Poster Session A
Epidemiology, health policy and outcomes
Session: (0117–0144) Epidemiology & Public Health Poster I
0133: COVID-19 Vaccination-related Delayed Adverse Events Among Patients with Systemic Lupus Erythematosus: Results from the COVAD Study
Sunday, November 12, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- MF
Madiha Fathima, MD
Dubai Medical College & Karolinska Institute
Lombard, IL, United StatesDisclosure information not submitted.
Abstract Poster Presenter(s)
Madiha Fathima1, Bohdana Doskaliuk2, Julius Lindblom1, Elena Nikiphorou3, Chris Wincup4, Sreoshy Saha5, Syahrul Sazliyana Shaharir6, Wanruchada Katchamart7, Phonpen Akarawatcharangura Goo8, Lisa Traboco9, Yi-Ming Chen10, Kshitij Jagtap11, James Lilleker12, Arvind Nune13, John Pauling14, Vishwesh Agarwal15, Dey Dzifa16, CARLOS ENRIQUE TORO GUTIERREZ17, CARLO VINICIO CABALLERO18, Hector Chinoy19, Rohit Aggarwal20, Vikas Agarwal21, Latika Gupta22 and Ioannis Parodis1, 1Karolinska Institutet, Stockholm, Sweden, 2SHEI \"Ivano-Frankivsk national medical university\", Ivano-Frankivsk, Ukraine, 3King's College London, London, United Kingdom, 4King's College Hospital, London, United Kingdom, 5Mymensingh Medical College, Faridpur, Bangladesh, 6Faculty of Medicine, Universiti Kebangsaan Malaysia, 56000, Cheras, Kuala Lumpur, Malaysia, 7Mahidol University, Bangkok, Thailand, 8Department of Medicine, Queen Savang Vadhana Memorial Hospital, Chonburi, Thailand, 9University of the Philippines - Manila, St Luke's Medical Center - Bonifacio Global City, Paranaque, Manila, Philippines, 10Taichung Veterans General Hospital, Taichung, Taiwan, 11Seth Gordhandhas Sunderdas Medical College and King Edwards Memorial Hospital, Mumbai, India, 12Centre for Musculoskeletal Research, Division of Musculoskeletal and Dermatological Sciences, School of Biological Sciences, Faculty of Biology, Medicine and Health, Manchester Academic Health Science Centre, The University of Manchester, Manchester, UK; Manchester Centre for Clinical Neurosciences, Salford Royal NHS Foundation Trust, Salford, UK. Orcid ID: 0000-0002-9230-4137., Manchester, United Kingdom, 13Southport & Ormskirk NHS Foundation Trust, Liverpool, United Kingdom, 14North Bristol NHS Trust, Bristol, United Kingdom, 15Mahatma Gandhi Missions Medical College, Lucknow, India, 16Department of Medicine and Therapeutics, University of Ghana School of Medicine and Dentistry, College of Health Sciences, Korle-Bu, Accra, Ghana, 17Centro de Estudios de Reumatología y Dermatología SAS, Cali, Colombia, 18REUMACARIBE IPS, Barranquilla, Colombia, 19The University of Manchester, Sale, United Kingdom, 20University of Pittsburgh, Pittsburgh, PA, 21Sanjay Gandhi Postgraduate Institute of Medical Sciences (SGPGIMS), Lucknow, India, 22Royal Wolverhampton Trust, Wolverhampton/University of Manchester, United Kingdom
Background/Purpose: COVID-19 vaccines have been proven to be safe in healthy populations. However, data on delayed adverse effects (AEs) in people with autoimmune diseases (AIDs), including SLE has been lacking.
Methods: COVID-19 vaccination-related AEs reported longer than 7 days post-vaccination were assessed in the “COVID-19 Vaccination in Autoimmune Diseases 2” (COVAD-2) study in patients with SLE, rheumatic autoimmune diseases other than SLE (rAIDs), non-rheumatic autoimmune diseases (nrAIRDs), and healthy controls (HC). rAIDs included connective tissue diseases, inflammatory myopathies, and inflammatory arthritis, yet not SLE, whereas nrAIDs included inflammatory bowel disease and multiple sclerosis, among others. The COVAD-2 study comprised 157 collaborators across 106 countries and was conducted between February and June 2022. An online survey captured self-reported data related to COVID-19 vaccination-associated AEs in SLE, AIDs, and HC. We compared COVID-19 vaccination-related delayed AEs between groups using multivariable binary regression adjusting for age, gender, and ethnicity.
