Poster Session B
Pediatric autoimmune diseases: Kawasaki disease, juvenile dermatomyositis and juvenile localized scleroderma
Session: (1221–1255) Pediatric Rheumatology – Clinical Poster II: Connective Tissue Disease
1229: Longitudinal Assessment of Self-reported Executive Function in Youth with Childhood-Onset Lupus
Monday, November 13, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- JL
Justine Ledochowski, PhD
The Hospital for Sick Children
Toronto, ON, CanadaDisclosure information not submitted.
Abstract Poster Presenter(s)
Tala El Tal1, justine ledochowski2, Sarah Mossad3, Victoria Lishak3, Ibrahim Mohamed4, Joanna Law3, Lawrence Ng3, Paris Moaf3, Asha Jeyanathan3, Adrienne Davis3, Linda Hiraki3, Deborah Levy3, Zahi Touma5, Natoshia Cunningham6, Ashley Danguecan3 and Andrea Knight3, 1Division of Rheumatology, The Hospital for Sick Children, andDivision of Rheumatology, Children’s Hospital of Eastern Ontario, Ottawa, ON, Canada, 2Department of Psychology, The Hospital for Sick Children, Toronto, ON, Canada, 3The Hospital for Sick Children, Toronto, ON, Canada, 4The Hospital for Sick Children, Brampton, ON, Canada, 5University of Toronto, Toronto, ON, Canada, 6Michigan State University, Grand Rapids, MI
Background/Purpose: Cognitive dysfunction (including executive dysfunction) affects up to 60% of youth with childhood-onset systemic lupus erythematosus (cSLE), with potential adverse effects on health-related quality of life, and medication adherence. Patients with cSLE are often diagnosed during a critical period of neurodevelopment, and changes in executive function (EF) over time have not been well-studied in cSLE compared to typically developing children. We aimed to characterize the course of EF over time in cSLE patients matched to healthy controls.
Methods: We examined a prospective longitudinal sample of patients with cSLE (ages 12-17, met ACR or SLICC classification criteria for SLE), along with age and sex-matched healthy controls over two time points (baseline and 1 year later) collected from January 2020 to May 2023. Participants completed the self-report Behavior Rating Inventory of Executive Function 2 (BRIEF-2) that generated overall EF difficulties index score (global executive composite (GEC)), and three sub-indices – behavioral regulation index (BRI), cognitive regulation index (CRI), and emotion regulation index (ERI)). T-scores are transformations of raw scores based on age and sex norms. A higher T-score indicates a greater degree of difficulty in executive functioning. Disease measures including SLE Disease Activity Index 2000 (SLEDAI-2K), SLICC/ACR damage index (SDI), glucocorticoid use at visit and cumulative use, and disease duration were collected. The primary outcome was EF measured by GEC, BRI, CRI, and ERI T-scores, with a focus on GEC. T-tests were performed to examine within-group and between-group comparisons of mean scores at baseline and 1-year, and of change in mean scores (1 year minus baseline) respectively, for cSLE participants and controls.
