Poster Session A
Infection-related rheumatic syndromes
Session: (0196–0228) Infection-related Rheumatic Disease Poster
0218: Incidence and Risk Factors for Breakthrough COVID-19 After Tixagevimab/Cilgavimab Among Patients with Systemic Autoimmune Rheumatic Diseases
Sunday, November 12, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- YK
Yumeko Kawano, MD
Brigham and Women's Hospital
Boston, MA, United StatesDisclosure information not submitted.
Abstract Poster Presenter(s)
Yumeko Kawano1, Xiaosong Wang1, Naomi Patel2, Grace Qian1, Emily Kowalski1, Katarina Bade3, Kathleen Vanni1, Accelerating Medicines Partnership (AMP): RA/SLE1, Zachary Williams2, Claire Cook2, Shruthi Srivatsan2, Zachary Wallace4 and Jeffrey Sparks5, 1Brigham and Women's Hospital, Boston, MA, 2Massachusetts General Hospital, Boston, MA, 3Brigham and Women's Hospital, Boston, MA, 4Massachusetts General Hospital, Newton, MA, 5Division of Rheumatology, Inflammation, and Immunity, Brigham and Women’s Hospital and Harvard Medical School, Boston, MA
Background/Purpose: During the height of the COVID-19 pandemic in the United States, tixagevimab/cilgavimab (Evusheld), a combination of monoclonal antibodies directed against the SARS-CoV2 spike protein, received emergency use authorization (EUA) for use as pre-exposure prophylaxis (PrEP) against COVID-19. However, little is known regarding real-world experience with PrEP in patients with systemic autoimmune rheumatic diseases (SARDs). We aimed to determine the incidence of and risk factors for breakthrough COVID-19 after tixagevimab/cilgavimab among patients with SARDs.