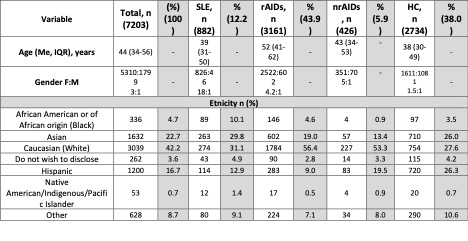
Results: Among 7203 participants, 882 (12.2%) SLE, 3161 (43.9%) rAIDs, 426 (5.9%) nrAIDs, and 2734 (38%) HC were included from a total of 10 783 respondents, with 74% female and 42.2% Caucasian (Table 1). People with SLE were young and middle-aged adults [median age 39 (31–50); rAIDs, 52 (41–62); nrAIDs 43 (34–53); HC 38 (30–49)].
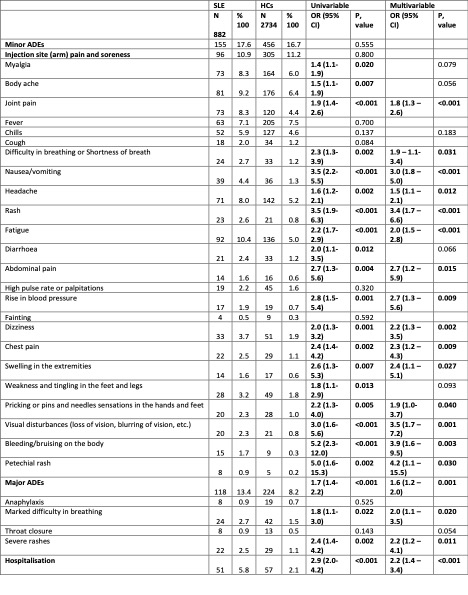
When compared to HC, people with SLE reported higher overall major [OR 1.6 (1.2–2.0)] and minor AEs (Table 2). SLE patients when compared to rAIDS reported more frequent episodes of severe rashes [OR 2.4 (1.3–4.2)], while compared to nrAIDs, SLE individuals reported an increased number of several minor AEs (joint pain [OR 2.4 (1.3–4.3)], fatigue [OR 2.0 (1.2–3.2)], body ache [OR 2.1 (1.2–3.7)]), and hospitalization [OR 2.3 (1.1–4.9)].
Vaccination with ChadOx1 nCOV-19 (Oxford/ AstraZeneca) in SLE patients led to a higher frequency in the incidence of diarrhea compared to other vaccines [OR 2.6 (1.1 – 6.2)], whereas the Moderna vaccine was associated with increased hospitalization frequencies [OR 3.0 (1.6–5.6)].
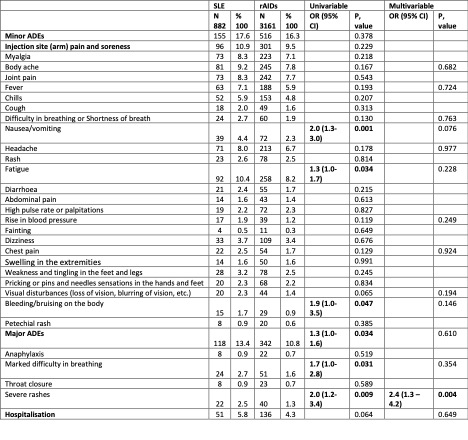
Patients with active versus non-active SLE reported similar frequencies of COVID-19 vaccine-related delayed minor AEs [17.5% versus 17.6%; OR 1.1 (0.8–1.6)]. SLE patients without autoimmune comorbidities reported fewer major and minor AEs compared to SLE patients with other concurrent rAIDs (Table 3). People with SLE and mental health disorders reported a higher frequency of severe rash [OR 3.2 (1.3–7.5)] and headache [OR 2.2 (1.3–3.6)].
Conclusion: COVID-19 vaccination is linked with increased risks of delayed AEs in patients with SLE compared to HC. Co-existence of rAIDs other than SLE yielded an increased risk for delayed AEs. By contrast, the degree of disease activity did not appear to have an impact.
