Poster Session A
Infection-related rheumatic syndromes
Session: (0196–0228) Infection-related Rheumatic Disease Poster
0213: When Should I Get My Next Booster? Active Surveillance of COVID-19 Breakthrough Infections in Canadian Patients with Immune-Mediated Inflammatory Diseases
Sunday, November 12, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- JT
Jeremiah Tan, BSc, -None-
University of British Columbia
Vancouver, BC, CanadaDisclosure information not submitted.
Abstract Poster Presenter(s)
Jeremiah Tan1, J. Antonio Avina-Zubieta2, Jennifer LF Lee3, Paul R. Fortin4, Ines Colmegna5, Lourdes Gonzalez Arreola2, Claudie Berger3, Maggie Larche6, Carol Hitchon7, Dawn Richards8, Nadine Lalonde9 and Sasha Bernatsky10, 1Arthritis Research Canada, Vancouver, Canada, Vancouver, BC, Canada, 2Arthritis Research Canada, Vancouver, BC, Canada, 3RI-MUHC, Montreal, QC, Canada, 4Centre ARThrite - CHU de Québec - Université Laval, Quebec City, QC, Canada, 5The Research Institute of the McGill University Health Centre, Montréal, QC, Canada, 6McMaster University, Hamilton, ON, Canada, 7University of Manitoba, Winnipeg, MB, Canada, 8Canadian Arthritis Patient Alliance, Toronto, ON, Canada, 9COVID-19 Global Rheumatology Alliance, London, ON, Canada, 10Research Institute of the McGill University Health Centre, Montreal, QC, Canada
Background/Purpose: Breakthrough COVID-19 infections are still a risk after vaccination and may be more common in patients with immune-mediated inflammatory diseases (IMIDs) than in the general population. Previous studies on breakthrough infections in IMID relied on retrospective databases; however, these are subject to limitations on COVID-19 test availability, making it hard to determine true infection prevalence as asymptomatic cases may have gone untested. We performed active surveillance of breakthrough COVID-19 infections, analyzing saliva samples via quantitative polymerase chain reaction (qPCR) in vaccinated individuals with IMIDs (systemic lupus erythematosus, rheumatic arthritis, psoriatic arthritis, spondylarthritis, inflammatory bowel disease, and scleroderma).