Methods: We conducted a retrospective cohort study of patients with SARDs at a large U.S. healthcare system. We identified all patients who received tixagevimab/cilgavimab as PrEP between 1/2/2022 and 11/16/2022. The index date was the date of the first dose of tixagevimab/cilgavimab. We collected demographics, medications, prior COVID-19, and SARS-CoV2 spike antibody levels prior to index date from electronic query. SARD diagnoses, the presence of additional PrEP indications (e.g., prior organ transplant, active cancer treatment), and COVID-19 course were confirmed by medical record review. The primary outcome was breakthrough COVID-19 confirmed by polymerase chain reaction or an antigen test reported to a physician. Censoring occurred at earliest of breakthrough COVID-19, death, or end of study (November 16, 2022). We used multivariable Cox regression models adjusting for baseline factors to identify risk factors for breakthrough COVID-19.
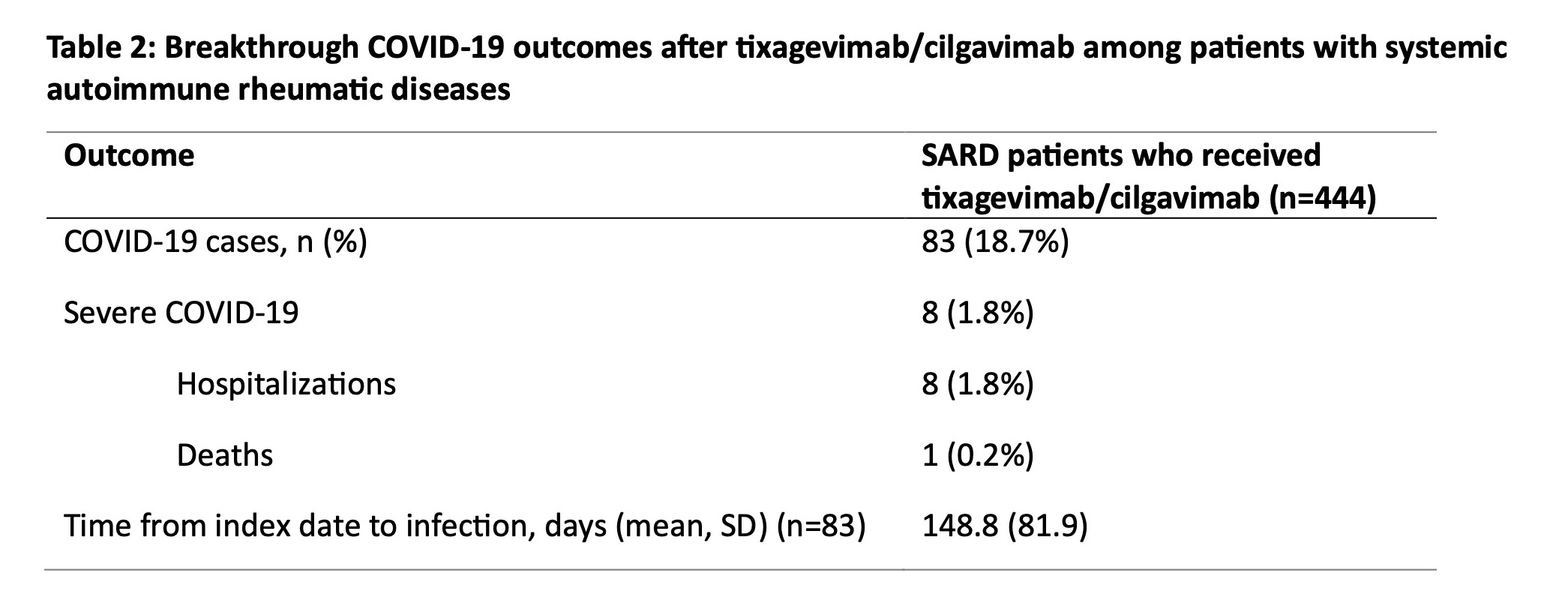
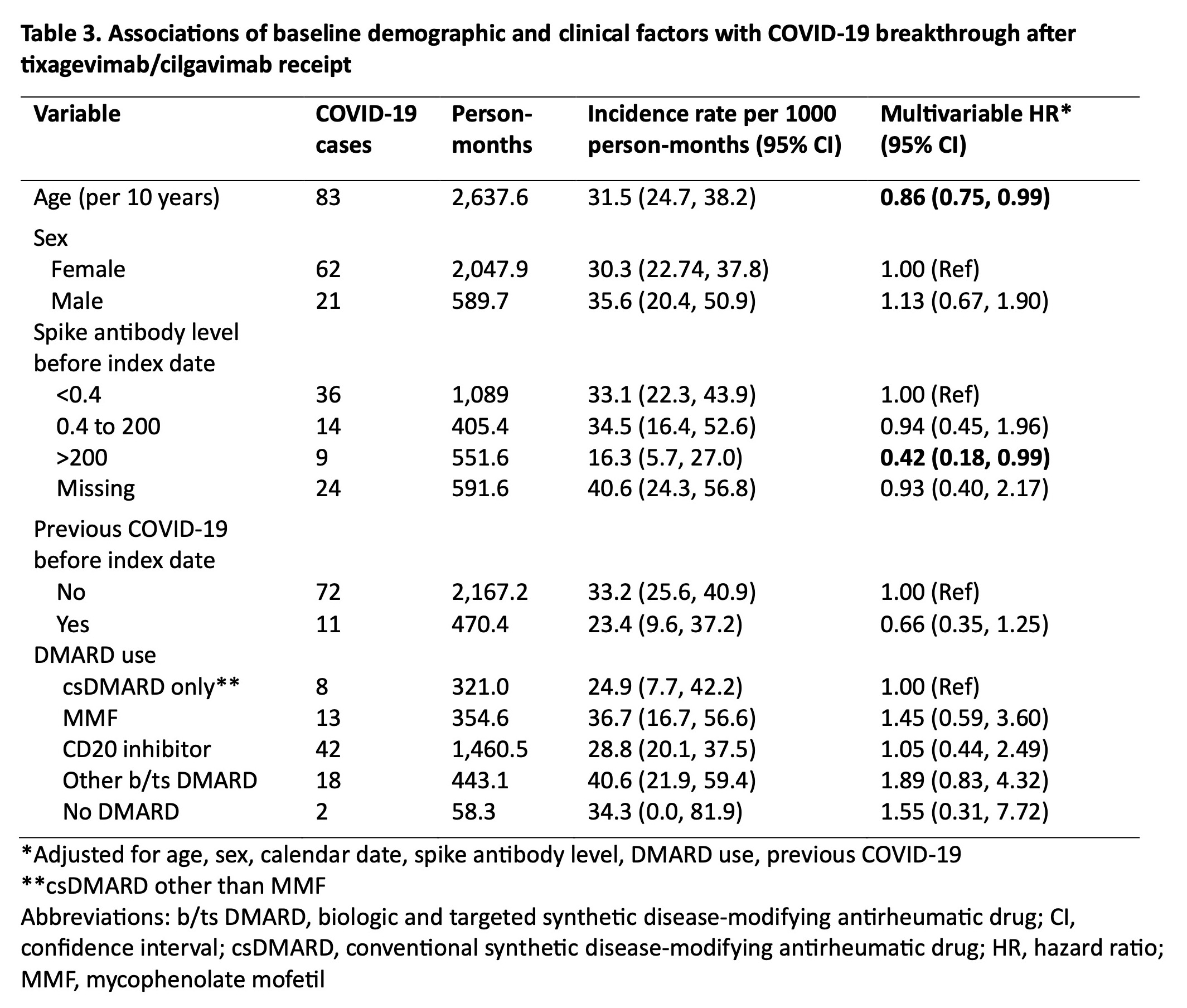
Results: We identified 444 patients with SARDs who received tixagevimab/cilgavimab (mean age 62.0 years, 78.2% female, 79.3% white) (Table 1). The most common SARD diagnoses were rheumatoid arthritis (43.7%), systemic lupus erythematosus (14.9%), and ANCA-associated vasculitis (11.7%). There were 83 (18.7%) breakthrough COVID-19 cases, of which 8 (1.8%) required hospitalization and 1 (0.2%) died (Table 2). The overall incidence rate was 31.5 per 1000 person-months (95% CI 24.70, 38.24). Older age was inversely associated with breakthrough COVID-19 (adjusted hazard ratio [aHR] 0.86 per 10 years, 