Poster Session B
Epidemiology, health policy and outcomes
Session: (1200–1220) Patient Outcomes, Preferences, & Attitudes Poster II
1215: Efficacy of Pharmacological Interventions: A Systematic Review Informing the 2023 EULAR Recommendations for the Management of Fatigue in People with Inflammatory Rheumatic and Musculoskeletal Diseases
Monday, November 13, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- BF
Bayram Farisogullari, MD
Hacettepe University, Faculty of Medicine, Department of Internal Medicine, Division of Rheumatology
Ankara, TurkeyDisclosure information not submitted.
Abstract Poster Presenter(s)
Bayram Farisogullari1, Eduardo Santos2, Emma Dures3, Rinie Geenen4 and Pedro Machado5, 1Hacettepe University, Faculty of Medicine, Department of Internal Medicine, Division of Rheumatology, Ankara, Turkey, 2Health Sciences Research Unit: Nursing (UICISA: E), Nursing School of Coimbra (ESEnfC), Coimbra, Portugal, 3Academic Rheumatology, Bristol Royal Infirmary; and Faculty of Health and Applied Sciences, University of the West of England, Bristol, United Kingdom, 4Utrecht University, Vorstenbosch, Netherlands, 5Centre for Rheumatology & Department of Neuromuscular Diseases, University College London, London, UK. Department of Rheumatology, Northwick Park Hospital, London North West University Healthcare NHS trust, London, UK., London, United Kingdom
Background/Purpose: To identify the best evidence on the efficacy of pharmacological interventions in reducing fatigue in people with I-RMDs and to summarise their safety in the identified studies to inform EULAR recommendations for the management of fatigue in people with inflammatory rheumatic and musculoskeletal disease (I-RMD).
Methods: Systematic review of adults with I-RMD conducted according to the Cochrane Handbook. Search strategy ran in Medline, Embase, Cochrane Library, CINAHL Complete, PEDro, OTseeker and PsycINFO. Assessment of risk of bias, data extraction, and synthesis performed by two reviewers independently. Data pooled in statistical meta-analyses.
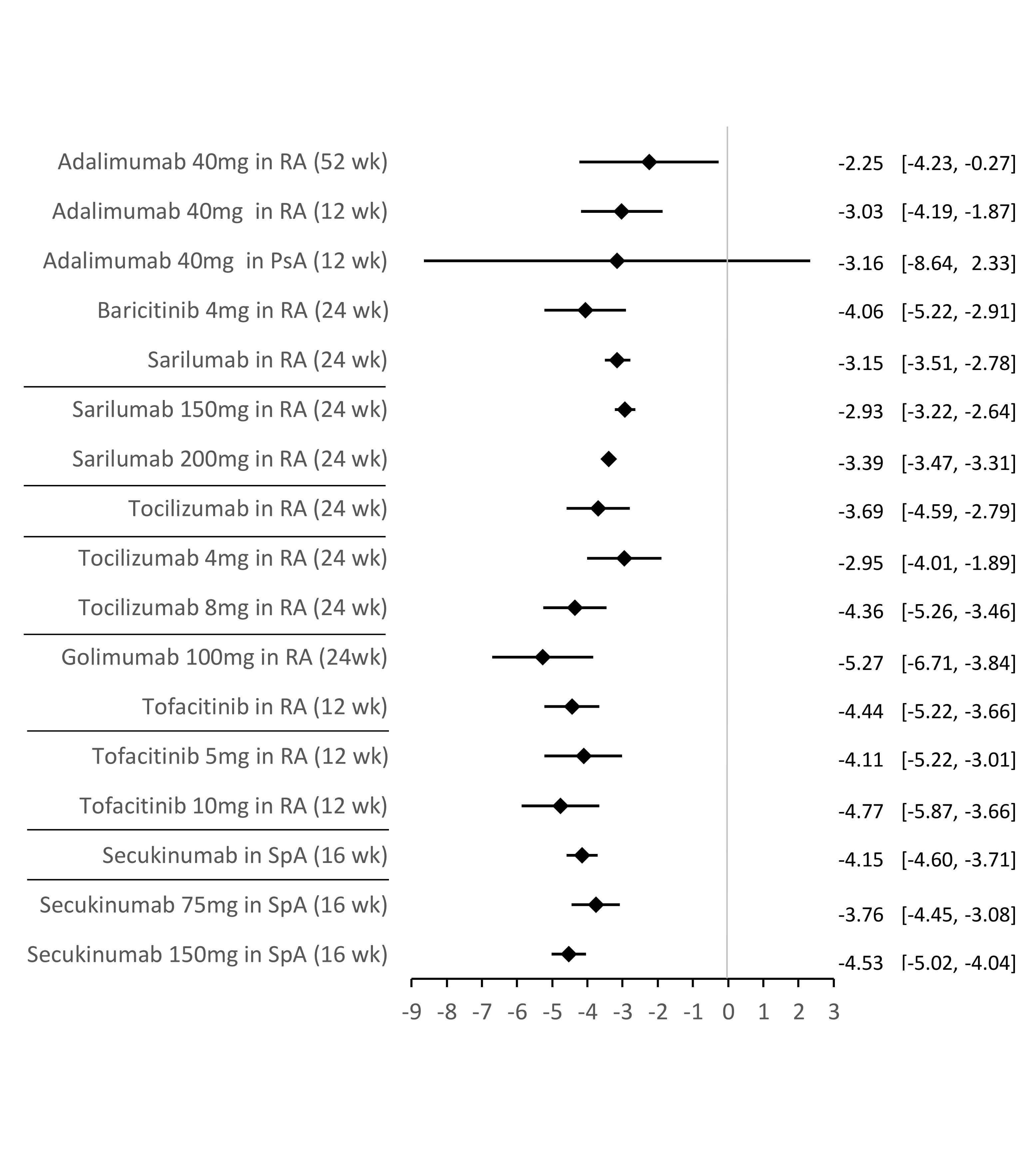
Results: From a total of 4,150 records, 454 were selected for full-text review, 105 fulfilled the inclusion criteria, and 19 RCTs were included in meta-analyses. Adalimumab was superior to placebo in reducing fatigue at 52 and 12 weeks (wk) in rheumatoid arthritis (RA) (mean difference [MD]=-2.25, p=0.03; MD=-3.03, p< 0.001; respectively) and psoriatic arthritis (MD=-3.16, p=0.26). Golimumab (24wk: MD=-5.27, p< 0.001), baricitinib (24wk: MD=-4.06, p< 0.001), sarilumab (24wk: MD=-3.15, p< 0.001), tocilizumab (24wk: MD=-3.69, p< 0.001) and tofacitinib (12wk: MD=-4.44, p< 0.001) were also superior to placebo in reducing fatigue in RA. A dose/effect relationship was observed for sarilumab, tocilizumab and tofacitinib. In spondyloarthritis, secukinumab was superior to placebo in reducing fatigue at 16wk (MD=-4.15, p< 0.001), with a dose/effect relationship also observed (Figure). The narrative results of the RCTs not included in the meta-analysis indicated that several other pharmacological interventions were efficacious in reducing fatigue, with reassuring safety results.
