Poster Session C
Vasculitis
Session: (2387–2424) Vasculitis – Non-ANCA-Associated & Related Disorders Poster III
2391: Epidemiology and Predictors of Relapse in Giant Cell Arteritis: Results of the ARTESER Register
Tuesday, November 14, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- JN
Javier Narvaez, MD, PhD
Hospital Universitario de Bellvitge
Barcelona, SpainDisclosure information not submitted.
Abstract Poster Presenter(s)
Javier Narvaez1, Jesús Sanchez-Costa2, Iñigo Hernández-Rodríguez3, Rafael Benito Melero-Gonzalez4, Eugenio De Miguel5, Irene Monjo6, Maite silva díaz7, LUCIA SILVA FERNANDEZ8, Joaquin Maria Belzunegui Otano9, Jesus Alejandro Valero-Jaimes10, Clara Moriano Morales11, Ismael González12, Julio Sanchez Martin13, Tomas Almorza Hidalgo14, Judit Lluch15, Eva Galindez-Agirregoikoa16, Itziar Calvo-Zorrilla16, Vicente Aldasoro17, Javier Mendizabal18, LYDIA ABASOLO19, PIA LOIS20, Javier Loricera13, Noemi Garrido-Puñal21, ALBERTO MARIANO RUIZ ROMAN22, Santos Castañeda23, Cristina Valero23, Patricia Moya Albarado24, Hector Corominas25, Maria J. García-Villanueva26, Carmen Larena27, Paula V. Estrada-Alarcón28, Vanessa Navarro29, Carles Galisteo30, Anne Riveros-Frutos31, Francisco Miguel Ortiz Sanjuan32, Selene Labrada-Arrabal33, María Alcalde34, Clara Molina Almela35, Carlos Garcia Porrua36, Maria García-González37, Margarida Rocha38, Antonio Juan Mas39, Miguel A Gonzalez-Gay40 and Ricardo Blanco41, 1Hospital Universitario de Bellvitge, Barcelona, Spain, 2Unidad de Investigación de la Sociedad Española de Reumatología, Madrid, Spain, 3Hospital do Meixoeiro. Complejo Hospitalario Universitario de Vigo, Vigo, Spain, 4CHU Vigo, O Carballino, Spain, 5Hospital Universitario La Paz, Madrid, Spain, 6University Hospital La Paz, Madrid, Spain, 7Rheumatology department, Complexo Hospitalario Universitario A Coruña (CHUAC). Instituto de Investigación Biomédica A Coruña (INIBIC)., A Coruña, Spain, 8Complejo Hospitalario Universitario de la Coruña, Pontevedra, Spain, 9University Hospital Donostia, Donostia-San Sebasti, Spain, 10Hospital Bidasoa, Irún, Spain, 11Rheumatology, Hospital Universitario de León, León, Spain, 12Hospital Universitario de León, León, Spain, 13Hospital Universitario Marqués de Valdecilla, Santander, Spain, 14Hospital Universitario 12 de Octubre, Madrid, Spain, 15Department of Rheumatology, Hospital Universitario de Bellvitge, Barcelona, Spain, 16Basurto University Hospital, Bilbao, Spain, 17Hospital Universitario de Navarra, Pamplona, Spain, 18Complejo Hospitalario de Navarra, Pamplona, Spain, 19Hospital Clínico San Carlos, Madrid, Spain, 20Hospital Clinico San Carlos, Madrid, Spain, 21Hospital Universitario Virgen del Rocío, Sevilla, Spain, 22Department of Rheumatology, Hospital Universitario Virgen del Rocío, Sevilla, Spain, 23Hospital Universitario de la Princesa, Madrid, Spain, 24Hospital Universitario de Sant Pau, Barcelona, Spain, 25Hospital Universitario Sant Pau, Rheumatology, Barcelona, Spain, 26Hospital Ramón y Cajal, Madrid, Spain, 27Hospital Universitario Ramon y Cajal, Madrid, Spain, 28Hospital de San Juan Despí Moisès Broggi, Barcelona, Spain, 29Hospital de Sant Joan Despí – Moises Broggi, Barcelona, Spain, 30Hospital Universitario Parc Taulí, Sabadell, Spain, 31Hospital Universitario Germans Trias i Pujol, Barcelona, Spain, 32Hospital Universitario y Politecnico La Fe, Valencia, Spain, 33Hospital del Mar, Barcelona, Spain, 34Hospital Severo Ochoa, Madrid, Spain, 35Hospital General de Valencia, Valencia, Spain, 36Hospital Universitario Lucus Augusti, Lugo, Spain, 37Hospital Universitario de Canarias, Santa Cruz de Tenerife, Spain, 38Osakidetza, Bilbo, Spain, 39Hospital Universitario Son Llàtzer, Mallorca, Spain, 40IDIVAL and School of Medicine, UC, Santander; Department of Rheumatology, IIS-Fundación Jiménez Díaz, Madrid, Santander, Spain, 41Hospital Universitario Marqués de Valdecilla, IDIVAL, Santander, Spain
Background/Purpose: The relapse rate of treated patients with giant cell arteritis (GCA) varied widely in observational series and randomized controlled trials. This study aimed to estimate the frequency and timing of relapse, the prevalence of multiple relapses, the characteristics of flares, and the predictors of relapse in a large cohort of Spanish patients with GCA.
