Poster Session B
Periodic fever syndromes, autoinflammatory diseases, Still’s disease and MAS/HLH
Session: (1124–1154) Miscellaneous Rheumatic & Inflammatory Diseases Poster II
1144: Deucravacitinib in Plaque Psoriasis: Maintenance of Response over 3 Years
Monday, November 13, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- MC
Matthew Colombo, PhD
Bristol Myers Squibb
Westampton, NJ, United StatesDisclosure information not submitted.
Abstract Poster Presenter(s)
Bruce Strober1, Howard Sofen2, Shinichi Imafuku3, Carle Paul4, Melinda Gooderham5, Lynda Spelman6, Seong Jun Seo7, Thierry Passeron8, Renata M. Kisa9, Victoria Berger9, Eleni Vritzali10, Kim Hoyt9, Matthew J. Colombo9, Subhashis Banerjee9, Matthias Augustin11, Linda Stein Gold12, Andrew Alexis13, Diamant Thaçi14, Andrew Blauvelt15 and Mark Lebwohl16, 1Yale University, New Haven, CT, 2University of California Los Angeles School of Medicine and Dermatology Research Associates, Los Angeles, CA, 3Fukuoka University Faculty of Medicine, Fukuoka, Japan, 4Toulouse University and CHU, Toulouse, France, 5SKiN Centre for Dermatology, Ontario, Canada, 6Veracity Clinical Research, Brisbane, Australia, 7Chung-Ang University Hospital, Seoul, South Korea, 8Université Côte d’Azur, University Hospital of Nice, Nice, France, 9Bristol Myers Squibb, Princeton, NJ, 10Bayer, Sissach, Switzerland, 11Institute of Health Care Research in Dermatology and Nursing, Hamburg, Germany, 12Henry Ford Health System, MI, 13Weill Cornell Medicine, New York, NY, 14Institute and Comprehensive Center for Inflammation Medicine, University of Lübeck, Lübeck, Germany, 15Oregon Medical Research Center, Portland, OR, 16Department of Dermatology, Icahn School of Medicine at Mount Sinai, New York, NY
Background/Purpose: Deucravacitinib, a first-in-class, oral, selective, allosteric tyrosine kinase 2 inhibitor, is approved in multiple countries for the treatment of adults with plaque psoriasis. Deucravacitinib was superior to placebo and apremilast in the global, 52-week, phase 3 POETYK PSO-1 (NCT03624127) and PSO-2 (NCT03611751) trials in psoriasis. Deucravacitinib is being investigated in several immune-mediated diseases and has shown efficacy compared with placebo in phase 2 trials for SLE (NCT03252587) and PsA (NCT03881059). The POETYK long-term extension (LTE) trial (NCT04036435) showed that deucravacitinib maintained long-term efficacy through 2 years with no new safety signals. Here, we report clinical efficacy up to 3 years (148 weeks) in the POETYK LTE trial in a subset of patients who received continuous deucravacitinib from day 1 in the parent trials.
Methods: In POETYK PSO-1 and PSO-2, patients were randomized 1:2:1 to placebo, deucravacitinib 6 mg once daily (QD), or apremilast 30 mg twice daily. At week 52, patients could enter the POETYK LTE trial and receive open-label deucravacitinib 6 mg QD. Deucravacitinib efficacy through week 148 was evaluated in patients from pooled POETYK PSO-1 and PSO-2 populations who received continuous deucravacitinib from day 1, achieved ≥ 75% reduction from baseline in Psoriasis Area and Severity Index (PASI 75) at week 16 (primary endpoint) or week 24 (peak response), and enrolled in the POETYK LTE trial. Maintenance of response was assessed through data cutoff (June 15, 2022) and included PASI 75 and PASI 90 (≥ 90% reduction from baseline in PASI). Static Physician's Global Assessment (sPGA) score of 0 (clear) or 1 (almost clear) with a ≥ 2-point improvement from baseline was assessed.
