Poster Session A
Infection-related rheumatic syndromes
Session: (0196–0228) Infection-related Rheumatic Disease Poster
0207: Role of Pneumocystis Jirovecii Pneumonia Prophylaxis with Trimethoprim-Sulfamethoxazole Among Patients with Autoimmune Inflammatory Diseases Receiving High-Dose Glucocorticoids: A Systematic Review and Meta-Analysis
Sunday, November 12, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- AV
Athanasios Vassilopoulos, MD
Warren Alpert Medical School of Brown University
Providence, RI, United StatesDisclosure information not submitted.
Abstract Poster Presenter(s)
Athanasios Vassilopoulos1, Stephanos Vassilopoulos1, Fadi Shehadeh2, Markos Kalligeros1 and Eleftherios Mylonakis3, 1Division of Internal Medicine, Warren Alpert Medical School of Brown University, Rhode Island Hospital, Providence, RI, 2Houston Methodist Research Institute, Houston, TX, 3Department of Medicine, Houston Methodist Hospital, Houston, TX
Background/Purpose: Available immunosuppressive treatments for autoimmune inflammatory rheumatic diseases (AIRD) might increase the risk for opportunistic infections, such as Pneumocystis jirovecii pneumonia (PJP). Prophylactic trimethoprim-sulfamethoxazole (TMP/SMX) is often prescribed to prevent the occurrence of PJP among patients with AIRD. We calculate the incidence of PJP, PJP-related mortality, and TMP/SMX-related adverse events among patients with AIRD who received a prednisolone dose equivalent of ≥20 mg/day.
