Poster Session B
Fibrosing rheumatic diseases (scleroderma, MCTD, IgG4-related disease, scleroderma mimics)
Session: (0934–0964) Systemic Sclerosis & Related Disorders – Basic Science Poster
0949: Development of Interstitial Pneumonia with Autoimmune Features in Conditional Tgfb3 Deletion
Monday, November 13, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- TK
Abstract Poster Presenter(s)
Toshihiko Komai1, Tomohisa Okamura2, Masanori Kono1, Kazuhiko Yamamoto3 and Keishi Fujio1, 1Department of Allergy and Rheumatology, Graduate School of Medicine, The University of Tokyo, Tokyo, Japan, 2Department of Allergy and Rheumatology, Graduate School of Medicine, The University of Tokyo and Department of Functional Genomics and Immunological Diseases, Graduate School of Medicine, The University of Tokyo, Tokyo, Japan, 3Department of Allergy and Rheumatology, Graduate School of Medicine, The University of Tokyo and Laboratory for Autoimmune Diseases, Center for Integrative Medical Sciences, RIKEN, Yokohama, Japan
Background/Purpose: We found that TGF-β3 regulates humoral immunity in a context-dependent manner [1]. Recent genome-wide association study reveals that TGF-β3 might be a novel target in African American systemic sclerosis (SSc) patients [2]. Since Tgfb3-/- mice die within 20 hours of birth [3], the physiological roles of TGF-β3 have not clearly elucidated. In this study, we evaluated the immunological and fibrotic roles of TGF-β3 by using mice with conditional Tgfb3 deletion.
[1] Komai T, et al. Front Immunol. 2018;9:1364., [2] Kaundal U, et al. ACR2022, Arthritis Rheumatol. 2022; 74 (suppl 9), [3] Kaartinen V, et al. Nat Genet. 1995;11(4):415-21.
Methods: TGF-β3 conditional knockout mice in CD4+ T cells (Tgfb3fl/flCD4Cre) and B cells (Tgfb3fl/flCD19Cre) were created. Histopathology, expression of fibrosis-related genes in lung by qRT-PCR in lungs, and flow cytometric analysis of splenocytes from these mice were conducted. Also, a continuous bleomycin infusion delivered by osmotic minipumps to these mice was conducted as an induced SSc model. To evaluate the effects of overexpression of TGF-β3, either pCAGGS-Mock, pCAGGS-Tgfb3 plasmid vectors were intravenously administered to lupus prone MRL/lpr mice with spontaneous interstitial pneumonia, and lung pathologies were assessed.In vitro, normal human lung fibroblasts (NHLFs) cultured with and without TGF-β3 were evaluated.
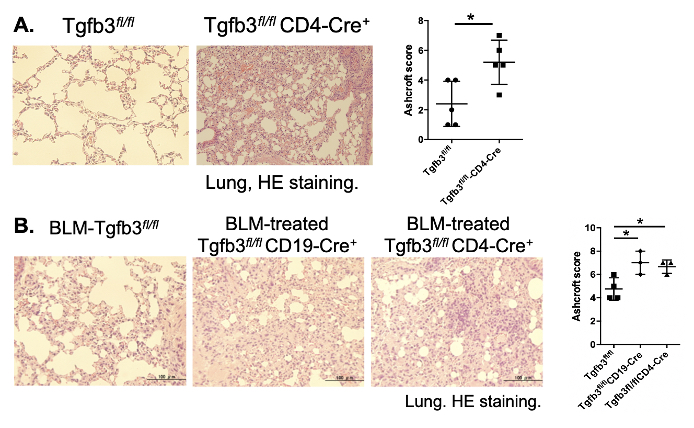
Results: Tgfb3fl/flCD4Cre mice developed inflammatory cell infiltration and fibrosis in lungs spontaneously (Figure 1A), and low-titer anti-dsDNA antibody production and increased marginal zone B cells was observed. The bleomycin-induction exacerbated interstitial pneumonia, and fibrosis-related genes of the lung tissue such as mCol1a1, mCol1a2, mSerpine1, and mSpp1 were up-regulated in both Tgfb3fl/flCD4Cre and Tgfb3fl/flCD19Cre mice in comparison to Tgfb3fl/flmice (Figure 1B). Further, the interstitial pneumonia of MRL/lpr mice was prone to be ameliorated by pCAGGS-Tgfb3 administration. In in vitro experiments, TGF-β3 inhibited the proliferation of NHLFs in a dose-dependent manner.
Conclusion: Our study suggested that TGF-β3 contributes to regulate systemic autoimmune and fibrotic responses. Anti-inflammatory and anti-fibrotic function of TGF-β3 might lead to future therapeutic target for interstitial pneumonia with autoimmune features.