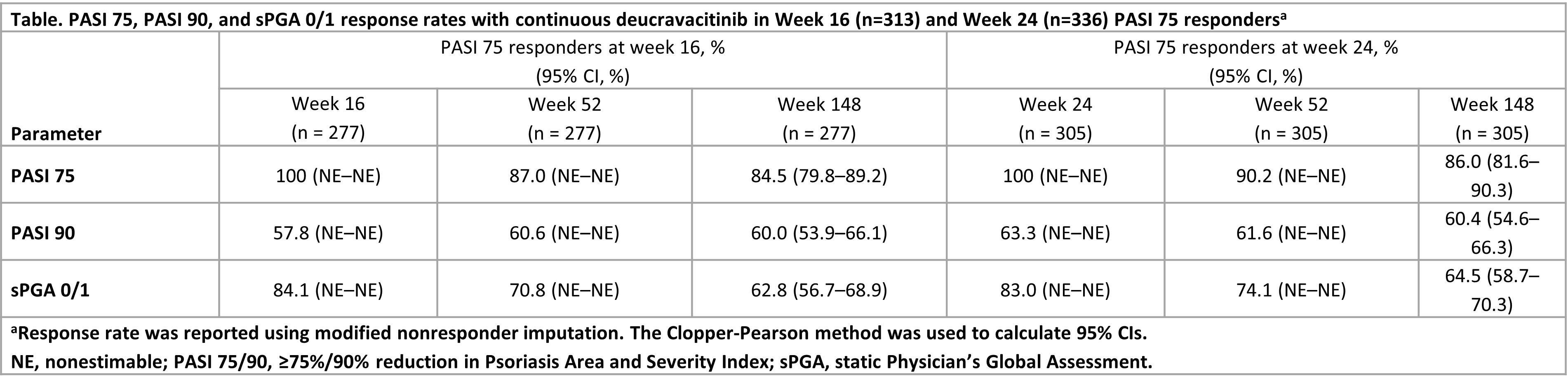
Results: A total of 513 patients completed 52 weeks in the parent trials and received continuous deucravacitinib from day 1, including 313 (61.4%) patients (95% CI, 57.0–65.6) who achieved PASI 75 at week 16 and 336 (66.5%) patients (95% CI, 62.2–70.6) who achieved PASI 75 at week 24. Among these patients, PASI 75 response rates were maintained from week 52 to week 148 (Table). PASI 90 response rates were maintained in > 50% of patients from the start of the POETYK LTE trial (Table). Response rates for sPGA 0/1 were maintained from week 52 to week 148 (Table).
Conclusion: Clinical efficacy was maintained for up to 148 weeks with continuous deucravacitinib in most patients who were week 16 and week 24 PASI 75 responders from the parent trials and enrolled in the POETYK LTE trial. These findings further support the long-term use of once-daily oral deucravacitinib as an effective treatment for patients with psoriasis.
B. Strober: AbbVie, 6, 6, Alamar, 6, Almirall, 6, Alumis, 6, Amgen, 6, Arcutis, 6, 6, Arena, 6, Aristea, 6, Asana, 6, Boehringer Ingelheim, 6, Bristol Myers Squibb, 12, Consultancy honoraria, Connect Biopharma, 6, 11, CorEvitas, 6, CorEvitas Psoriasis Registry, 12, Scientific Co-Director (consulting fee), 12, Investigator, Dermavant, 6, 6, Dermira, 12, Investigator, Eli Lilly, 6, 6, Evelo Biosciences, 6, Immunic Therapeutics, 6, Incyte, 6, Janssen, 6, 6, Journal of Psoriasis and Psoriatic Arthritis, 6, Leo, 6, Maruho, 6, Meiji Seika Pharma, 6, Mindera Health, 6, 11, Novartis, 6, Pfizer, 6, Protagonist, 6, Regeneron, 6, 6, Sanofi-Genzyme, 6, 6, Sun Pharma, 6, UCB Pharma, 6, Union Therapeutics, 6, Ventyxbio, 6, vTv Therapeutics, 6; H. Sofen: AbbVie/Abbott, 5, Amgen, 5, Boehringer-Ingelheim, 5, Bristol-Myers Squibb(BMS), 1, 2, Eli Lilly, 5, UCB, 5; S. Imafuku: AbbVie, 5, Amgen (Celgene), 12, Personal fees, Boehringer Ingelheim, 12, Personal fees, Bristol Myers Squibb, 12, Personal fees, Daiichi Sankyo, 12, Personal fees, Eisai, 5, Eli Lilly, 12, Personal fees, GSK, 12, Personal fees, Janssen, 5, Kyowa Kirin, 5, Leo Pharma, 5, Maruho, 5, Novartis, 12, Personal fees, Sun Pharma, 5, Taiho Yakuhin, 5, Tanabe Mitsubishi, 5, Torii Yakuhin, 5, UCB, 12, Personal fees; C. Paul: AbbVie, 2, 5, Almirall, 2, 5, Amgen, 2, 5, Boehringer-Ingelheim, 2, 5, Celgene, 2, 5, Eli Lilly, 2, 5, GlaxoSmithKlein(GSK), 2, 5, Janssen, 2, 5, Leo Pharma, 2, 5, Novartis, 2, 5, Pfizer, 2, 5, Pierre Fabre, 2, 5, Sanofi Regeneron, 2, 5, UCB, 2, 5; M. Gooderham: AbbVie, 1, 2, 12, Investigator, Lecture Fees, Akros Pharma, 2, 12, Investigator, Amgen, 1, 2, 12, Investigator, Lecture Fees, AnaptysBio, 12, Investigator, Arcutis, 1, 2, 12, Investigator, Lecture Fees, Aristea, 12, Investigator, Asana Biosciences, 1, Aslan, 2, 