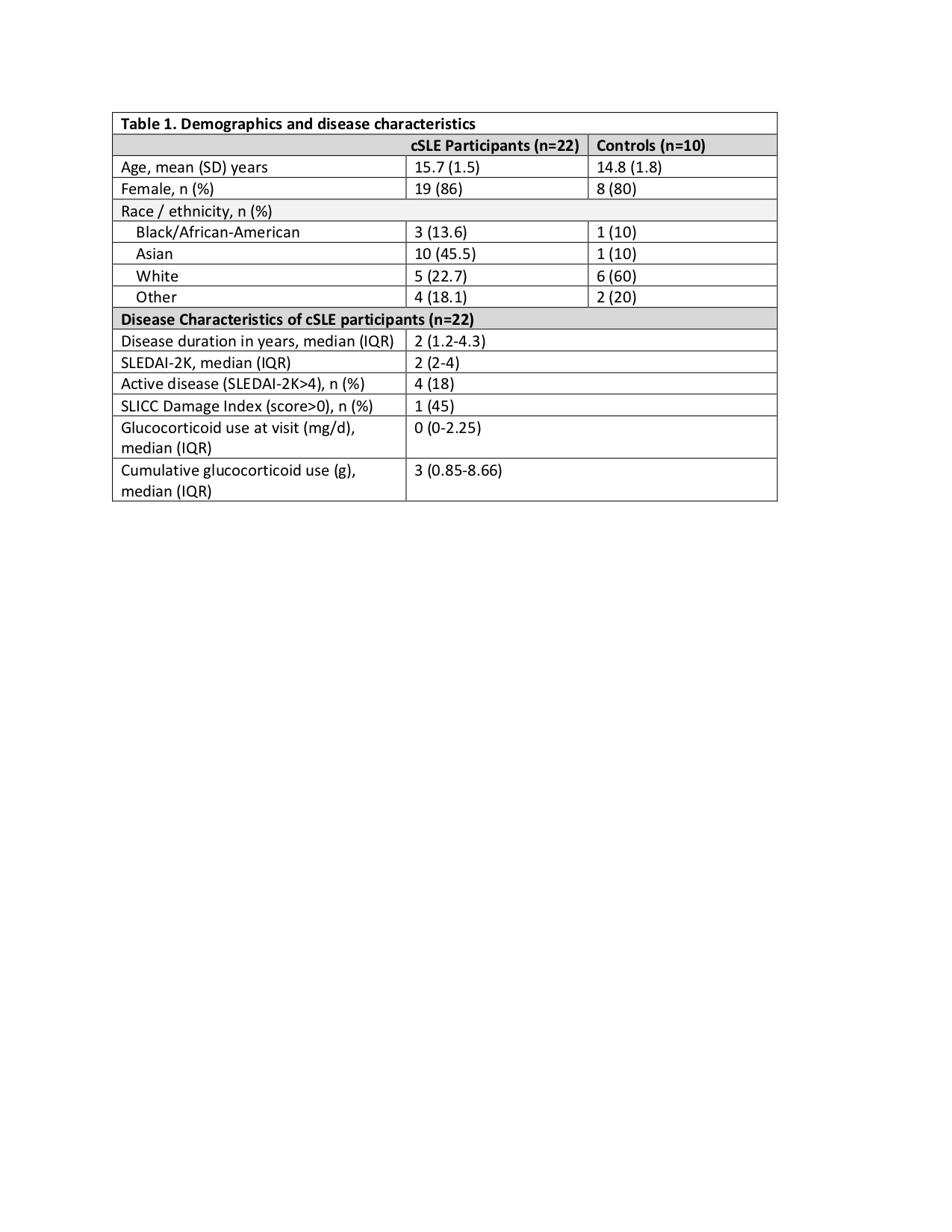
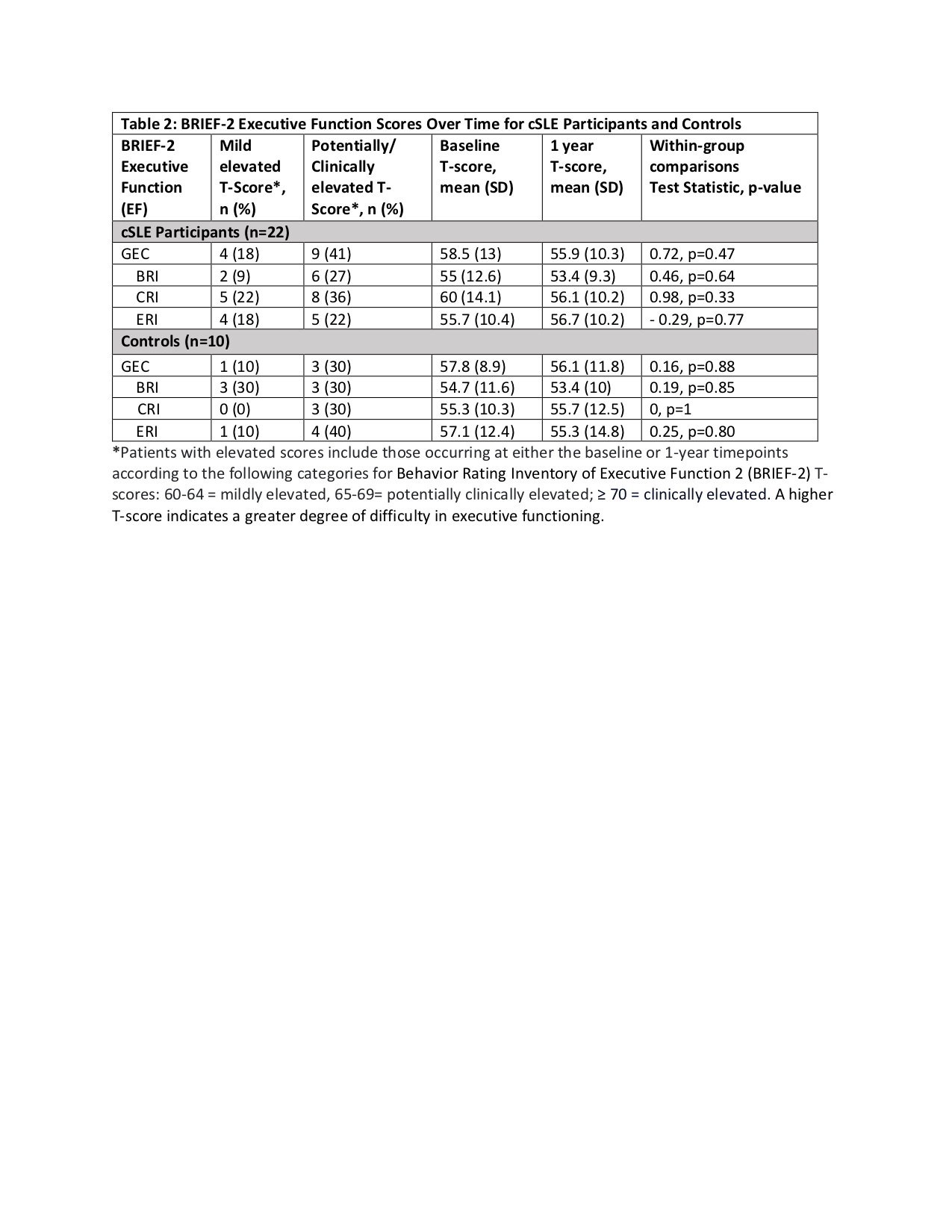
Results: Twenty-two patients with cSLE and 10 controls were included. For cSLE, the mean age was 15.7 ± SD 1.5 years, 86% were female, median SLEDAI-2K score was 2 (IQR 2-4) and median disease duration was 2 years (IQR 1.2-4.3) (Table1). In cSLE patients compared to controls, mildly elevated GEC T-scores were present in 18% (n=4) vs. 1 (10%); potentially and/or clinically elevated GEC T-scores were present in 41% (n=9) vs 30 %(n=3) (Table2) at baseline or 1-year timepoints. While the mean CRI T-score showed a small improvement from baseline and 1 year later (60 vs 56.1), there were no statistically significant differences for within-group comparisons of any of the BRIEF-T scores over time for both cSLE and controls (Table 2). There were also no significant differences between the cSLE and control groups for mean change in the BRIEF-T scores over time (GEC, t-value=0.78, p=0.43; BRI t-value= -0.28, p=0.77, CRI t-value= -1.2, p=0.20, ERI t-value 0.99, p=0.32).
Conclusion: Self-reported EF was stable over a one-year period in this small cohort of patients with cSLE and controls. Although no significant differences in mean scores for EF were found between cSLE and controls, 59% (versus 40% of HC) had at least mild difficulties in EF. More longitudinal research is needed with longer follow up to identify cognitive function trajectories and at-risk groups for executive dysfunction in cSLE.
