Poster Session B
Pediatric autoimmune diseases: Kawasaki disease, juvenile dermatomyositis and juvenile localized scleroderma
Session: (1221–1255) Pediatric Rheumatology – Clinical Poster II: Connective Tissue Disease
1250: Local Inflammatory Response Mediated by the Homing of Tfh17 Cells Is Involved in Tissue Injury of Immunoglobulin a Vasculitis
Monday, November 13, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- XM
Xiaoxue Ma
The First Hospital of China Medical University
Shenyang, ChinaDisclosure information not submitted.
Abstract Poster Presenter(s)
Xiaoxue Ma, Qinglian Jiang and Xuyang Chi, The First Hospital of China Medical University, Shenyang, China
Background/Purpose: Immunoglobulin A vasculitis (IgAV), also named Henoch–Schönlein purpura, is a systemic vasculitis characterized by the deposition of IgA1-dominant immune complexes in small vessels that often involves the skin, joints, gastrointestinal tract, and kidney. Research indicated increased frequencies of circulating activated B cells and plasmablasts in IgAV, which may serve as the source of
the rising IgA levels. T follicular helper (Tfh) 17 cells are considered to support B cells to switch to high-affinity IgA production. Previous study has confirmed that Tfh17 cells increase in the peripheral blood of IgAV patients. To evaluate the pathological role and the contribution in local tissue damage of Tfh17 cells in IgAV, we investigated the mechanism responsible for the differentiation of Tfh17 and the production of IgA in IgAV patients and IgAV rats respectively, and explored how to ameliorate IgAV by modulating Tfh17 generation.
Methods: Renal biopsy samples and peripheral blood mononuclear cells from IgAV patients were analyzed respectively by immunofluorescence staining and flow cytometry. In vitro culture was performed to assess the modulation of cytokine-induced phenotypes. IgAV rat model was established by intragastric administration of mixed solution, intraperitoneal injection of ovalbumin and Freund's adjuvant. IgAV rats were used to explore the therapeutic effects of regulating Tfh17 cells. Serum cytokine and IgA levels were measured by ELISA while histopathological changes were evaluated by H&E and PAS staining. Flow cytometry and immunofluorescence staining were used to detect T cell and GC B cell phenotypes and homing characteristics in circulation and tissues of IgAV rats.
Results: Frequency of CD4+CXCR5+CCR6+Tfh17 cells were increased in the circulation and organization of IgAV patients and associated with disease severity. The increased expressions of CD103, CCR6, CCR9 lead to the homing and residence characteristics of Tfh17 cells, resulting in local inflammatory response. Suppression of Tfh17 cells reduced the production of IgA and greatly ameliorated clinical symptoms and decreased IgA deposition and mesangial proliferation in the kidney in IgAV rats.
Conclusion: Our findings suggest that suppression of Tfh17 differentiation can alleviate IgA-mediated vasculitis and inflammatory tissue damage, which may permit the development of tailored medicines for treating IgAV.
.gif)
.gif)
X. Ma: None; Q. Jiang: None; X. Chi: None.
Background/Purpose: Immunoglobulin A vasculitis (IgAV), also named Henoch–Schönlein purpura, is a systemic vasculitis characterized by the deposition of IgA1-dominant immune complexes in small vessels that often involves the skin, joints, gastrointestinal tract, and kidney. Research indicated increased frequencies of circulating activated B cells and plasmablasts in IgAV, which may serve as the source of
the rising IgA levels. T follicular helper (Tfh) 17 cells are considered to support B cells to switch to high-affinity IgA production. Previous study has confirmed that Tfh17 cells increase in the peripheral blood of IgAV patients. To evaluate the pathological role and the contribution in local tissue damage of Tfh17 cells in IgAV, we investigated the mechanism responsible for the differentiation of Tfh17 and the production of IgA in IgAV patients and IgAV rats respectively, and explored how to ameliorate IgAV by modulating Tfh17 generation.