Methods: All patients in the ARTESER (Registry of Giant Cell Arteritis Patients of the Spanish Society of Rheumatology) were reviewed. ARTESER is a multicentre observational retrospective longitudinal study conducted in 26 hospitals that included all consecutive patients diagnosed with GCA between 1 June 2013 and 29 March 2019. All cases were aged 50 years or older and had a confirmed diagnosis of GCA, that is, they met at least one of the following criteria: objective confirmation of the presence of vasculitis in a diagnostic test, at least three 1990 ACR criteria for GCA satisfied, and/or a diagnosis made based on the clinical judgment of the investigator. We diagnosed disease relapses if all the following criteria were satisfied: 1) reappearance of signs/symptoms of GCA and/or polymyalgia rheumatica (PMR); 2) resolution of signs/symptoms after increasing glucocorticoids; 3) presence of raised acute-phase reactants ESR and/or CRP, and 4) exclusion of other causes. The data were obtained by review of medical records.
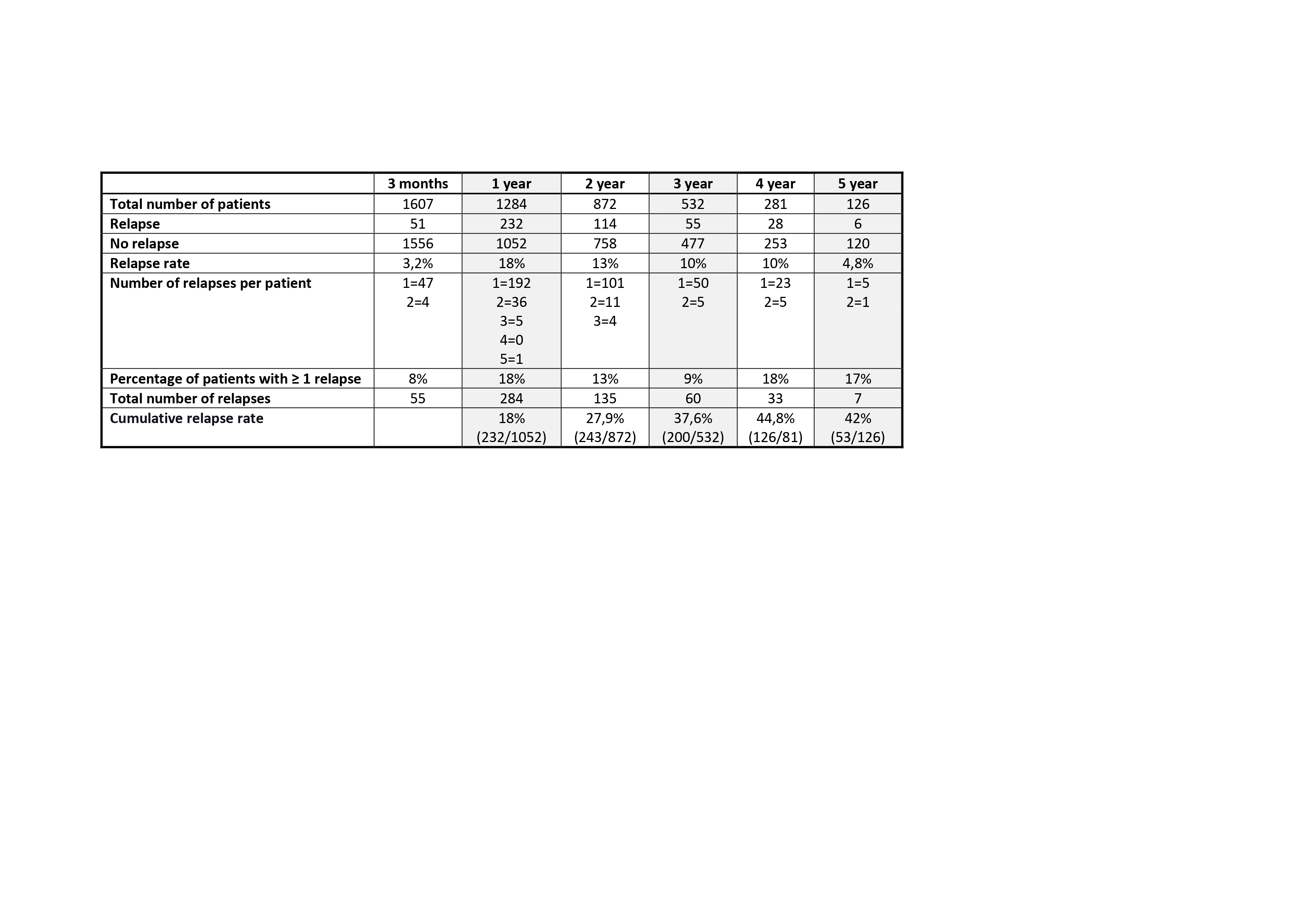
Results: In total, 1675 patients were included. They were predominantly female (70.3%), with a mean (±SD) age at diagnosis of 76.9±8.1 years. Of them, 1284 patients were followed up for one or more years. During follow-up, 574 relapses were observed in 334 (26%) patients. One-year, 2-year, 3-year, and 4-year cumulative relapse rates were 18%, 27.9%, 37.6%, and 44.8%, respectively (see Table 1). Most relapses (81.2%) occurred within the first two years after diagnosis (55.3% in the first year and 26% during the second). The mean dose of prednisone at the first relapse was 13.95 mg/day (IQR25%-75%: 5-20 mg). Fifteen percent (34/232) of patients experienced > 1 flare within the first year of treatment and 10.5% (12/114) during the second year. The majority of flares corresponded to a minor relapse according to the EULAR consensus definitions for disease activity states in GCA (minor relapses 73% / major relapses 27%) In the multivariate analysis, we did not identify any clinical factor at diagnosis that was a predictor of relapse during the first three years of evolution: nor the age, sex, presence of cranial manifestations, severe ischemic complications, large vessel involvement, or a strong initial systemic inflammatory response. By contrast, the administration of intravenous (IV)methylprednisolone (MP) boluses was negatively associated with the occurrence of relapses (OR: 0.356, 95% CI 0.182 to 0.696; p< 0.01).