12, Investigator, Bausch Health, 1, 2, 12, Investigator, Lecture Fees, Boehringer-Ingelheim, 1, 2, 12, Investigator, Lecture Fees, Bristol-Myers Squibb(BMS), 12, Investigator, Celgene, 1, 2, 12, Investigator, Lecture Fees, Coherus Biosciences, 12, Investigator, Dermira, 12, Investigator, Eli Lilly, 1, 2, 12, Investigator, Lecture Fees, Galderma, 1, 12, Investigator, Lecture Fees, GSK, 12, Investigator, Immunotherapeutics, 12, Investigator, Incyte, 12, Investigator, Janssen, 1, 2, 12, Investigator, Lecture Fees, Kyowa Kirin, 2, 12, Investigator, Leo Pharma, 1, 12, Investigator, Lecture Fees, MedImmune, 12, Investigator, Meiji Seika, 12, Investigator, Merck, 12, Investigator, MoonLake, 12, Investigator, Nimbus Therapeutics, 12, Investigator, Novartis, 1, 2, 12, Investigator, Lecture Fees, Pfizer, 1, 12, Investigator, Lecture Fees, Regeneron, 1, 12, Investigator, Lecture Fees, Sanofi, 1, 2, 12, Investigator, Lecture Fees, Sun Pharma, 1, 2, 12, Investigator, Lecture Fees, UCB, 1, 2, 12, Investigator, Lecture Fees; L. Spelman: AbbVie, 1, 2, 6, Amgen, 2, 6, 12, Investigator, Anacor, 2, 6, 12, Investigator, Astellas, 2, 6, 12, Investigator, AstraZeneca, 2, 6, 12, Investigator, Blaze Bioscience, 2, 6, 12, Investigator, Boehringer-Ingelheim, 2, 6, 12, Investigator, Botanix, 2, 6, 12, Investigator, Bristol Myers Squibb, 2, 6, 12, Investigator, Celgene, 2, 6, 12, Investigator, Dermira, 2, 6, 12, Investigator, Eli Lilly, 2, 6, 12, Investigator, Galderma, 2, 6, 12, Investigator, Genentech, 2, 6, 12, Investigator, GSK, 2, 6, 12, Investigator, Hexima, 2, 6, 12, Investigator, Janssen, 2, 6, 12, Investigator, Leo Pharma, 2, 6, 12, Investigator, Mayne Pharma, 2, 6, 12, Investigator, MedImmune, 2, 6, 12, Investigator, Merck, 2, 6, 12, Investigator, Merck-Serono, 2, 6, 12, Investigator, Novartis, 2, 6, 12, Investigator, Otsuka, 2, 6, 12, Investigator, Pfizer, 2, 6, 12, Investigator, Phosphagenics, 2, 6, 12, Investigator, Photon MD, 2, 6, 12, Investigator, Regeneron, 2, 6, 12, Investigator, Roche, 2, 6, 12, Investigator, Samumed, 2, 6, 12, Investigator, Sanofi Genzyme, 2, 6, 12, Investigator, SHR Pharmacy, 2, 6, 12, Investigator, Sun Pharma ANZ, 2, 6, 12, Investigator, Trius, 2, 6, 12, Investigator, UCB, 2, 6, 12, Investigator, Zai Lab, 2, 6, 12, Investigator; S. Seo: None; T. Passeron: AbbVie/Abbott, 2, 6, Amgen, 6, Boehringer-Ingelheim, 6, Bristol-Myers Squibb(BMS), 1, 5, 6, Celgene, 6, Eli Lilly, 1, 6, Janssen, 1, 6, Novartis, 1, 6, Pfizer, 6, UCB, 1, 5, 6; R. Kisa: Bristol-Myers Squibb(BMS), 3, 12, Shareholder; V. Berger: Bristol-Myers Squibb(BMS), 3, 11; E. Vritzali: Bristol-Myers Squibb(BMS), 3, 12, Shareholder; K. Hoyt: Bristol-Myers Squibb(BMS), 2; M. Colombo: Bristol-Myers Squibb(BMS), 3, 11; S. Banerjee: Bristol-Myers Squibb(BMS), 3, 