Methods: Renal biopsy samples and peripheral blood mononuclear cells from IgAV patients were analyzed respectively by immunofluorescence staining and flow cytometry. In vitro culture was performed to assess the modulation of cytokine-induced phenotypes. IgAV rat model was established by intragastric administration of mixed solution, intraperitoneal injection of ovalbumin and Freund's adjuvant. IgAV rats were used to explore the therapeutic effects of regulating Tfh17 cells. Serum cytokine and IgA levels were measured by ELISA while histopathological changes were evaluated by H&E and PAS staining. Flow cytometry and immunofluorescence staining were used to detect T cell and GC B cell phenotypes and homing characteristics in circulation and tissues of IgAV rats.
Results: Frequency of CD4+CXCR5+CCR6+Tfh17 cells were increased in the circulation and organization of IgAV patients and associated with disease severity. The increased expressions of CD103, CCR6, CCR9 lead to the homing and residence characteristics of Tfh17 cells, resulting in local inflammatory response. Suppression of Tfh17 cells reduced the production of IgA and greatly ameliorated clinical symptoms and decreased IgA deposition and mesangial proliferation in the kidney in IgAV rats.
Conclusion: Our findings suggest that suppression of Tfh17 differentiation can alleviate IgA-mediated vasculitis and inflammatory tissue damage, which may permit the development of tailored medicines for treating IgAV.
.gif)
Circulating CD4+CXCR5+CCR6+ Tfh17 cells are increased and associated with IgAV disease severity. PBMCs and serum were isolated from the peripheral blood of IgAV patients and HCs without incubation. (A) Representative flow cytometry plots showing the percentage of circulating PD-1+CXCR5+ Tfh cells in CD3+CD4+ T cells. (B) Scatter plots showing the frequencies of circulating PD-1+CXCR5+ Tfh and CD69+ activated cTfh cells in CD3+CD4+ T cells. (C, D) Representative flow cytometry plots and scatter plots showing the percentages of cTfh1, cTfh2, and cTfh17 subsets in circulating CD3+CD4+CXCR5+ Tfh cells. (E) Serum levels of IL-17A and IL-21 evaluated by ELISA (25 of 40 IgAV patients were randomly selected and tested). Correlations of (F) frequencies of CD19+ B cells in lymphocytes (32 of 40 IgAV patients were randomly selected and tested) and (G) serum CRP levels with the percentages of cTfh cells and cTfh17 cells from IgAV patients. (H) Frequencies of circulating CXCR3−CCR6+ Tfh17 cells in circulating CD3+CD4+CXCR5+ Tfh cells from HC and IgAV patients grouped by the organ involvement (OI) number or the presence of gastrointestinal bleeding (GIB). ns: not significant; *p<0.05; **p<0.01; ***p<0.001; ****p<0.0001.
.gif)
Suppressing Tfh17 using tocilizumab ameliorates symptoms in the IgAV rat. SD rats were respectively assigned to HC, IgAV, and TCZ groups. Representative photographs showing (A) hemorrhagic rash and (B) bloody stool from the three groups. Representative immunofluorescence images (C) showing renal IgA deposition from the three groups. (D, E) PBMCs and spleen cells were isolated from the three groups. Representative flow cytometry line graphs (D) and bar graphs (E) showing the expression of pSTAT1 and pSTAT3 in CD4+ T cells. (F) Serum levels of IL-6 and TGF-β in the three groups (the serum TGF-β level was tested in 5 rats from the HC group, 4 rats from the IgAV group, and 6 rats from the TCZ group). Representative photomicrographs showing H&E-stained sections of (G) skin and (H) intestinal (upper) and gastric (lower) tissues from the three groups. Black arrows indicate thickened vessel walls, hemangiectasis, and hyperemia; yellow arrows indicate extravasated erythrocytes. (I) Representative photomicrographs showing H&E-stained (upper) and PAS-stained (lower) sections of the kidney from the three groups. Scale bar = 50 μm. MFI: mean fluorescence intensity; ns: not significant; *p<0.05; **p<0.01; ***p<0.001; ****p<0.0001.