Methods: We searched PubMed and EMBASE for multiple-arm studies evaluating the efficacy and safety of TMP/SMX prophylaxis in individuals with AIRD. We stratified different prophylactic dose regimens of TMP/SMX in two groups: a "low-dose" group if the weekly dosage was lower than the equivalent of 400/80 mg/day, and a "standard-dose" group if the weekly dosage was equal or higher than the equivalent of 400/80 mg/day.
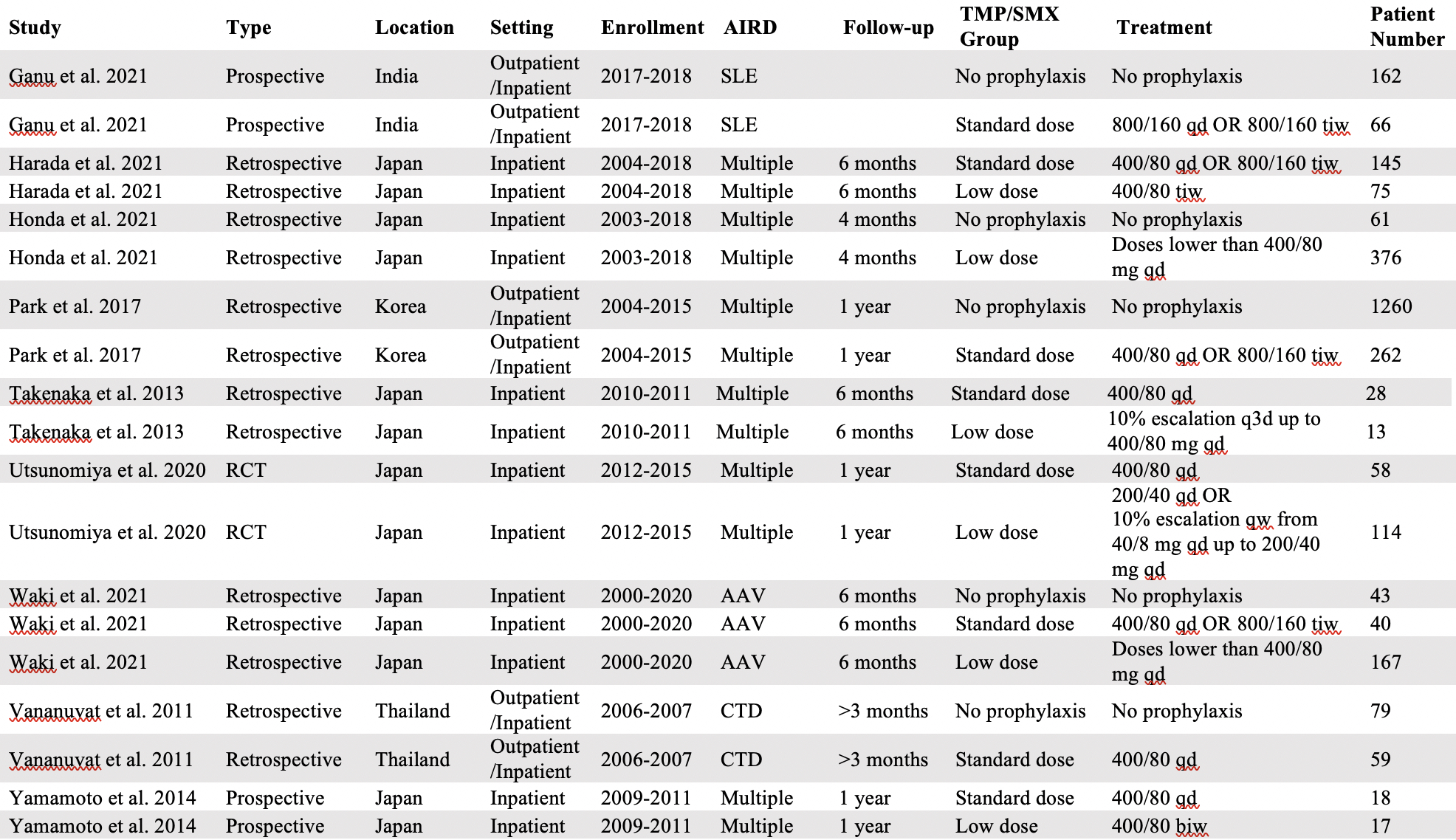
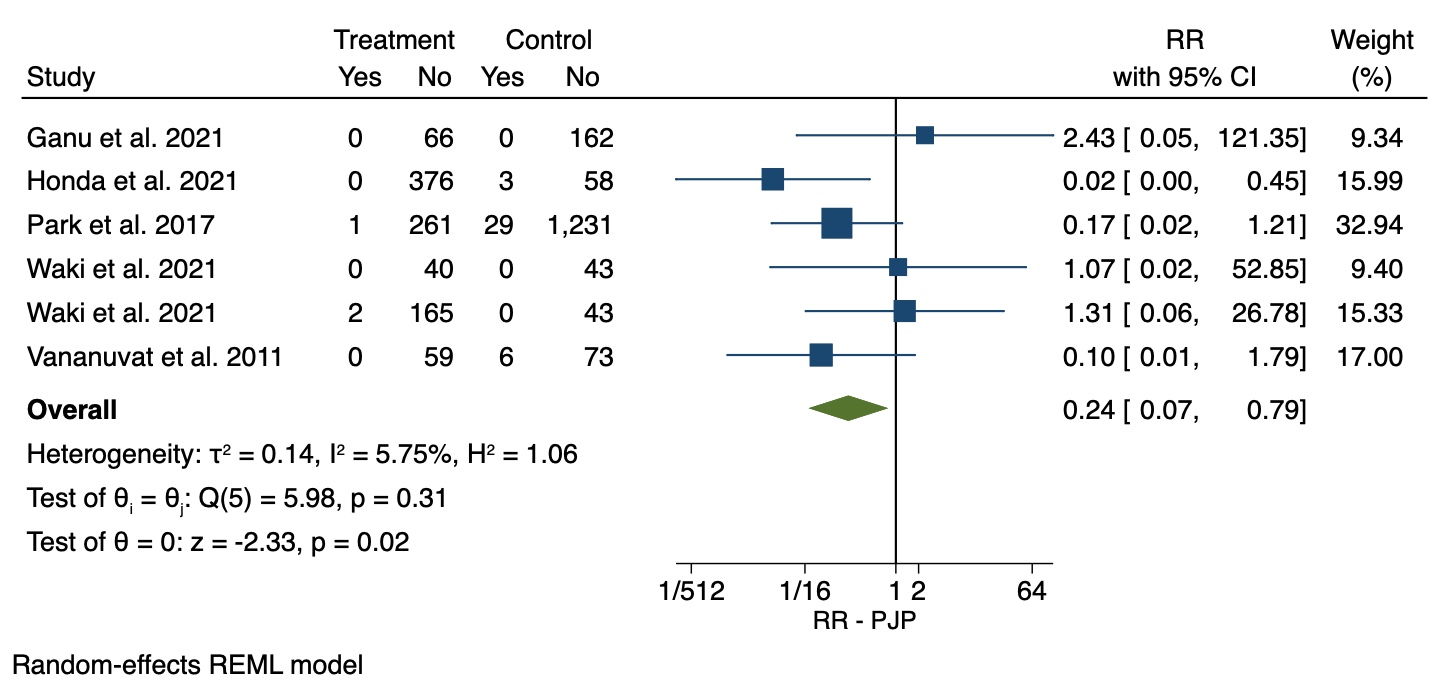
Results: Nine studies, performed in Japan, Korea, Thailand, and India, provided data on 1,438 individuals with AIRD receiving TMP/SMX and 1,605 individuals with AIRD not receiving prophylaxis. Studies were published between 2013 and 2021 and participants were followed from 3 to 12 months (77.75% for more than 6 months). At least one TMP/SMX treatment arm (i.e., "standard-dose", "low-dose" or both) was present in any included study. Specifically, eight studies included participants grouped in our "standard-dose" group, 6 studies included participants grouped in our "low-dose" group and 5 studies included participants that received no prophylaxis. The relative risk of PJP was 76% lower (Relative Risk 0.24; 95% CI: 0.07-0.79) among participants who received prophylactic TMP/SMX at any dose, compared to patients who did not receive any prophylaxis. The incidence of PJP for the initial 3-12 months of high-dose glucocorticoid treatment among patients who received no prophylaxis (2.95%, 95% CI: 0.65-6.46%) was significantly higher compared to the incidence of PJP among patients