11; M. Augustin: AbbVie, 1, 2, 5, 6, 12, Investigator, Almirall, 1, 2, 4, 5, 6, Amgen, 1, 2, 5, 6, 12, Investigator, Biogen, 2, 4, 5, 6, 12, Investigator, Boehringer-Ingelheim, 1, 5, 6, 12, Investigator, Bristol-Myers Squibb(BMS), 1, 2, 4, 5, 6, Celgene, 5, 12, Investigator, Eli Lilly, 2, 4, 6, 12, Investigator, Galderma, 2, 4, 6, 12, Investigator, GlaxoSmithKlein(GSK), 2, 4, 6, Janssen Biotech, 1, 2, 5, 6, 12, Investigator, Leo Pharma, 1, 2, 5, 6, 12, Investigator, Merck, 5, 12, Investigator, Mylan, 2, 4, 6, Novartis, 1, 2, 6, 12, Investigator, Pfizer, 2, 6, 12, Investigator, Sanofi, 1, 2, 6, 12, Investigator, UCB, 2, 6, 12, Investigator; L. Stein Gold: AbbVie/Abbott, 1, 2, 6, Amgen, 1, 2, 5, 6, Bristol-Myers Squibb(BMS), 1, 2, 6, Eli Lilly, 1, 2, 5, Janssen, 1, 2, 5, Novartis, 2, Pfizer, 1, 2, 5, 6, UCB, 1, 2, 5, 6; A. Alexis: AbbVie, 1, 2, 5, Allergan, 1, 2, Almirall, 1, 2, 5, Amgen, 1, 2, 5, Arcutis, 1, 2, 5, Bausch Health, 1, 2, Beiersdorf, 1, 2, Bristol Myers Squibb, 1, 2, 5, 6, Cara Therapeutics, 1, 2, 5, Castle Biosciences, 1, 2, 5, Cutera, 1, 2, Dermavant, 1, 2, 5, Eli Lilly, 1, 2, EPI Health, 1, 2, Galderma, 1, 2, 5, Incyte, 1, 2, Janssen, 1, 2, L’Oréal, 1, 2, Leo Pharma, 1, 2, 5, Novartis, 5, Ortho, 1, 2, Pfizer, 1, 2, 6, Regeneron, 6, Sanofi-Genzyme, 6, Sanofi-Regeneron, 1, 2, Springer, 9, Swiss American, 1, 2, UCB, 1, 2, Valeant (Bausch Health), 5, VisualDx, 1, 2, Vyne, 1, 2, 5, Wiley-Blackwell, 9, Wolters Kluwer Health, 9; D. Thaçi: AbbVie, 1, 2, 5, 12, Investigator, Almirall, 1, 2, 12, Investigator, Amgen, 1, 2, 12, Investigator, Boehringer-Ingelheim, 1, 2, 12, Investigator, Bristol-Myers Squibb(BMS), 1, 2, 12, Investigator, Celltrion, 1, 2, 12, Investigator, Eli Lilly, 1, 2, 12, Investigator, Galapagos, 1, 2, 12, Investigator, Galderma, 1, 2, 5, 12, Investigator, Janssen-Cilag, 1, 2, 12, Investigator, LEO Pharma, 1, 2, 5, 12, Investigator, Novartis, 1, 2, 5, 12, Investigator, Pfizer, 1, 2, 12, Investigator, Regeneron, 1, 2, 12, Investigator, Samsung, 1, 2, 12, Investigator, Sandoz, 1, 2, 12, Investigator, Sanofi, 1, 2, 12, Investigator, Target-Solution, 1, 2, 12, Investigator, UCB, 1, 2, 12, Investigator; A. Blauvelt: AbbVie/Abbott, 5, 6, Abcentra, 6, Acelyrin, 12, Clinical study investigator, Aclaris, 6, Affibody, 6, Aligos, 6, Allakos, 12, Clinical study investigator, Almirall, 6, Alumis, 6, Amgen, 5, 6, Anaptysbio, 6, Apogee, 6, Arcutis, 5, 6, Arena, 6, Aslan, 6, Athenex, 5, 6, 12, Clinical study investigator, Bluefin, 6, Boehringer-Ingelheim, 5, 6, Bristol-Myers Squibb(BMS), 5, 6, Cara Therapeutics, 6, Concert, 12, Clinical study investigator, CTI Biopharma, 6, Dermavant, 5, 6, EcoR1, 6, Eli Lilly, 5, 6, Escient, 6, Evelo, 6, Evommune, 6, Forte, 6, Galderma, 5, 6, Highlightll Pharma, 6, Incyte, 5, 6, InnoventBio, 