Abstract Session
Epidemiology, health policy and outcomes
Session: Abstracts: Epidemiology & Public Health I (0739–0744)
0739: Major Adverse Cardiovascular Event and Venous Thromboembolism Risk Comparing Advanced Therapies Among Individuals with Axial Spondylarthritis and Psoriatic Arthritis
Sunday, November 12, 2023
2:00 PM - 2:10 PM PT
Location: Room 33A-C
- SM
Sali Merjanah, MD
Boston University
Newmarket, NH, United StatesDisclosure information not submitted.
Presenting Author(s)
Sali Merjanah1, Devin Driscoll2, Christine Peloquin3, Jean liew3 and Maureen Dubreuil4, 1Boston University Chobanian & Avedisian School of Medicine, Boston, MA, 2Boston University/Boston Medical Center, Boston, MA, 3Boston University, Boston, MA, 4Department of Rheumatology, Boston University School of Medicine, Milton, MA
Background/Purpose: Individuals with axial spondyloarthritis (axSpA) and psoriatic arthritis (PsA) have increased cardiovascular risk compared to the general population, which is partly explained by systemic inflammation. While emerging evidence suggests tumor necrosis factor inhibitors (TNFi) may be cardioprotective, there are safety concerns for Janus kinase inhibitors (JAKibs) in regard to major adverse cardiovascular events (MACE) and venous thromboembolism (VTE). We aimed to assess the risk of MACE and VTE in axSpA and PsA comparing users of JAKibs versus TNFi.
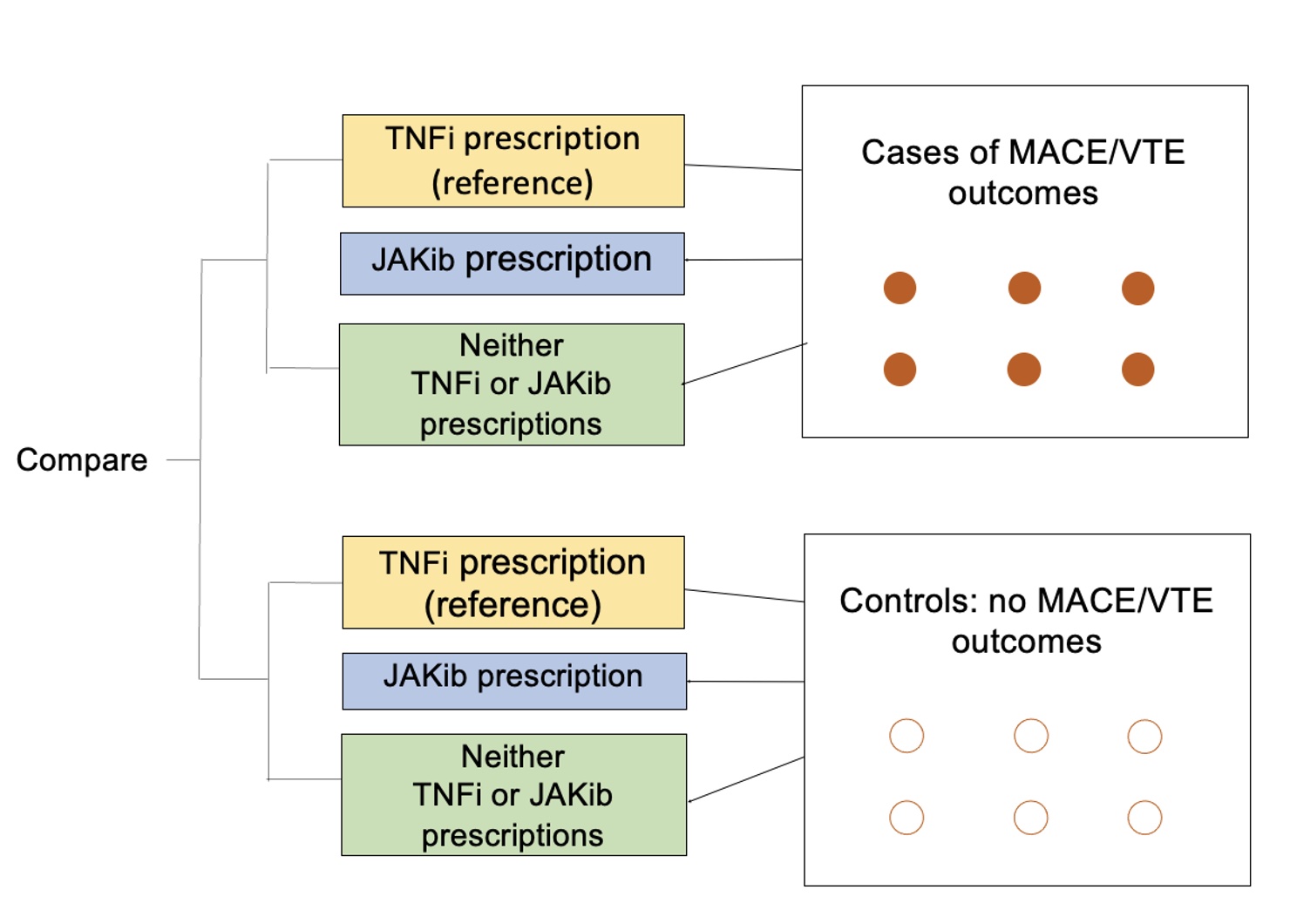
Methods: We conducted two nested case-control studies (Figure 1) using 2006-2021 data from the MarketScan Commercial Claims and Encounters (Merative) Database, which includes de-identified, patient-specific data of reimbursed healthcare claims. We included adults aged 18-65 years with ≥1 inpatient or 2 outpatient axSpA or PsA ICD-9/-10 diagnosis codes separated by ≥7 days.
For the first case control study, MACE was defined as the first inpatient claim for myocardial infarction or stroke/transient ischemic attack; for the second case control study, VTE was defined using the first inpatient/outpatient claim. The index date was the date of