T. El Tal: None; j. ledochowski: None; S. Mossad: None; V. Lishak: None; I. Mohamed: None; J. Law: None; L. Ng: None; P. Moaf: None; A. Jeyanathan: None; A. Davis: None; L. Hiraki: None; D. Levy: None; Z. Touma: AstraZeneca, 2, GSK, 2; N. Cunningham: DoD, 12, DoD Transformative Vision Award (starting Oct 2023) and CARRA Transdisciplinary Research Grant (NCE). I also have other current funding support.; A. Danguecan: None; A. Knight: Pfizer, 6.
Background/Purpose: Cognitive dysfunction (including executive dysfunction) affects up to 60% of youth with childhood-onset systemic lupus erythematosus (cSLE), with potential adverse effects on health-related quality of life, and medication adherence. Patients with cSLE are often diagnosed during a critical period of neurodevelopment, and changes in executive function (EF) over time have not been well-studied in cSLE compared to typically developing children. We aimed to characterize the course of EF over time in cSLE patients matched to healthy controls.
Methods: We examined a prospective longitudinal sample of patients with cSLE (ages 12-17, met ACR or SLICC classification criteria for SLE), along with age and sex-matched healthy controls over two time points (baseline and 1 year later) collected from January 2020 to May 2023. Participants completed the self-report Behavior Rating Inventory of Executive Function 2 (BRIEF-2) that generated overall EF difficulties index score (global executive composite (GEC)), and three sub-indices – behavioral regulation index (BRI), cognitive regulation index (CRI), and emotion regulation index (ERI)). T-scores are transformations of raw scores based on age and sex norms. A higher T-score indicates a greater degree of difficulty in executive functioning. Disease measures including SLE Disease Activity Index 2000 (SLEDAI-2K), SLICC/ACR damage index (SDI), glucocorticoid use at visit and cumulative use, and disease duration were collected. The primary outcome was EF measured by GEC, BRI, CRI, and ERI T-scores, with a focus on GEC. T-tests were performed to examine within-group and between-group comparisons of mean scores at baseline and 1-year, and of change in mean scores (1 year minus baseline) respectively, for cSLE participants and controls.
Results: Twenty-two patients with cSLE and 10 controls were included. For cSLE, the mean age was 15.7 ± SD 1.5 years, 86% were female, median SLEDAI-2K score was 2 (IQR 2-4) and median disease duration was 2 years (IQR 1.2-4.3) (Table1). In cSLE patients compared to controls, mildly elevated GEC T-scores were present in 18% (n=4) vs. 1 (10%); potentially and/or clinically elevated GEC T-scores were present in 41% (n=9) vs 30 %(n=3) (Table2) at baseline or 1-year timepoints. While the mean CRI T-score showed a small improvement from baseline and 1 year later (60 vs 56.1), there were no statistically significant differences for within-group comparisons of any of the BRIEF-T scores over time for both cSLE and controls (Table 2). There were also no significant differences between the cSLE and control groups for mean change in the BRIEF-T scores over time (GEC, t-value=0.78, p=0.43; BRI t-value= -0.28, p=0.77, CRI t-value= -1.2, p=0.20, ERI t-value 0.99, p=0.32).
Conclusion: Self-reported EF was stable over a one-year period in this small cohort of patients with cSLE and controls. Although no significant differences in mean scores for EF were found between cSLE and controls, 59% (versus 40% of HC) had at least mild difficulties in EF. More longitudinal research is needed with longer follow up to identify cognitive function trajectories and at-risk groups for executive dysfunction in cSLE.


T. El Tal: None; j. ledochowski: None; S. Mossad: None; V. Lishak: None; I. Mohamed: None; J. Law: None; L. Ng: None; P. Moaf: None; A. Jeyanathan: None; A. Davis: None; L. Hiraki: None; D. Levy: None; Z. Touma: AstraZeneca, 2, GSK, 2; N. Cunningham: DoD, 12, DoD Transformative Vision Award (starting Oct 2023) and CARRA Transdisciplinary Research Grant (NCE). I also have other current funding support.; A. Danguecan: None; A. Knight: Pfizer, 6.