95% CI 0.75, 0.99). Higher baseline spike antibody levels were associated with lower risk of breakthrough COVID-19 (aHR 0.42, 95% CI 0.18, 0.99 for spike antibody levels >200 units vs. < 0.4 units). CD20 inhibitor users had a similar risk of breakthrough COVID-19 (aHR 1.05, 95% CI 0.44, 2.49) compared to patients on conventional synthetic DMARDs (Table 3).
Conclusion: In this study spanning the period of viral neutralizing abilityfor tixagevimab/cilgavimab, we found that patients with SARDs had frequent breakthrough COVID-19 after PrEP, but the proportion of severe COVID-19 was low. Further, we found no associations between DMARD type, including anti-CD20 inhibitors, with the risk of breakthrough COVID-19. Older age and higher baseline spike antibody level were protective against breakthrough infection, highlighting the continued need for a multimodal approach (e.g. shielding behaviors, vaccinations) to prevent COVID-19 in this vulnerable population as newer generation PrEP monoclonal antibodies against SARS-CoV2 are being developed.

Y. Kawano: None; X. Wang: None; N. Patel: Arrivo Bio, 2, Chronius Health, 2, FVC Health, 2; G. Qian: None; E. Kowalski: None; K. Bade: None; K. Vanni: None; A. Medicines Partnership (AMP): RA/SLE: None; Z. Williams: None; C. Cook: None; S. Srivatsan: None; Z. Wallace: BioCryst, 2, Bristol-Myers Squibb(BMS), 5, Horizon, 1, 2, 5, MedPace, 2, Novartis, 1, PPD, 2, Sanofi, 1, 5, Shionogi, 1, Visterra, 1, 2, Zenas, 1, 2; J. Sparks: AbbVie, 2, Amgen, 2, Boehringer Ingelheim, 2, Bristol-Myers Squibb, 2, 5, Gilead, 2, Inova Diagnostics, 2, Janssen, 2, Optum, 2, Pfizer, 2, ReCor, 2.