Conclusion: Pharmacological interventions are efficacious and safe for the management of fatigue in people with I-RMD.
B. Farisogullari: None; E. Santos: None; E. Dures: None; R. Geenen: None; P. Machado: AbbVie/Abbott, 2, 6, Bristol-Myers Squibb(BMS), 2, 6, Celgene, 2, 6, Eli Lilly, 2, 6, Galapagos, 2, 6, Janssen, 2, 6, Merck/MSD, 2, 6, Novartis, 2, 6, Orphazyme, 2, 6, Pfizer, 2, 6, Roche, 2, 6, UCB, 2, 6.
Background/Purpose: To identify the best evidence on the efficacy of pharmacological interventions in reducing fatigue in people with I-RMDs and to summarise their safety in the identified studies to inform EULAR recommendations for the management of fatigue in people with inflammatory rheumatic and musculoskeletal disease (I-RMD).
Methods: Systematic review of adults with I-RMD conducted according to the Cochrane Handbook. Search strategy ran in Medline, Embase, Cochrane Library, CINAHL Complete, PEDro, OTseeker and PsycINFO. Assessment of risk of bias, data extraction, and synthesis performed by two reviewers independently. Data pooled in statistical meta-analyses.
Results: From a total of 4,150 records, 454 were selected for full-text review, 105 fulfilled the inclusion criteria, and 19 RCTs were included in meta-analyses. Adalimumab was superior to placebo in reducing fatigue at 52 and 12 weeks (wk) in rheumatoid arthritis (RA) (mean difference [MD]=-2.25, p=0.03; MD=-3.03, p< 0.001; respectively) and psoriatic arthritis (MD=-3.16, p=0.26). Golimumab (24wk: MD=-5.27, p< 0.001), baricitinib (24wk: MD=-4.06, p< 0.001), sarilumab (24wk: MD=-3.15, p< 0.001), tocilizumab (24wk: MD=-3.69, p< 0.001) and tofacitinib (12wk: MD=-4.44, p< 0.001) were also superior to placebo in reducing fatigue in RA. A dose/effect relationship was observed for sarilumab, tocilizumab and tofacitinib. In spondyloarthritis, secukinumab was superior to placebo in reducing fatigue at 16wk (MD=-4.15, p< 0.001), with a dose/effect relationship also observed (Figure). The narrative results of the RCTs not included in the meta-analysis indicated that several other pharmacological interventions were efficacious in reducing fatigue, with reassuring safety results.
Conclusion: Pharmacological interventions are efficacious and safe for the management of fatigue in people with I-RMD.

B. Farisogullari: None; E. Santos: None; E. Dures: None; R. Geenen: None; P. Machado: AbbVie/Abbott, 2, 6, Bristol-Myers Squibb(BMS), 2, 6, Celgene, 2, 6, Eli Lilly, 2, 6, Galapagos, 2, 6, Janssen, 2, 6, Merck/MSD, 2, 6, Novartis, 2, 6, Orphazyme, 2, 6, Pfizer, 2, 6, Roche, 2, 6, UCB, 2, 6.