M. Fathima: None; B. Doskaliuk: None; J. Lindblom: None; E. Nikiphorou: AbbVie/Abbott, 6, Celltrion, 6, Eli Lilly, 6, fresenius, 6, Galapagos, 6, Gilead, 1, 6, Pfizer, 6, Sanofi, 6; C. Wincup: None; S. Saha: None; S. Shaharir: None; W. Katchamart: None; P. Akarawatcharangura Goo: None; L. Traboco: None; Y. Chen: None; K. Jagtap: None; J. Lilleker: None; A. Nune: None; J. Pauling: AstraZeneca, 2, Boehringer-Ingelheim, 2, IsoMab, 2, Janssen, 2, 6, Permeatus, 2, Sojournix Pharma, 2; V. Agarwal: None; D. Dzifa: Roche, 6; C. TORO GUTIERREZ: AbbVie/Abbott, 6, Boehringer-Ingelheim, 6, Janssen, 6; C. CABALLERO: None; H. Chinoy: AstraZeneca, 1, Biogen, 2, Eli Lilly, 5, GlaxoSmithKlein(GSK), 6, Novartis, 2, Orphazyme, 2, Pfizer, 1, UCB, 6; R. Aggarwal: Actigraph, 2, Alexion, 2, ANI Pharmaceuticals, 2, Argenx, 2, AstraZeneca, 2, Boehringer-Ingelheim, 2, 5, Bristol-Myers Squibb(BMS), 2, 5, CabalettaBio, 2, Capella Bioscience, 2, Corbus, 2, CSL Behring, 2, EMD Serono, 2, 5, Galapagos, 2, Horizon Therapeutics, 2, I-Cell, 2, Janssen, 2, 5, Kezar, 2, Kyverna, 2, Mallinckrodt, 5, Merck, 2, Octapharma, 2, Pfizer, 2, 5, Q32, 5, Roivant, 2, Sanofi, 2, Teva, 2; V. Agarwal: None; L. Gupta: None; I. Parodis: Amgen, 5, 6, AstraZeneca, 5, 6, Aurinia Pharmaceuticals, 5, 6, Bristol-Myers Squibb(BMS), 5, 6, Elli Lilly and Company, 5, 6, F. Hoffmann-La Roche AG, 5, 6, Gilead Sciences, 5, 6, GSK, 5, 6, Janssen Pharmaceuticals, 5, 6, Novartis, 5, 6, Otsuka Pharmaceutical, 5, 6.
Background/Purpose: COVID-19 vaccines have been proven to be safe in healthy populations. However, data on delayed adverse effects (AEs) in people with autoimmune diseases (AIDs), including SLE has been lacking.
Methods: COVID-19 vaccination-related AEs reported longer than 7 days post-vaccination were assessed in the “COVID-19 Vaccination in Autoimmune Diseases 2” (COVAD-2) study in patients with SLE, rheumatic autoimmune diseases other than SLE (rAIDs), non-rheumatic autoimmune diseases (nrAIRDs), and healthy controls (HC). rAIDs included connective tissue diseases, inflammatory myopathies, and inflammatory arthritis, yet not SLE, whereas nrAIDs included inflammatory bowel disease and multiple sclerosis, among others. The COVAD-2 study comprised 157 collaborators across 106 countries and was conducted between February and June 2022. An online survey captured self-reported data related to COVID-19 vaccination-associated AEs in SLE, AIDs, and HC. We compared COVID-19 vaccination-related delayed AEs between groups using multivariable binary regression adjusting for age, gender, and ethnicity.
Results: Among 7203 participants, 882 (12.2%) SLE, 3161 (43.9%) rAIDs, 426 (5.9%) nrAIDs, and 2734 (38%) HC were included from a total of 10 783 respondents, with 74% female and 42.2% Caucasian (Table 1). People with SLE were young and middle-aged adults [median age 39 (31–50); rAIDs, 52 (41–62); nrAIDs 43 (34–53); HC 38 (30–49)].
When compared to HC, people with SLE reported higher overall major [OR 1.6 (1.2–2.0)] and minor AEs (Table 2). SLE patients when compared to rAIDS reported more frequent episodes of severe rashes [OR 2.4 (1.3–4.2)], while compared to nrAIDs, SLE individuals reported an increased number of several minor AEs (joint pain [OR 2.4 (1.3–4.3)], fatigue [OR 2.0 (1.2–3.2)], body ache [OR 2.1 (1.2–3.7)]), and hospitalization [OR 2.3 (1.1–4.9)].