who received "standard-dose" (0.00%, 95% CI: 0.00-0.37%, p-value=0.009) or "low-dose" (0.00%, 95% CI: 0.00-0.04%, p-value=0.003) TMP/SMX. PJP-related mortality was 0.62% (95% CI: 0.00-1.98%) among individuals who received no prophylaxis and 0.00% among patients who received "standard-dose" (95% CI: 0.00-0.06%, p-value=0.073) or "low-dose" TMP/SMX (95% CI: 0.00-0.00%, p-value=0.038). We found the incidence of any TMP/SMX-related adverse event was 23.59% (95% CI: 9.77-40.88%) among patients who received "standard-dose" TMP/SMX and 24.06% (95% CI: 11.90-38.63%) among patients who received "low-dose" TMP/SMX, a non-significant difference (p-value=0.946).
Conclusion: Among individuals with AIRD who were included in studies evaluating the use of prophylactic TMP/SMX, the risk for PJP was lower among participants who received TMP/SMX compared to participants who did not receive prophylaxis. The rate of TMP/SMX-related adverse events did not differ among the low-dose and standard-dose TMP/SMX subgroups. Clinical trials evaluating the efficacy and safety of different prophylactic TMP/SMX dose regimens with long term follow-up are needed to study TMP/SMX utility for preventing PJP among patients with AIRD.
A. Vassilopoulos: None; S. Vassilopoulos: None; F. Shehadeh: None; M. Kalligeros: None; E. Mylonakis: BARDA, 5, Basilea, 1, Chemic Labs/KODA Therapeutics, 5, Cidara, 5, Leidos Biomedical Research Inc./NCI, 5, NIH/NIAID, 5, NIH/NIGMS, 5, Pfizer, 5, Regeneron Pharmaceuticals, Inc., 5, SciClone Pharmaceuticals, 5.