the MACE or VTE outcome among cases; or matching the case's outcome date among controls. Cases and controls were matched 1:4 by age, gender and axSpA/PsA diagnosis date using risk-set sampling. We evaluated drug exposure (TNFi or JAKib use or neither) using pharmacy claims and CPT codes within 6 months prior to the index date.
We assessed the association of drug exposure (referent: TNFi use) with the outcomes of MACE and VTE in separate models, using conditional logistic regression with adjustment for potential confounders (age, cardiovascular disease history, comorbidities, and tobacco use) to calculate odds ratios (OR) and 95% confidence intervals (CI).
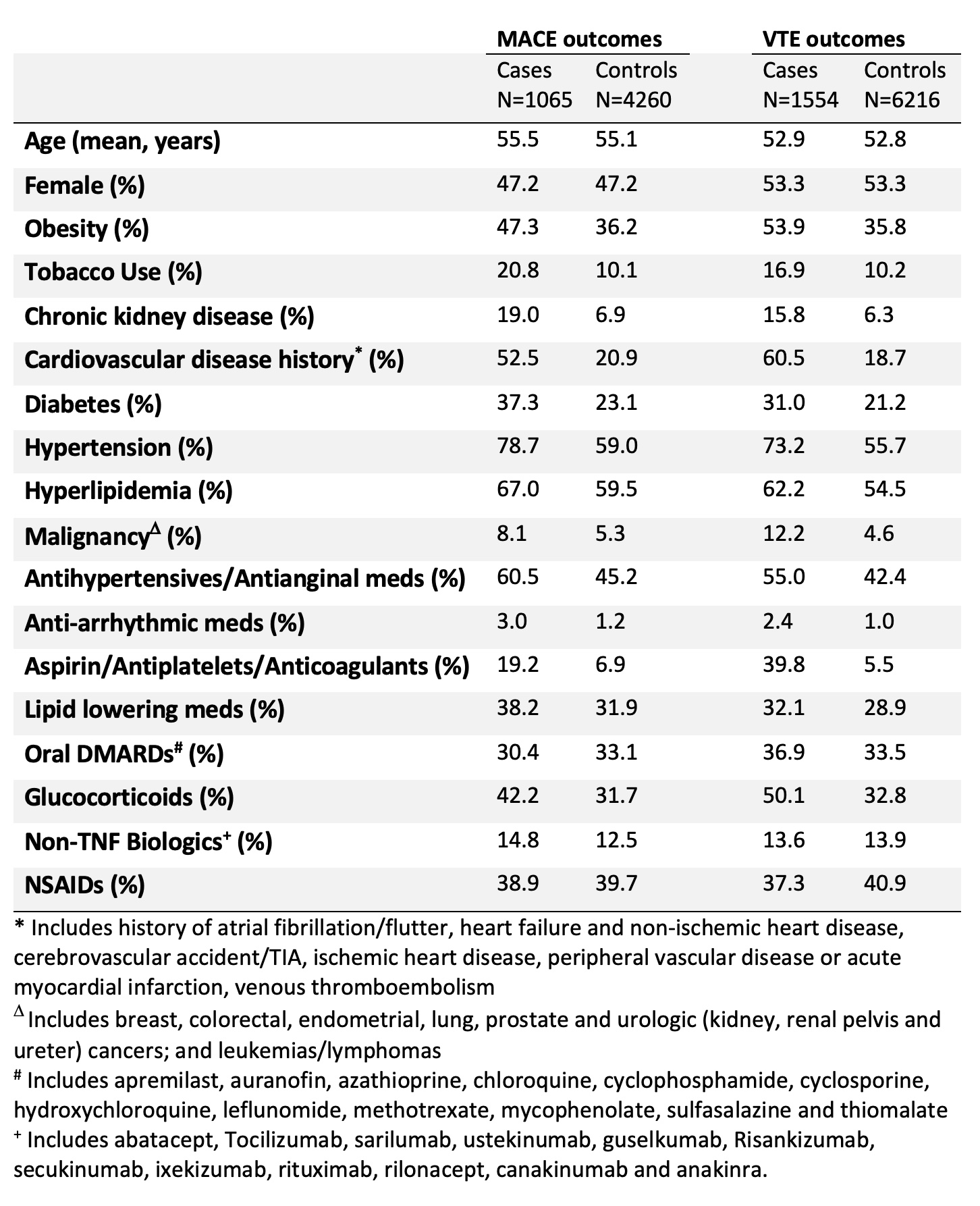
Results: We identified 1065 MACE cases among 5325 adults with AxSpA or PSA and 1554 VTE cases among 7770 axSpA/PsA subjects. Demographics and clinical characteristics are shown in Table 1. Comorbidities including cardiovascular disease history, obesity, diabetes and hypertension were more commonly seen in cases versus controls.