T. Komai: Amgen, 6, Asahi Kasei, 6, Chugai, 5, 6, Daiichi-Sankyo, 6, Eisai, 6, Eli Lilly, 1, 6, GlaxoSmithKlein(GSK), 5, 6, Janssen, 6, Novartis, 6, Tanabe Mitsubishi, 6; T. Okamura: Chugai Pharmaceutical., 12, belong to the Social Cooperation Program, Department of Functional Genomics and Immunological Diseases, supported by Chugai Pharmaceutical.; M. Kono: Asahi Kasei Pharma, 6, Eli Lilly, 6, GlaxoSmithKlein(GSK), 6; K. Yamamoto: AbbVie, 6, Pfizer Japan Inc, 12, Outsourcing contract, RegCell, 1, Sun Pharmaceutical Industries Ltd, 6; K. Fujio: AbbVie/Abbott, 6, Asahi Kasei, 5, 6, Astellas, 6, AstraZeneca, 6, Ayumi, 6, Bristol-Myers Squibb(BMS), 5, 6, Chugai Pharmaceutical., 5, 6, Daiichi-Sankyo, 6, Eisai, 5, 6, Eli Lilly, 5, 6, Janssen, 6, Novartis, 6, Ono, 6, Pfizer, 6, Sanofi, 6, Tanabe Mitsubishi, 5, 6, Tsumura, 5.
Background/Purpose: We found that TGF-β3 regulates humoral immunity in a context-dependent manner [1]. Recent genome-wide association study reveals that TGF-β3 might be a novel target in African American systemic sclerosis (SSc) patients [2]. Since Tgfb3-/- mice die within 20 hours of birth [3], the physiological roles of TGF-β3 have not clearly elucidated. In this study, we evaluated the immunological and fibrotic roles of TGF-β3 by using mice with conditional Tgfb3 deletion.
[1] Komai T, et al. Front Immunol. 2018;9:1364., [2] Kaundal U, et al. ACR2022, Arthritis Rheumatol. 2022; 74 (suppl 9), [3] Kaartinen V, et al. Nat Genet. 1995;11(4):415-21.
Methods: TGF-β3 conditional knockout mice in CD4+ T cells (Tgfb3fl/flCD4Cre) and B cells (Tgfb3fl/flCD19Cre) were created. Histopathology, expression of fibrosis-related genes in lung by qRT-PCR in lungs, and flow cytometric analysis of splenocytes from these mice were conducted. Also, a continuous bleomycin infusion delivered by osmotic minipumps to these mice was conducted as an induced SSc model. To evaluate the effects of overexpression of TGF-β3, either pCAGGS-Mock, pCAGGS-Tgfb3 plasmid vectors were intravenously administered to lupus prone MRL/lpr mice with spontaneous interstitial pneumonia, and lung pathologies were assessed.In vitro, normal human lung fibroblasts (NHLFs) cultured with and without TGF-β3 were evaluated.
Results: Tgfb3fl/flCD4Cre mice developed inflammatory cell infiltration and fibrosis in lungs spontaneously (Figure 1A), and low-titer anti-dsDNA antibody production and increased marginal zone B cells was observed. The bleomycin-induction exacerbated interstitial pneumonia, and fibrosis-related genes of the lung tissue such as mCol1a1, mCol1a2, mSerpine1, and mSpp1 were up-regulated in both Tgfb3fl/flCD4Cre and Tgfb3fl/flCD19Cre mice in comparison to Tgfb3fl/flmice (Figure 1B). Further, the interstitial pneumonia of MRL/lpr mice was prone to be ameliorated by pCAGGS-Tgfb3 administration. In in vitro experiments, TGF-β3 inhibited the proliferation of NHLFs in a dose-dependent manner.
Conclusion: Our study suggested that TGF-β3 contributes to regulate systemic autoimmune and fibrotic responses. Anti-inflammatory and anti-fibrotic function of TGF-β3 might lead to future therapeutic target for interstitial pneumonia with autoimmune features.

Figure 1. Exacerbation of interstitial pneumonia by conditional Tgfb3 deletion.
T. Komai: Amgen, 6, Asahi Kasei, 6, Chugai, 5, 6, Daiichi-Sankyo, 6, Eisai, 6, Eli Lilly, 1, 6, GlaxoSmithKlein(GSK), 5, 6, Janssen, 6, Novartis, 6, Tanabe Mitsubishi, 6; T. Okamura: Chugai Pharmaceutical., 12, belong to the Social Cooperation Program, Department of Functional Genomics and Immunological Diseases, supported by Chugai Pharmaceutical.; M. Kono: Asahi Kasei Pharma, 6, Eli Lilly, 6, GlaxoSmithKlein(GSK), 6; K. Yamamoto: AbbVie, 6, Pfizer Japan Inc, 12, Outsourcing contract, RegCell, 1, Sun Pharmaceutical Industries Ltd, 6; K. Fujio: AbbVie/Abbott, 6, Asahi Kasei, 5, 6, Astellas, 6, AstraZeneca, 6, Ayumi, 6, Bristol-Myers Squibb(BMS), 5, 6, Chugai Pharmaceutical., 5, 6, Daiichi-Sankyo, 6, Eisai, 5, 6, Eli Lilly, 5, 6, Janssen, 6, Novartis, 6, Ono, 6, Pfizer, 6, Sanofi, 6, Tanabe Mitsubishi, 5, 6, Tsumura, 5.