Methods: Adults with IMID were recruited from Canadian clinics and registries between September 2022 and March 2023 and asked to self-collect saliva samples monthly. Samples underwent batch qPCR testing, with each sample being tested twice, to qualitatively detect SARS-CoV-2 nucleic acids indicators (N1 and N2).
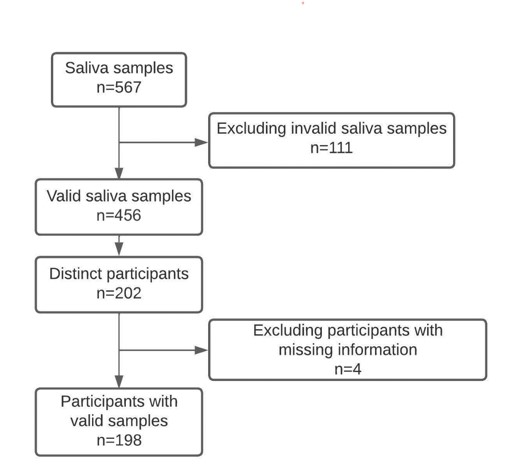
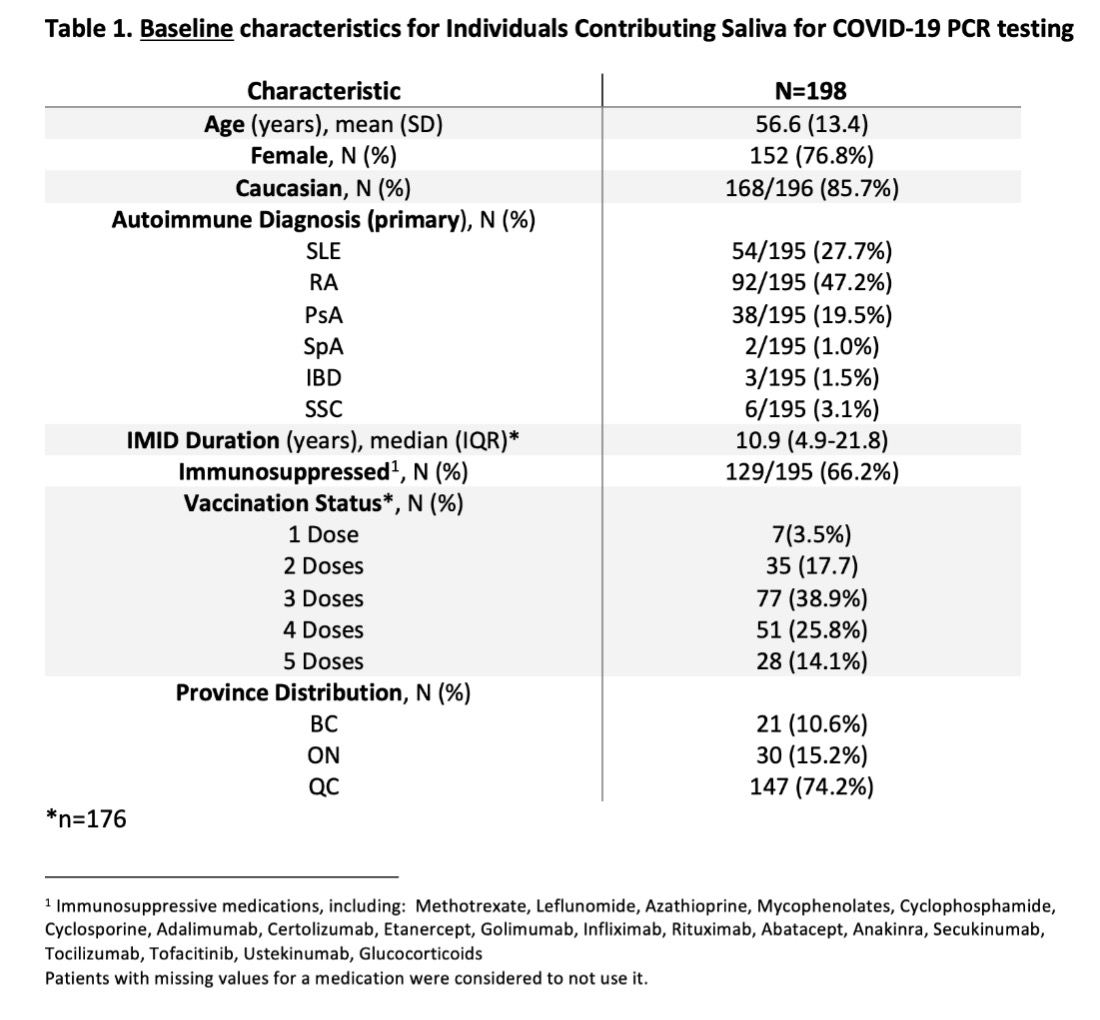
Results: To date, 202 patients have been enrolled, providing 458 valid samples (Fig. 1, Table 1). Most (66.2%) were on immunomodulatory medications, and the majority (78.8%) had received 3 or more vaccine doses. Only 5% of participants (n=10/202) met the N1 and N2 thresholds required to confirm COVID-19 positivity. Given the small number of positive results, we were unable to ascertain significant differences between demographic factors, including age, sex, immunosuppression, or vaccination status. However, participants who tested positive had a median time since their last vaccination that was considerably longer (278 days) than those who tested negative (165 days) (95% CI for difference,18-205). The majority (three-quarters) of positive saliva samples occurred in patients who were beyond 217 days of their last vaccine.
Conclusion: A 5% COVID-19 breakthrough infection rate aligns with a Canadian population-based cohort where breakthrough infections occurred in 5.4%-6.5% of fully vaccinated IMID patients (1). In our sample, those with breakthrough infections had a longer median time since vaccination (by 112 days), corroborating the currently held belief that protection against COVID wanes in the 3-4 months post-vaccination and beyond. Most infections occurred 7-8 months after the last vaccine dose. These findings will help patients, clinicians, and other stakeholders with decision-making in 2023-2024 and beyond.