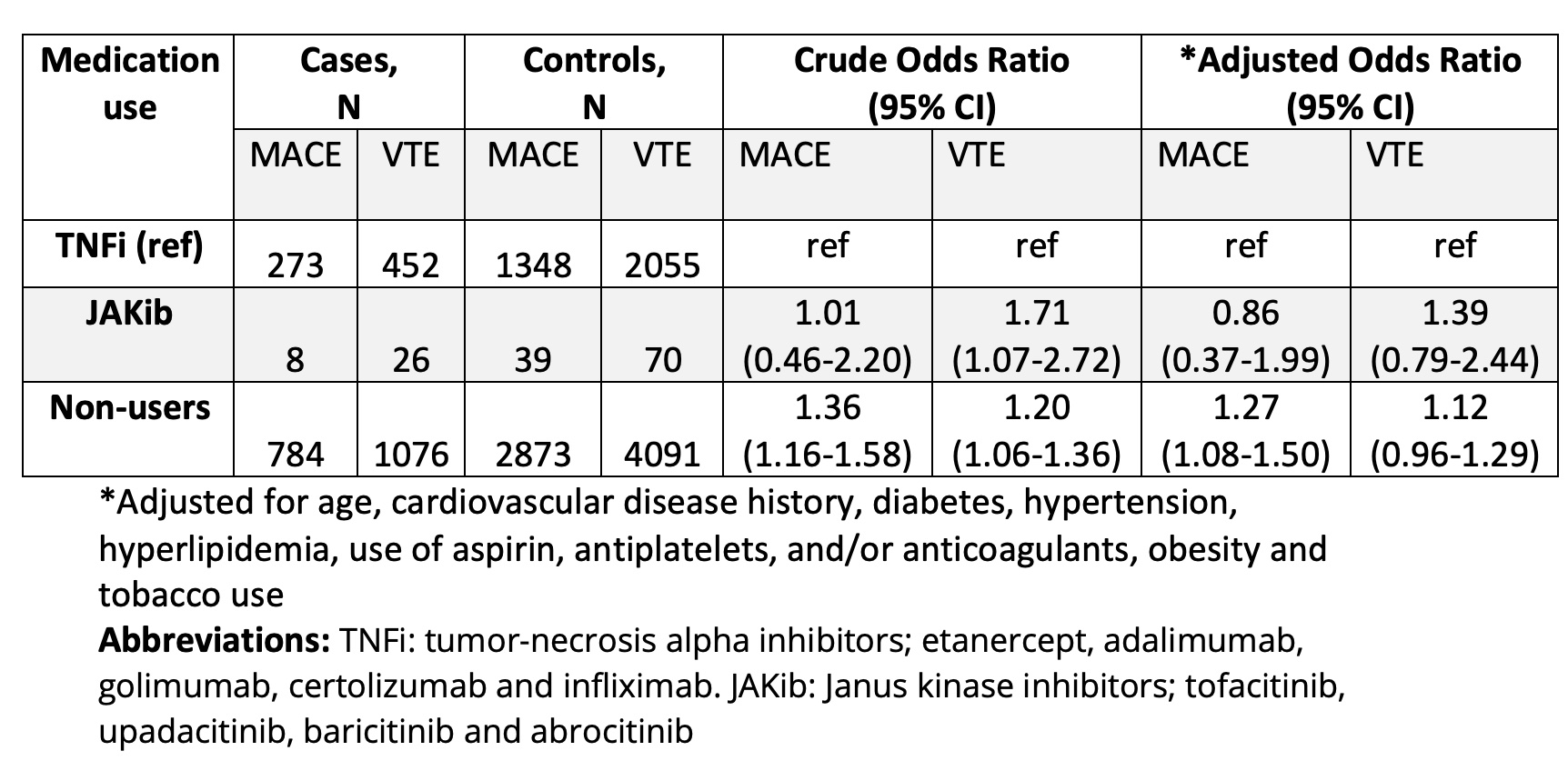
In the MACE nested case control study, 47 (0.9%) were JAKib users, 1621 (30.4%) were TNFi users and 3657 (68.7%) were non-users of JAKib and TNFi. In the VTE sample, 96 (1.2%) were JAKib users, 2507 (32.3%) were TNFi users and 5167 (66.5%) were non-users.
Non-users of TNFi and JAKibs had 1.3 times the odds of MACE relative to TNFi users (95% CI 1.08-1.50; Table 2). In JAKib users versus TNFi users, the odds of MACE was not increased (OR 0.86, 95% CI 0.37-1.99). The odds ratio for VTE was 1.39 among JAKib users however, this was not statistically significant after confounder adjustment (95% CI 0.79-2.44).
Conclusion: Using a large US insurance claims database, we did not find a statistically significant association between JAKib use and risk of MACE or VTE, compared with TNFi use among individuals with axSpA and PsA, although there was a trend toward increased VTE events among JAKib users. The accrual of further data is needed to better study the comparative safety of JAKibs versus TNFi in this study population.