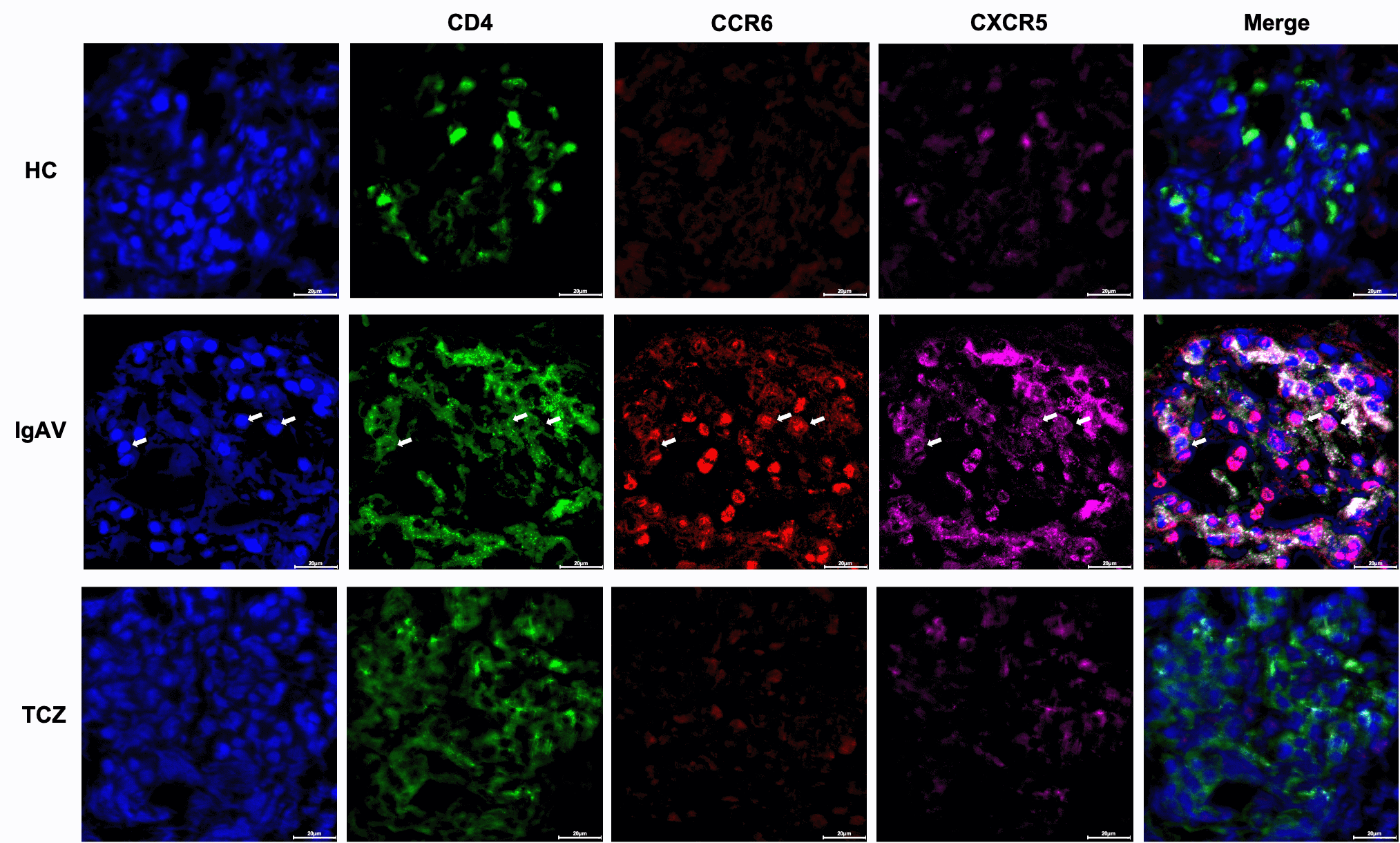
Expression of Tfh17 cells in the renal tissue of IgAV rat. Multichannel confocal
immunofluorescence staining of human renal tissue for CD4, CCR6 and CXCR5 with composite multiplexed images (scale bar: 20 lm). Arrowheads
indicate representative CD4+CCR6+ CXCR5+ Tfh17 cells.
immunofluorescence staining of human renal tissue for CD4, CCR6 and CXCR5 with composite multiplexed images (scale bar: 20 lm). Arrowheads
indicate representative CD4+CCR6+ CXCR5+ Tfh17 cells.
X. Ma: None; Q. Jiang: None; X. Chi: None.