6, Janssen, 5, 6, Landos, 6, Leo, 5, 6, Merck/MSD, 5, 6, Novartis, 5, 6, Pfizer, 5, 6, Rani, 6, Rapt, 6, Regeneron, 5, 6, Sanofi Genzyme, 6, Spherix Global Insights, 6, Sun Pharma, 5, 6, TLL Pharmaceutical, 6, TrialSpark, 6, UCB, 5, 5, 6, 6, Union, 6, Ventyx, 6, Vibliome, 6, Xencor, 6; M. Lebwohl: Mark Lebwohl is an employee of Mount Sinai and receives research funds fromAbbvie, Amgen, Arcutis, Avotres, Boehringer Ingelheim, Cara therapeutics,, 2.
Background/Purpose: Deucravacitinib, a first-in-class, oral, selective, allosteric tyrosine kinase 2 inhibitor, is approved in multiple countries for the treatment of adults with plaque psoriasis. Deucravacitinib was superior to placebo and apremilast in the global, 52-week, phase 3 POETYK PSO-1 (NCT03624127) and PSO-2 (NCT03611751) trials in psoriasis. Deucravacitinib is being investigated in several immune-mediated diseases and has shown efficacy compared with placebo in phase 2 trials for SLE (NCT03252587) and PsA (NCT03881059). The POETYK long-term extension (LTE) trial (NCT04036435) showed that deucravacitinib maintained long-term efficacy through 2 years with no new safety signals. Here, we report clinical efficacy up to 3 years (148 weeks) in the POETYK LTE trial in a subset of patients who received continuous deucravacitinib from day 1 in the parent trials.
Methods: In POETYK PSO-1 and PSO-2, patients were randomized 1:2:1 to placebo, deucravacitinib 6 mg once daily (QD), or apremilast 30 mg twice daily. At week 52, patients could enter the POETYK LTE trial and receive open-label deucravacitinib 6 mg QD. Deucravacitinib efficacy through week 148 was evaluated in patients from pooled POETYK PSO-1 and PSO-2 populations who received continuous deucravacitinib from day 1, achieved ≥ 75% reduction from baseline in Psoriasis Area and Severity Index (PASI 75) at week 16 (primary endpoint) or week 24 (peak response), and enrolled in the POETYK LTE trial. Maintenance of response was assessed through data cutoff (June 15, 2022) and included PASI 75 and PASI 90 (≥ 90% reduction from baseline in PASI). Static Physician's Global Assessment (sPGA) score of 0 (clear) or 1 (almost clear) with a ≥ 2-point improvement from baseline was assessed.
Results: A total of 513 patients completed 52 weeks in the parent trials and received continuous deucravacitinib from day 1, including 313 (61.4%) patients (95% CI, 57.0–65.6) who achieved PASI 75 at week 16 and 336 (66.5%) patients (95% CI, 62.2–70.6) who achieved PASI 75 at week 24. Among these patients, PASI 75 response rates were maintained from week 52 to week 148 (Table). PASI 90 response rates were maintained in > 50% of patients from the start of the POETYK LTE trial (Table). Response rates for sPGA 0/1 were maintained from week 52 to week 148 (Table).