Background/Purpose: Available immunosuppressive treatments for autoimmune inflammatory rheumatic diseases (AIRD) might increase the risk for opportunistic infections, such as Pneumocystis jirovecii pneumonia (PJP). Prophylactic trimethoprim-sulfamethoxazole (TMP/SMX) is often prescribed to prevent the occurrence of PJP among patients with AIRD. We calculate the incidence of PJP, PJP-related mortality, and TMP/SMX-related adverse events among patients with AIRD who received a prednisolone dose equivalent of ≥20 mg/day.
Methods: We searched PubMed and EMBASE for multiple-arm studies evaluating the efficacy and safety of TMP/SMX prophylaxis in individuals with AIRD. We stratified different prophylactic dose regimens of TMP/SMX in two groups: a "low-dose" group if the weekly dosage was lower than the equivalent of 400/80 mg/day, and a "standard-dose" group if the weekly dosage was equal or higher than the equivalent of 400/80 mg/day.
Results: Nine studies, performed in Japan, Korea, Thailand, and India, provided data on 1,438 individuals with AIRD receiving TMP/SMX and 1,605 individuals with AIRD not receiving prophylaxis. Studies were published between 2013 and 2021 and participants were followed from 3 to 12 months (77.75% for more than 6 months). At least one TMP/SMX treatment arm (i.e., "standard-dose", "low-dose" or both) was present in any included study. Specifically, eight studies included participants grouped in our "standard-dose" group, 6 studies included participants grouped in our "low-dose" group and 5 studies included participants that received no prophylaxis. The relative risk of PJP was 76% lower (Relative Risk 0.24; 95% CI: 0.07-0.79) among participants who received prophylactic TMP/SMX at any dose, compared to patients who did not receive any prophylaxis. The incidence of PJP for the initial 3-12 months of high-dose glucocorticoid treatment among patients who received no prophylaxis (2.95%, 95% CI: 0.65-6.46%) was significantly higher compared to the incidence of PJP among patients who received "standard-dose" (0.00%, 95% CI: 0.00-0.37%, p-value=0.009) or "low-dose" (0.00%, 95% CI: 0.00-0.04%, p-value=0.003) TMP/SMX. PJP-related mortality was 0.62% (95% CI: 0.00-1.98%) among individuals who received no prophylaxis and 0.00% among patients who received "standard-dose" (95% CI: 0.00-0.06%, p-value=0.073) or "low-dose" TMP/SMX (95% CI: 0.00-0.00%, p-value=0.038). We found the incidence of any TMP/SMX-related adverse event was 23.59% (95% CI: 9.77-40.88%) among patients who received "standard-dose" TMP/SMX and 24.06% (95% CI: 11.90-38.63%) among patients who received "low-dose" TMP/SMX, a non-significant difference (p-value=0.946).
Conclusion: Among individuals with AIRD who were included in studies evaluating the use of prophylactic TMP/SMX, the risk for PJP was lower among participants who received TMP/SMX compared to participants who did not receive prophylaxis. The rate of TMP/SMX-related adverse events did not differ among the low-dose and standard-dose TMP/SMX subgroups. Clinical trials evaluating the efficacy and safety of different prophylactic TMP/SMX dose regimens with long term follow-up are needed to study TMP/SMX utility for preventing PJP among patients with AIRD.

Table 1 Baseline characteristics of included studies

Figure 1 Relative risk of Pneumocystis jirovecii pneumonia among patients receiving trimethoprim-sulfamethoxazole prophylaxis compared to patients not receiving prophylaxis.
A. Vassilopoulos: None; S. Vassilopoulos: None; F. Shehadeh: None; M. Kalligeros: None; E. Mylonakis: BARDA, 5, Basilea, 1, Chemic Labs/KODA Therapeutics, 5, Cidara, 5, Leidos Biomedical Research Inc./NCI, 5, NIH/NIAID, 5, NIH/NIGMS, 5, Pfizer, 5, Regeneron Pharmaceuticals, Inc., 5, SciClone Pharmaceuticals, 5.