Conclusion: Relapses among patients with GCA are common, mainly during the first two years after diagnosis. Between 8 and 18% of patients had multiple relapses. Despite the relevance of this problem, we do not yet have any predictive factors to identify patients with a higher risk of flare. Induction treatment with high-dose IV MP boluses appears to decrease the risk of relapses.
J. Narvaez: None; J. Sanchez-Costa: None; I. Hernández-Rodríguez: None; R. Melero-Gonzalez: None; E. De Miguel: None; I. Monjo: Amgen, 6, Gedeon Richter, 6, Janssen, 6, Novartis, 6, Roche, 6, UCB, 6; M. silva díaz: None; L. SILVA FERNANDEZ: None; J. Belzunegui Otano: None; J. Valero-Jaimes: None; C. Moriano Morales: None; I. González: None; J. Sanchez Martin: None; T. Almorza Hidalgo: None; J. Lluch: None; E. Galindez-Agirregoikoa: None; I. Calvo-Zorrilla: None; V. Aldasoro: None; J. Mendizabal: None; L. ABASOLO: None; P. LOIS: None; J. Loricera: None; N. Garrido-Puñal: None; A. RUIZ ROMAN: None; S. Castañeda: None; C. Valero: None; P. Moya Albarado: None; H. Corominas: None; M. García-Villanueva: AstraZeneca, 6, GSK, 6, Otsuka, 1; C. Larena: None; P. Estrada-Alarcón: None; V. Navarro: None; C. Galisteo: None; A. Riveros-Frutos: None; F. Ortiz Sanjuan: None; S. Labrada-Arrabal: None; M. Alcalde: None; C. Molina Almela: None; C. Garcia Porrua: None; M. García-González: None; M. Rocha: None; A. Mas: None; M. Gonzalez-Gay: AbbVie/Abbott, 5, 6, Amgen, 5, 6, Pfizer, 5, 6; R. Blanco: AbbVie, 5, 6, Amgen, 6, AstraZeneca, 2, BMS, 6, Eli Lilly, 6, Galapagos, 2, 6, Janssen, 2, 6, MSD, 6, Novartis, 2, 6, Pfizer, 2, 6, Roche, 5, 6, Sanofi, 6.
Background/Purpose: The relapse rate of treated patients with giant cell arteritis (GCA) varied widely in observational series and randomized controlled trials. This study aimed to estimate the frequency and timing of relapse, the prevalence of multiple relapses, the characteristics of flares, and the predictors of relapse in a large cohort of Spanish patients with GCA.
