Poster Session A
Fibrosing rheumatic diseases (scleroderma, MCTD, IgG4-related disease, scleroderma mimics)
Session: (0609–0672) Systemic Sclerosis & Related Disorders – Clinical Poster I: Research
0664: Quantitative Hand Held Dynamometry to Assess Muscle Strength in Scleroderma Associated Myopathy
Sunday, November 12, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- AN
Anshula Nallapati, BS
Johns Hopkins University
Baltimore, MD, United StatesDisclosure information not submitted.
Abstract Poster Presenter(s)
Julie Paik1, Christopher Mecoli2, Anshula Nallapati1, Ami Shah3, Fred Wigley4, Zsuzsanna McMahan5 and Laura Hummers1, 1Johns Hopkins University, Baltimore, MD, 2Johns Hopkins University School of Medicine, Baltimore, MD, 3Department of Medicine, Division of Rheumatology, Johns Hopkins University School of Medicine, Ellicott City, MD, 4Department of Rheumatology, Johns Hopkins Medicine, Baltimore, MD, 5Johns Hopkins Rheumatology, Lutherville, MD
Background/Purpose: Manual muscle strength testing can be challenging in SSc associated myopathy (SSc-AM) due to contractures or skin tightening. Since there is currently no validated strength assessment in SSc, the purpose of this study is to investigate the reliability and validity of hand held dynamometry (HHD) in SSc-AM.
Methods: This was a prospective, cross-sectional study of 36 patients with SSc-AM enrolled at a single-center from August 2021 to May 2022. Inclusion criteria required patients to fulfill the 2013 ACR/EULAR criteria for SSc, and have skeletal myopathy as defined by a history of proximal weakness and/or elevated creatine kinase (CK) with at least one abnormal objective test result such as myopathic findings on EMG, muscle edema on muscle MRI, or inflammation, necrosis, or fibrosis on muscle biopsy.
Quantitative muscle strength testing of 3 proximal muscle groups [neck flexors, iliopsoas (bilateral hip flexors), and deltoid (bilateral arm abductors)], was assessed using a Micro-FET2 HHD and compared to the validated Manual Muscle Testing of 8 muscles (MMT-8). Each patient underwent 3 serial measurements for each of the three proximal muscle groups. A second blinded investigator performed the same assessments on 7 patients to assess inter-rater reliability. Both investigators had prior training using HHD and MMT-8.
The final HHD score was an average of each proximal muscle group (with or without neck flexors). Intra-rater reliability was assessed with a Pearson correlation and the intraclass correlation coefficient (ICC). Inter-rater reliability was also assessed with ICC. To determine how well the HHD score correlated with MMT-8, Pearson correlation was used to quantify the magnitude of correlation.
Results: Thirty-six patients with SSc-AM were enrolled. The average age at enrollment was 52.5 ± 11.1 years and at SSc diagnosis was 44.2 ± 11.9 years. 75% (27 of 36) were female and 44% (16 of 36) were Black while 37% (17 of 36) were Caucasian. 63.8% had diffuse SSc with a maximum modified Rodnan skin score (mRSS) of 13.7 ± 10.1. Maximum CK was 311 ± 425 U/L and aldolase was 12.6 ± 8.9 U/L . Closest CK at time of enrollment was 165 ± 166 U/L. Available commercial autoantibody testing and Euroimmun SSc profile demonstrated that 15% were anti-Scl-70 positive, 15% anti-PM-Scl positive, 6.25% anti-centromere B positive, 15% anti-RNA Poly III positive, 22% anti-U3RNP positive, 5.5% positive for U1RNP.
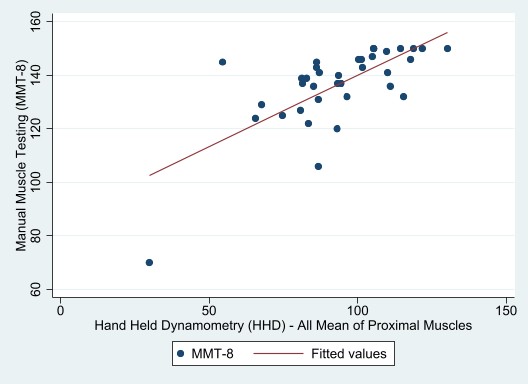
HHD strongly correlated with MMT-8 with neck flexors (r=0.70, p< 0.00001) and without neck flexors (r=0.68, p< 0.00001) (See Figure 1). Intra-rater reliability of HHD for the three consecutive assessments was also strong for the neck flexors and arm abductors with ICC of 0.96 (CI 0.94, 0.98) and hip flexors was ICC of 0.87 (CI 0.79,0.93), p=0.001. The inter-rater reliability of HHD between two examiners was good for the neck flexors [ICC of 0.87 (CI 0.23-0.97), p=0.01], arm abductors [ICC of 0.88 (CI 0.39-0.98), p=0.006] and hip flexors with [ICC of 0.73 (CI -0.56-0.95, p=0.07)].