Conclusion: Clinical efficacy was maintained for up to 148 weeks with continuous deucravacitinib in most patients who were week 16 and week 24 PASI 75 responders from the parent trials and enrolled in the POETYK LTE trial. These findings further support the long-term use of once-daily oral deucravacitinib as an effective treatment for patients with psoriasis.

B. Strober: AbbVie, 6, 6, Alamar, 6, Almirall, 6, Alumis, 6, Amgen, 6, Arcutis, 6, 6, Arena, 6, Aristea, 6, Asana, 6, Boehringer Ingelheim, 6, Bristol Myers Squibb, 12, Consultancy honoraria, Connect Biopharma, 6, 11, CorEvitas, 6, CorEvitas Psoriasis Registry, 12, Scientific Co-Director (consulting fee), 12, Investigator, Dermavant, 6, 6, Dermira, 12, Investigator, Eli Lilly, 6, 6, Evelo Biosciences, 6, Immunic Therapeutics, 6, Incyte, 6, Janssen, 6, 6, Journal of Psoriasis and Psoriatic Arthritis, 6, Leo, 6, Maruho, 6, Meiji Seika Pharma, 6, Mindera Health, 6, 11, Novartis, 6, Pfizer, 6, Protagonist, 6, Regeneron, 6, 6, Sanofi-Genzyme, 6, 6, Sun Pharma, 6, UCB Pharma, 6, Union Therapeutics, 6, Ventyxbio, 6, vTv Therapeutics, 6; H. Sofen: AbbVie/Abbott, 5, Amgen, 5, Boehringer-Ingelheim, 5, Bristol-Myers Squibb(BMS), 1, 2, Eli Lilly, 5, UCB, 5; S. Imafuku: AbbVie, 5, Amgen (Celgene), 12, Personal fees, Boehringer Ingelheim, 12, Personal fees, Bristol Myers Squibb, 12, Personal fees, Daiichi Sankyo, 12, Personal fees, Eisai, 5, Eli Lilly, 12, Personal fees, GSK, 12, Personal fees, Janssen, 5, Kyowa Kirin, 5, Leo Pharma, 5, Maruho, 5, Novartis, 12, Personal fees, Sun Pharma, 5, Taiho Yakuhin, 5, Tanabe Mitsubishi, 5, Torii Yakuhin, 5, UCB, 12, Personal fees; C. Paul: AbbVie, 2, 5, Almirall, 2, 5, Amgen, 2, 5, Boehringer-Ingelheim, 2, 5, Celgene, 2, 5, Eli Lilly, 2, 5, GlaxoSmithKlein(GSK), 2, 5, Janssen, 2, 5, Leo Pharma, 2, 5, Novartis, 2, 5, Pfizer, 2, 5, Pierre Fabre, 2, 5, Sanofi Regeneron, 2, 5, UCB, 2, 5; M. Gooderham: AbbVie, 1, 2, 12, Investigator, Lecture Fees, Akros Pharma, 2, 12, Investigator, Amgen, 1, 2, 12, Investigator, Lecture Fees, AnaptysBio, 12, Investigator, Arcutis, 1, 2, 12, Investigator, Lecture Fees, Aristea, 12, Investigator, Asana Biosciences, 1, Aslan, 2, 12, Investigator, Bausch Health, 1, 2, 12, Investigator, Lecture Fees, Boehringer-Ingelheim, 1, 2, 12, Investigator, Lecture Fees, Bristol-Myers Squibb(BMS), 12, Investigator, Celgene, 1, 2, 12, Investigator, Lecture Fees, Coherus Biosciences, 12, Investigator, Dermira, 12, Investigator, Eli Lilly, 1, 2, 12, Investigator, Lecture Fees, Galderma, 1, 12, Investigator, Lecture Fees, GSK, 12, Investigator, Immunotherapeutics, 12, Investigator, Incyte, 12, Investigator, Janssen, 1, 2, 12, Investigator, Lecture Fees, Kyowa Kirin, 2, 12, Investigator, Leo