Methods: All patients in the ARTESER (Registry of Giant Cell Arteritis Patients of the Spanish Society of Rheumatology) were reviewed. ARTESER is a multicentre observational retrospective longitudinal study conducted in 26 hospitals that included all consecutive patients diagnosed with GCA between 1 June 2013 and 29 March 2019. All cases were aged 50 years or older and had a confirmed diagnosis of GCA, that is, they met at least one of the following criteria: objective confirmation of the presence of vasculitis in a diagnostic test, at least three 1990 ACR criteria for GCA satisfied, and/or a diagnosis made based on the clinical judgment of the investigator. We diagnosed disease relapses if all the following criteria were satisfied: 1) reappearance of signs/symptoms of GCA and/or polymyalgia rheumatica (PMR); 2) resolution of signs/symptoms after increasing glucocorticoids; 3) presence of raised acute-phase reactants ESR and/or CRP, and 4) exclusion of other causes. The data were obtained by review of medical records.
Results: In total, 1675 patients were included. They were predominantly female (70.3%), with a mean (±SD) age at diagnosis of 76.9±8.1 years. Of them, 1284 patients were followed up for one or more years. During follow-up, 574 relapses were observed in 334 (26%) patients. One-year, 2-year, 3-year, and 4-year cumulative relapse rates were 18%, 27.9%, 37.6%, and 44.8%, respectively (see Table 1). Most relapses (81.2%) occurred within the first two years after diagnosis (55.3% in the first year and 26% during the second). The mean dose of prednisone at the first relapse was 13.95 mg/day (IQR25%-75%: 5-20 mg). Fifteen percent (34/232) of patients experienced > 1 flare within the first year of treatment and 10.5% (12/114) during the second year. The majority of flares corresponded to a minor relapse according to the EULAR consensus definitions for disease activity states in GCA (minor relapses 73% / major relapses 27%) In the multivariate analysis, we did not identify any clinical factor at diagnosis that was a predictor of relapse during the first three years of evolution: nor the age, sex, presence of cranial manifestations, severe ischemic complications, large vessel involvement, or a strong initial systemic inflammatory response. By contrast, the administration of intravenous (IV)methylprednisolone (MP) boluses was negatively associated with the occurrence of relapses (OR: 0.356, 95% CI 0.182 to 0.696; p< 0.01).
Conclusion: Relapses among patients with GCA are common, mainly during the first two years after diagnosis. Between 8 and 18% of patients had multiple relapses. Despite the relevance of this problem, we do not yet have any predictive factors to identify patients with a higher risk of flare. Induction treatment with high-dose IV MP boluses appears to decrease the risk of relapses.

J. Narvaez: None; J. Sanchez-Costa: None; I. Hernández-Rodríguez: None; R. Melero-Gonzalez: None; E. De Miguel: None; I. Monjo: Amgen, 6, Gedeon Richter, 6, Janssen, 6, Novartis, 6, Roche, 6, UCB, 6; M. silva díaz: None; L. SILVA FERNANDEZ: None; J. Belzunegui Otano: None; J. Valero-Jaimes: None; C. Moriano Morales: None; I. González: None; J. Sanchez Martin: None; T. Almorza Hidalgo: None; J. Lluch: None; E. Galindez-Agirregoikoa: None; I. Calvo-Zorrilla: None; V. Aldasoro: None; J. Mendizabal: None; L. ABASOLO: None; P. LOIS: None; J. Loricera: None; N. Garrido-Puñal: None; A. RUIZ ROMAN: None; S. Castañeda: None; C. Valero: None; P. Moya Albarado: None; H. Corominas: None; M. García-Villanueva: AstraZeneca, 6, GSK, 6, Otsuka, 1; C. Larena: None; P. Estrada-Alarcón: None; V. Navarro: None; C. Galisteo: None; A. Riveros-Frutos: None; F. Ortiz Sanjuan: None; S. Labrada-Arrabal: None; M. Alcalde: None; C. Molina Almela: None; C. Garcia Porrua: None; M. García-González: None; M. Rocha: None; A. Mas: None; M. Gonzalez-Gay: AbbVie/Abbott, 5, 6, Amgen, 5, 6, Pfizer, 5, 6; R. Blanco: AbbVie, 5, 6, Amgen, 6, AstraZeneca, 2, BMS, 6, Eli Lilly, 6, Galapagos, 2, 6, Janssen, 2, 6, MSD, 6, Novartis, 2, 6, Pfizer, 2, 6, Roche, 5, 6, Sanofi, 6.