Reference
J. Tan: None; J. Avina-Zubieta: None; J. Lee: None; P. Fortin: AbbVie, 1, AstraZeneca, 1, 6, GlaxoSmithKlein(GSK), 1, 6, Roche-Genentech, 1; I. Colmegna: None; L. Gonzalez Arreola: None; C. Berger: None; M. Larche: None; C. Hitchon: Astra Zeneca, 1, Pfizer, 5; D. Richards: None; N. Lalonde: Greenwish Life Science, 11, Merck/MSD, 11, Mind Medicine, 11, Predictmedix, 11, Takeda Pharma, 11, Tilray Brands, 11; S. Bernatsky: None.
Background/Purpose: Breakthrough COVID-19 infections are still a risk after vaccination and may be more common in patients with immune-mediated inflammatory diseases (IMIDs) than in the general population. Previous studies on breakthrough infections in IMID relied on retrospective databases; however, these are subject to limitations on COVID-19 test availability, making it hard to determine true infection prevalence as asymptomatic cases may have gone untested. We performed active surveillance of breakthrough COVID-19 infections, analyzing saliva samples via quantitative polymerase chain reaction (qPCR) in vaccinated individuals with IMIDs (systemic lupus erythematosus, rheumatic arthritis, psoriatic arthritis, spondylarthritis, inflammatory bowel disease, and scleroderma).
Methods: Adults with IMID were recruited from Canadian clinics and registries between September 2022 and March 2023 and asked to self-collect saliva samples monthly. Samples underwent batch qPCR testing, with each sample being tested twice, to qualitatively detect SARS-CoV-2 nucleic acids indicators (N1 and N2).
Results: To date, 202 patients have been enrolled, providing 458 valid samples (Fig. 1, Table 1). Most (66.2%) were on immunomodulatory medications, and the majority (78.8%) had received 3 or more vaccine doses. Only 5% of participants (n=10/202) met the N1 and N2 thresholds required to confirm COVID-19 positivity. Given the small number of positive results, we were unable to ascertain significant differences between demographic factors, including age, sex, immunosuppression, or vaccination status. However, participants who tested positive had a median time since their last vaccination that was considerably longer (278 days) than those who tested negative (165 days) (95% CI for difference,18-205). The majority (three-quarters) of positive saliva samples occurred in patients who were beyond 217 days of their last vaccine.
Conclusion: A 5% COVID-19 breakthrough infection rate aligns with a Canadian population-based cohort where breakthrough infections occurred in 5.4%-6.5% of fully vaccinated IMID patients (1). In our sample, those with breakthrough infections had a longer median time since vaccination (by 112 days), corroborating the currently held belief that protection against COVID wanes in the 3-4 months post-vaccination and beyond. Most infections occurred 7-8 months after the last vaccine dose. These findings will help patients, clinicians, and other stakeholders with decision-making in 2023-2024 and beyond.
Reference
- Widdifield J, Kwong JC, Chen S, Eder L, Benchimol EI, Kaplan GG, et al. Vaccine effectiveness against SARS-CoV-2 infection and severe outcomes among individuals with immune-mediated inflammatory diseases tested between March 1 and Nov 22, 2021, in Ontario, Canada: a population-based analysis. The Lancet Rheumatology [Internet]. 2022 Apr 14; Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9009845/

Figure 1: Participants and samples

Table 1. Baseline characteristics for individuals contributing saliva for COVID-19 PCR testing
J. Tan: None; J. Avina-Zubieta: None; J. Lee: None; P. Fortin: AbbVie, 1, AstraZeneca, 1, 6, GlaxoSmithKlein(GSK), 1, 6, Roche-Genentech, 1; I. Colmegna: None; L. Gonzalez Arreola: None; C. Berger: None; M. Larche: None; C. Hitchon: Astra Zeneca, 1, Pfizer, 5; D. Richards: None; N. Lalonde: Greenwish Life Science, 11, Merck/MSD, 11, Mind Medicine, 11, Predictmedix, 11, Takeda Pharma, 11, Tilray Brands, 11; S. Bernatsky: None.