Vaccination with ChadOx1 nCOV-19 (Oxford/ AstraZeneca) in SLE patients led to a higher frequency in the incidence of diarrhea compared to other vaccines [OR 2.6 (1.1 – 6.2)], whereas the Moderna vaccine was associated with increased hospitalization frequencies [OR 3.0 (1.6–5.6)].
Patients with active versus non-active SLE reported similar frequencies of COVID-19 vaccine-related delayed minor AEs [17.5% versus 17.6%; OR 1.1 (0.8–1.6)]. SLE patients without autoimmune comorbidities reported fewer major and minor AEs compared to SLE patients with other concurrent rAIDs (Table 3). People with SLE and mental health disorders reported a higher frequency of severe rash [OR 3.2 (1.3–7.5)] and headache [OR 2.2 (1.3–3.6)].
Conclusion: COVID-19 vaccination is linked with increased risks of delayed AEs in patients with SLE compared to HC. Co-existence of rAIDs other than SLE yielded an increased risk for delayed AEs. By contrast, the degree of disease activity did not appear to have an impact.

Table 1: Socio-demographic and vaccination data of survey respondents. HC: health controls; SLE: Systemic Lupus Erythematosus; nrAID: nonrheumatic autoimmune disease; rAID: rheumatic autoimmune disease.

Table 2: Effects of COVID-19 vaccination in patients with Systemic Lupus Erythematosus (SLE) vs healthy controls (HCs). Factors included as covariates in multivariable binary logistic regression analysis included age, sex, and ethnicity.

Table 3: Effects of COVID-19 vaccination in patients with Systemic Lupus Erythematosus (SLE) and no autoimmune comorbidities vs SLE with rheumatic autoimmune disease (rAIDs). Factors included as covariates in multivariable binary logistic regression analysis included age, sex, and ethnicity.
M. Fathima: None; B. Doskaliuk: None; J. Lindblom: None; E. Nikiphorou: AbbVie/Abbott, 6, Celltrion, 6, Eli Lilly, 6, fresenius, 6, Galapagos, 6, Gilead, 1, 6, Pfizer, 6, Sanofi, 6; C. Wincup: None; S. Saha: None; S. Shaharir: None; W. Katchamart: None; P. Akarawatcharangura Goo: None; L. Traboco: None; Y. Chen: None; K. Jagtap: None; J. Lilleker: None; A. Nune: None; J. Pauling: AstraZeneca, 2, Boehringer-Ingelheim, 2, IsoMab, 2, Janssen, 2, 6, Permeatus, 2, Sojournix Pharma, 2; V. Agarwal: None; D. Dzifa: Roche, 6; C. TORO GUTIERREZ: AbbVie/Abbott, 6, Boehringer-Ingelheim, 6, Janssen, 6; C. CABALLERO: None; H. Chinoy: AstraZeneca, 1, Biogen, 2, Eli Lilly, 5, GlaxoSmithKlein(GSK), 6, Novartis, 2, Orphazyme, 2, Pfizer, 1, UCB, 6; R. Aggarwal: Actigraph, 2, Alexion, 2, ANI Pharmaceuticals, 2, Argenx, 2, AstraZeneca, 2, Boehringer-Ingelheim, 2, 5, Bristol-Myers Squibb(BMS), 2, 5, CabalettaBio, 2, Capella Bioscience, 2, Corbus, 2, CSL Behring, 2, EMD Serono, 2, 5, Galapagos, 2, Horizon Therapeutics, 2, I-Cell, 2, Janssen, 2, 5, Kezar, 2, Kyverna, 2, Mallinckrodt, 5, Merck, 2, Octapharma, 2, Pfizer, 2, 5, Q32, 5, Roivant, 2, Sanofi, 2, Teva, 2; V. Agarwal: None; L. Gupta: None; I. Parodis: Amgen, 5, 6, AstraZeneca, 5, 6, Aurinia Pharmaceuticals, 5, 6, Bristol-Myers Squibb(BMS), 5, 6, Elli Lilly and Company, 5, 6, F. Hoffmann-La Roche AG, 5, 6, Gilead Sciences, 5, 6, GSK, 5, 6, Janssen Pharmaceuticals, 5, 6, Novartis, 5, 6, Otsuka Pharmaceutical, 5, 6.