Background/Purpose: During the height of the COVID-19 pandemic in the United States, tixagevimab/cilgavimab (Evusheld), a combination of monoclonal antibodies directed against the SARS-CoV2 spike protein, received emergency use authorization (EUA) for use as pre-exposure prophylaxis (PrEP) against COVID-19. However, little is known regarding real-world experience with PrEP in patients with systemic autoimmune rheumatic diseases (SARDs). We aimed to determine the incidence of and risk factors for breakthrough COVID-19 after tixagevimab/cilgavimab among patients with SARDs.
Methods: We conducted a retrospective cohort study of patients with SARDs at a large U.S. healthcare system. We identified all patients who received tixagevimab/cilgavimab as PrEP between 1/2/2022 and 11/16/2022. The index date was the date of the first dose of tixagevimab/cilgavimab. We collected demographics, medications, prior COVID-19, and SARS-CoV2 spike antibody levels prior to index date from electronic query. SARD diagnoses, the presence of additional PrEP indications (e.g., prior organ transplant, active cancer treatment), and COVID-19 course were confirmed by medical record review. The primary outcome was breakthrough COVID-19 confirmed by polymerase chain reaction or an antigen test reported to a physician. Censoring occurred at earliest of breakthrough COVID-19, death, or end of study (November 16, 2022). We used multivariable Cox regression models adjusting for baseline factors to identify risk factors for breakthrough COVID-19.
Results: We identified 444 patients with SARDs who received tixagevimab/cilgavimab (mean age 62.0 years, 78.2% female, 79.3% white) (Table 1). The most common SARD diagnoses were rheumatoid arthritis (43.7%), systemic lupus erythematosus (14.9%), and ANCA-associated vasculitis (11.7%). There were 83 (18.7%) breakthrough COVID-19 cases, of which 8 (1.8%) required hospitalization and 1 (0.2%) died (Table 2). The overall incidence rate was 31.5 per 1000 person-months (95% CI 24.70, 38.24). Older age was inversely associated with breakthrough COVID-19 (adjusted hazard ratio [aHR] 0.86 per 10 years, 95% CI 0.75, 0.99). Higher baseline spike antibody levels were associated with lower risk of breakthrough COVID-19 (aHR 0.42, 95% CI 0.18, 0.99 for spike antibody levels >200 units vs. < 0.4 units). CD20 inhibitor users had a similar risk of breakthrough COVID-19 (aHR 1.05, 95% CI 0.44, 2.49) compared to patients on conventional synthetic DMARDs (Table 3).
Conclusion: In this study spanning the period of viral neutralizing abilityfor tixagevimab/cilgavimab, we found that patients with SARDs had frequent breakthrough COVID-19 after PrEP, but the proportion of severe COVID-19 was low. Further, we found no associations between DMARD type, including anti-CD20 inhibitors, with the risk of breakthrough COVID-19. Older age and higher baseline spike antibody level were protective against breakthrough infection, highlighting the continued need for a multimodal approach (e.g. shielding behaviors, vaccinations) to prevent COVID-19 in this vulnerable population as newer generation PrEP monoclonal antibodies against SARS-CoV2 are being developed.



Y. Kawano: None; X. Wang: None; N. Patel: Arrivo Bio, 2, Chronius Health, 2, FVC Health, 2; G. Qian: None; E. Kowalski: None; K. Bade: None; K. Vanni: None; A. Medicines Partnership (AMP): RA/SLE: None; Z. Williams: None; C. Cook: None; S. Srivatsan: None; Z. Wallace: BioCryst, 2, Bristol-Myers Squibb(BMS), 5, Horizon, 1, 2, 5, MedPace, 2, Novartis, 1, PPD, 2, Sanofi, 1, 5, Shionogi, 1, Visterra, 1, 2, Zenas, 1, 2; J. Sparks: AbbVie, 2, Amgen, 2, Boehringer Ingelheim, 2, Bristol-Myers Squibb, 2, 5, Gilead, 2, Inova Diagnostics, 2, Janssen, 2, Optum, 2, Pfizer, 2, ReCor, 2.