S. Merjanah: None; D. Driscoll: None; C. Peloquin: None; J. liew: None; M. Dubreuil: Amgen, 2, Pfizer, 5, UCB Pharma, 2.
Background/Purpose: Individuals with axial spondyloarthritis (axSpA) and psoriatic arthritis (PsA) have increased cardiovascular risk compared to the general population, which is partly explained by systemic inflammation. While emerging evidence suggests tumor necrosis factor inhibitors (TNFi) may be cardioprotective, there are safety concerns for Janus kinase inhibitors (JAKibs) in regard to major adverse cardiovascular events (MACE) and venous thromboembolism (VTE). We aimed to assess the risk of MACE and VTE in axSpA and PsA comparing users of JAKibs versus TNFi.
Methods: We conducted two nested case-control studies (Figure 1) using 2006-2021 data from the MarketScan Commercial Claims and Encounters (Merative) Database, which includes de-identified, patient-specific data of reimbursed healthcare claims. We included adults aged 18-65 years with ≥1 inpatient or 2 outpatient axSpA or PsA ICD-9/-10 diagnosis codes separated by ≥7 days.
For the first case control study, MACE was defined as the first inpatient claim for myocardial infarction or stroke/transient ischemic attack; for the second case control study, VTE was defined using the first inpatient/outpatient claim. The index date was the date of the MACE or VTE outcome among cases; or matching the case's outcome date among controls. Cases and controls were matched 1:4 by age, gender and axSpA/PsA diagnosis date using risk-set sampling. We evaluated drug exposure (TNFi or JAKib use or neither) using pharmacy claims and CPT codes within 6 months prior to the index date.
We assessed the association of drug exposure (referent: TNFi use) with the outcomes of MACE and VTE in separate models, using conditional logistic regression with adjustment for potential confounders (age, cardiovascular disease history, comorbidities, and tobacco use) to calculate odds ratios (OR) and 95% confidence intervals (CI).
Results: We identified 1065 MACE cases among 5325 adults with AxSpA or PSA and 1554 VTE cases among 7770 axSpA/PsA subjects. Demographics and clinical characteristics are shown in Table 1. Comorbidities including cardiovascular disease history, obesity, diabetes and hypertension were more commonly seen in cases versus controls.
In the MACE nested case control study, 47 (0.9%) were JAKib users, 1621 (30.4%) were TNFi users and 3657 (68.7%) were non-users of JAKib and TNFi. In the VTE sample, 96 (1.2%) were JAKib users, 2507 (32.3%) were TNFi users and 5167 (66.5%) were non-users.
Non-users of TNFi and JAKibs had 1.3 times the odds of MACE relative to TNFi users (95% CI 1.08-1.50; Table 2). In JAKib users versus TNFi users, the odds of MACE was not increased (OR 0.86, 95% CI 0.37-1.99). The odds ratio for VTE was 1.39 among JAKib users however, this was not statistically significant after confounder adjustment (95% CI 0.79-2.44).
Conclusion: Using a large US insurance claims database, we did not find a statistically significant association between JAKib use and risk of MACE or VTE, compared with TNFi use among individuals with axSpA and PsA, although there was a trend toward increased VTE events among JAKib users. The accrual of further data is needed to better study the comparative safety of JAKibs versus TNFi in this study population.

Figure 1. Study design for nested case-control study with cases and controls identified by outcome status and 3-level medication exposure status assessed in 6 months prior to outcome (index date).

Table 1. Baseline demographics and clinical characteristics of study subjects for each case-control sample stratified by MACE and VTE outcomes.

Table 2. Association of each drug treatment category with major cardiovascular event and venous thromboembolism outcomes, among individuals with axial spondyloarthritis and psoriatic arthritis
S. Merjanah: None; D. Driscoll: None; C. Peloquin: None; J. liew: None; M. Dubreuil: Amgen, 2, Pfizer, 5, UCB Pharma, 2.



