (no fiduciary responsibilities), ZenasBio, 2; A. Kronbichler: Catalyst Biosciences, 2, CSL Vifor, 2, Delta4, 2, GlaxoSmithKlein(GSK), 2, Otsuka, 2, Waiden Biosciences, 2.