Pharma, 1, 12, Investigator, Lecture Fees, MedImmune, 12, Investigator, Meiji Seika, 12, Investigator, Merck, 12, Investigator, MoonLake, 12, Investigator, Nimbus Therapeutics, 12, Investigator, Novartis, 1, 2, 12, Investigator, Lecture Fees, Pfizer, 1, 12, Investigator, Lecture Fees, Regeneron, 1, 12, Investigator, Lecture Fees, Sanofi, 1, 2, 12, Investigator, Lecture Fees, Sun Pharma, 1, 2, 12, Investigator, Lecture Fees, UCB, 1, 2, 12, Investigator, Lecture Fees; L. Spelman: AbbVie, 1, 2, 6, Amgen, 2, 6, 12, Investigator, Anacor, 2, 6, 12, Investigator, Astellas, 2, 6, 12, Investigator, AstraZeneca, 2, 6, 12, Investigator, Blaze Bioscience, 2, 6, 12, Investigator, Boehringer-Ingelheim, 2, 6, 12, Investigator, Botanix, 2, 6, 12, Investigator, Bristol Myers Squibb, 2, 6, 12, Investigator, Celgene, 2, 6, 12, Investigator, Dermira, 2, 6, 12, Investigator, Eli Lilly, 2, 6, 12, Investigator, Galderma, 2, 6, 12, Investigator, Genentech, 2, 6, 12, Investigator, GSK, 2, 6, 12, Investigator, Hexima, 2, 6, 12, Investigator, Janssen, 2, 6, 12, Investigator, Leo Pharma, 2, 6, 12, Investigator, Mayne Pharma, 2, 6, 12, Investigator, MedImmune, 2, 6, 12, Investigator, Merck, 2, 6, 12, Investigator, Merck-Serono, 2, 6, 12, Investigator, Novartis, 2, 6, 12, Investigator, Otsuka, 2, 6, 12, Investigator, Pfizer, 2, 6, 12, Investigator, Phosphagenics, 2, 6, 12, Investigator, Photon MD, 2, 6, 12, Investigator, Regeneron, 2, 6, 12, Investigator, Roche, 2, 6, 12, Investigator, Samumed, 2, 6, 12, Investigator, Sanofi Genzyme, 2, 6, 12, Investigator, SHR Pharmacy, 2, 6, 12, Investigator, Sun Pharma ANZ, 2, 6, 12, Investigator, Trius, 2, 6, 12, Investigator, UCB, 2, 6, 12, Investigator, Zai Lab, 2, 6, 12, Investigator; S. Seo: None; T. Passeron: AbbVie/Abbott, 2, 6, Amgen, 6, Boehringer-Ingelheim, 6, Bristol-Myers Squibb(BMS), 1, 5, 6, Celgene, 6, Eli Lilly, 1, 6, Janssen, 1, 6, Novartis, 1, 6, Pfizer, 6, UCB, 1, 5, 6; R. Kisa: Bristol-Myers Squibb(BMS), 3, 12, Shareholder; V. Berger: Bristol-Myers Squibb(BMS), 3, 11; E. Vritzali: Bristol-Myers Squibb(BMS), 3, 12, Shareholder; K. Hoyt: Bristol-Myers Squibb(BMS), 2; M. Colombo: Bristol-Myers Squibb(BMS), 3, 11; S. Banerjee: Bristol-Myers Squibb(BMS), 3, 11; M. Augustin: AbbVie, 1, 2, 5, 6, 12, Investigator, Almirall, 1, 2, 4, 5, 6, Amgen, 1, 2, 5, 6, 12, Investigator, Biogen, 2, 4, 5, 6, 12, Investigator, Boehringer-Ingelheim, 1, 5, 6, 12, Investigator, Bristol-Myers Squibb(BMS), 1, 2, 4, 5, 6, Celgene, 5, 12, Investigator, Eli Lilly, 2, 4, 6, 12, Investigator, Galderma, 2, 4, 6, 12, Investigator, GlaxoSmithKlein(GSK), 2, 4, 6, Janssen Biotech, 1, 2, 5, 6, 12, Investigator, Leo Pharma, 1, 2, 5, 6, 12, Investigator, Merck, 5, 12, Investigator, Mylan, 2, 