Conclusion: HHD highly correlates with the validated MMT-8 in SSc-AM and demonstrates good intra-rater and inter-rater reliability, thereby making it a feasible alternative to quantify and measure muscle strength in scleroderma.
J. Paik: None; C. Mecoli: None; A. Nallapati: None; A. Shah: Arena Pharmaceuticals, 5, Eicos Sciences, 5, Kadmon Corporation, 5, Medpace LLC, 5; F. Wigley: None; Z. McMahan: Boehringer-Ingelheim, 12, medical writing support for a different manuscript; L. Hummers: AbbVie/Abbott, 1, Biotest, 2, Boehringer-Ingelheim, 1, 5, CSL Behring, 1, Cumberland Pharmaceuticals, 5, GlaxoSmithKlein(GSK), 5, Kadmon Corporation, 5, Medpace, 5, Mitsubishi Tanabe, 5, Prometheus, 5.
Background/Purpose: Manual muscle strength testing can be challenging in SSc associated myopathy (SSc-AM) due to contractures or skin tightening. Since there is currently no validated strength assessment in SSc, the purpose of this study is to investigate the reliability and validity of hand held dynamometry (HHD) in SSc-AM.
Methods: This was a prospective, cross-sectional study of 36 patients with SSc-AM enrolled at a single-center from August 2021 to May 2022. Inclusion criteria required patients to fulfill the 2013 ACR/EULAR criteria for SSc, and have skeletal myopathy as defined by a history of proximal weakness and/or elevated creatine kinase (CK) with at least one abnormal objective test result such as myopathic findings on EMG, muscle edema on muscle MRI, or inflammation, necrosis, or fibrosis on muscle biopsy.
Quantitative muscle strength testing of 3 proximal muscle groups [neck flexors, iliopsoas (bilateral hip flexors), and deltoid (bilateral arm abductors)], was assessed using a Micro-FET2 HHD and compared to the validated Manual Muscle Testing of 8 muscles (MMT-8). Each patient underwent 3 serial measurements for each of the three proximal muscle groups. A second blinded investigator performed the same assessments on 7 patients to assess inter-rater reliability. Both investigators had prior training using HHD and MMT-8.
The final HHD score was an average of each proximal muscle group (with or without neck flexors). Intra-rater reliability was assessed with a Pearson correlation and the intraclass correlation coefficient (ICC). Inter-rater reliability was also assessed with ICC. To determine how well the HHD score correlated with MMT-8, Pearson correlation was used to quantify the magnitude of correlation.
Results: Thirty-six patients with SSc-AM were enrolled. The average age at enrollment was 52.5 ± 11.1 years and at SSc diagnosis was 44.2 ± 11.9 years. 75% (27 of 36) were female and 44% (16 of 36) were Black while 37% (17 of 36) were Caucasian. 63.8% had diffuse SSc with a maximum modified Rodnan skin score (mRSS) of 13.7 ± 10.1. Maximum CK was 311 ± 425 U/L and aldolase was 12.6 ± 8.9 U/L . Closest CK at time of enrollment was 165 ± 166 U/L. Available commercial autoantibody testing and Euroimmun SSc profile demonstrated that 15% were anti-Scl-70 positive, 15% anti-PM-Scl positive, 6.25% anti-centromere B positive, 15% anti-RNA Poly III positive, 22% anti-U3RNP positive, 5.5% positive for U1RNP.
HHD strongly correlated with MMT-8 with neck flexors (r=0.70, p< 0.00001) and without neck flexors (r=0.68, p< 0.00001) (See Figure 1). Intra-rater reliability of HHD for the three consecutive assessments was also strong for the neck flexors and arm abductors with ICC of 0.96 (CI 0.94, 0.98) and hip flexors was ICC of 0.87 (CI 0.79,0.93), p=0.001. The inter-rater reliability of HHD between two examiners was good for the neck flexors [ICC of 0.87 (CI 0.23-0.97), p=0.01], arm abductors [ICC of 0.88 (CI 0.39-0.98), p=0.006] and hip flexors with [ICC of 0.73 (CI -0.56-0.95, p=0.07)].
Conclusion: HHD highly correlates with the validated MMT-8 in SSc-AM and demonstrates good intra-rater and inter-rater reliability, thereby making it a feasible alternative to quantify and measure muscle strength in scleroderma.

J. Paik: None; C. Mecoli: None; A. Nallapati: None; A. Shah: Arena Pharmaceuticals, 5, Eicos Sciences, 5, Kadmon Corporation, 5, Medpace LLC, 5; F. Wigley: None; Z. McMahan: Boehringer-Ingelheim, 12, medical writing support for a different manuscript; L. Hummers: AbbVie/Abbott, 1, Biotest, 2, Boehringer-Ingelheim, 1, 5, CSL Behring, 1, Cumberland Pharmaceuticals, 5, GlaxoSmithKlein(GSK), 5, Kadmon Corporation, 5, Medpace, 5, Mitsubishi Tanabe, 5, Prometheus, 5.