4, 6, Novartis, 1, 2, 6, 12, Investigator, Pfizer, 2, 6, 12, Investigator, Sanofi, 1, 2, 6, 12, Investigator, UCB, 2, 6, 12, Investigator; L. Stein Gold: AbbVie/Abbott, 1, 2, 6, Amgen, 1, 2, 5, 6, Bristol-Myers Squibb(BMS), 1, 2, 6, Eli Lilly, 1, 2, 5, Janssen, 1, 2, 5, Novartis, 2, Pfizer, 1, 2, 5, 6, UCB, 1, 2, 5, 6; A. Alexis: AbbVie, 1, 2, 5, Allergan, 1, 2, Almirall, 1, 2, 5, Amgen, 1, 2, 5, Arcutis, 1, 2, 5, Bausch Health, 1, 2, Beiersdorf, 1, 2, Bristol Myers Squibb, 1, 2, 5, 6, Cara Therapeutics, 1, 2, 5, Castle Biosciences, 1, 2, 5, Cutera, 1, 2, Dermavant, 1, 2, 5, Eli Lilly, 1, 2, EPI Health, 1, 2, Galderma, 1, 2, 5, Incyte, 1, 2, Janssen, 1, 2, L’Oréal, 1, 2, Leo Pharma, 1, 2, 5, Novartis, 5, Ortho, 1, 2, Pfizer, 1, 2, 6, Regeneron, 6, Sanofi-Genzyme, 6, Sanofi-Regeneron, 1, 2, Springer, 9, Swiss American, 1, 2, UCB, 1, 2, Valeant (Bausch Health), 5, VisualDx, 1, 2, Vyne, 1, 2, 5, Wiley-Blackwell, 9, Wolters Kluwer Health, 9; D. Thaçi: AbbVie, 1, 2, 5, 12, Investigator, Almirall, 1, 2, 12, Investigator, Amgen, 1, 2, 12, Investigator, Boehringer-Ingelheim, 1, 2, 12, Investigator, Bristol-Myers Squibb(BMS), 1, 2, 12, Investigator, Celltrion, 1, 2, 12, Investigator, Eli Lilly, 1, 2, 12, Investigator, Galapagos, 1, 2, 12, Investigator, Galderma, 1, 2, 5, 12, Investigator, Janssen-Cilag, 1, 2, 12, Investigator, LEO Pharma, 1, 2, 5, 12, Investigator, Novartis, 1, 2, 5, 12, Investigator, Pfizer, 1, 2, 12, Investigator, Regeneron, 1, 2, 12, Investigator, Samsung, 1, 2, 12, Investigator, Sandoz, 1, 2, 12, Investigator, Sanofi, 1, 2, 12, Investigator, Target-Solution, 1, 2, 12, Investigator, UCB, 1, 2, 12, Investigator; A. Blauvelt: AbbVie/Abbott, 5, 6, Abcentra, 6, Acelyrin, 12, Clinical study investigator, Aclaris, 6, Affibody, 6, Aligos, 6, Allakos, 12, Clinical study investigator, Almirall, 6, Alumis, 6, Amgen, 5, 6, Anaptysbio, 6, Apogee, 6, Arcutis, 5, 6, Arena, 6, Aslan, 6, Athenex, 5, 6, 12, Clinical study investigator, Bluefin, 6, Boehringer-Ingelheim, 5, 6, Bristol-Myers Squibb(BMS), 5, 6, Cara Therapeutics, 6, Concert, 12, Clinical study investigator, CTI Biopharma, 6, Dermavant, 5, 6, EcoR1, 6, Eli Lilly, 5, 6, Escient, 6, Evelo, 6, Evommune, 6, Forte, 6, Galderma, 5, 6, Highlightll Pharma, 6, Incyte, 5, 6, InnoventBio, 6, Janssen, 5, 6, Landos, 6, Leo, 5, 6, Merck/MSD, 5, 6, Novartis, 5, 6, Pfizer, 5, 6, Rani, 6, Rapt, 6, Regeneron, 5, 6, Sanofi Genzyme, 6, Spherix Global Insights, 6, Sun Pharma, 5, 6, TLL Pharmaceutical, 6, TrialSpark, 6, UCB, 5, 5, 6, 6, Union, 6, Ventyx, 6, Vibliome, 6, Xencor, 6; M. Lebwohl: Mark Lebwohl is an employee of Mount Sinai and receives research funds fromAbbvie, Amgen, Arcutis, Avotres, Boehringer Ingelheim, Cara therapeutics,, 2.



