Poster Session A
Fibrosing rheumatic diseases (scleroderma, MCTD, IgG4-related disease, scleroderma mimics)
Session: (0609–0672) Systemic Sclerosis & Related Disorders – Clinical Poster I: Research
0644: Prediction of Stable SSc-ILD Depends on Definition of ILD Progression
Sunday, November 12, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- AH
Anna-Maria Hoffmann-Vold, MD, PhD
Oslo University Hospital
Oslo, NorwayDisclosure information not submitted.
Abstract Poster Presenter(s)
Anna-Maria Hoffmann-Vold1, Liubov Petelytska2, Havard Fretheim1, Trond Mogens Aaløkken3, Mike Becker4, Cathrine Brunborg5, Cosimo Bruni6, Christian Clarenbach7, Phuong Phuong Diep8, Rucsandra Dobrota6, Michael Durheim9, Muriel Elhai10, Thomas Frauenfelder11, Suzana Jordan6, Emily Langballe12, Carina Mihai6, Oyvind Midtvedt1, Oyvind Molberg13 and Oliver Distler6, 1Oslo University Hospital, Oslo, Norway, 2Bogomolets National Medical University, Kyiv, Ukraine, 3Dept of Radiology, Oslo University Hospital, Oslo, Norway, 4Department of Rheumatology, University Hospital Zurich, University of Zurich, Zürich, Switzerland, 5Oslo Centre for Biostatistics and Epidemiology, Research Support Services, Oslo University Hospital - Rikshospitalet, Oslo, Norway, 6Department of Rheumatology, University Hospital Zurich, University of Zurich, Zurich, Switzerland, 7Dept of Pulmonology, University Hospital Zurich, University of Zurich, Zurich, Switzerland, 8Dept of Respiratory diseases, University Hospital Oslo, Oslo, Norway, 9Dept of Respiratory diseases, Oslo University Hospital, Oslo, Norway, 10University Hospital zurich, University of Zurich, Zurich, Switzerland, 11Dept of Radiology, University Hospital Zurich, University of Zurich, Zurich, Switzerland, 12Universitetet i Oslo, Oslo, Norway, 13Dept of Rheumatology, University Hospital Oslo, Oslo, Norway
Background/Purpose: Progression of interstitial lung disease (ILD) reduces long-term survival in patients with systemic sclerosis (SSc), and aggressive treatment and tight monitoring should be considered. Conversely, identifying stable SSc-ILD patients over time is important in clinical practice to avoid overtreatment and facilitate inclusion into clinical trials. The objective was to determine predictive factors for stable ILD in SSc.
Methods: We included all SSc patients who had ILD on HRCT from two expert SSc centers with well characterized SSc cohorts. Consecutive annual lung function tests including forced vital capacity (FVC) and diffusing capacity for carbon monoxide (DLCO), and comprehensive serial clinical and imaging assessments were evaluated. Patients were defined as long-term stable ILD if no progression was observed over three years, using the following definitions for ILD progression:
(A) FVC decline ≥5% over 12 months
(B) 2022 ATS/ERS/JRS/ALAT guideline progressive pulmonary fibrosis (PPF) criteria with (1) worsening of respiratory symptoms; (2) absolute decline in FVC ≥5% or in DLCO≥10% and (3) disease progression on HRCT over 12 months
(C) INBUILD progressive fibrosing ILD (PF-ILD) criteria with (1) relative FVC decline ≥10%, (2) relative FVC decline ≥5-< 10% and worsening of respiratory symptoms or an increased extent of fibrosis on HRCT, or (3) worsening of respiratory symptoms and an increased extent of fibrosis within 24 months.
Multivariable logistic regression was applied, adjusting for known risk factors for ILD progression, including treatment, to identify predictors of stable ILD.
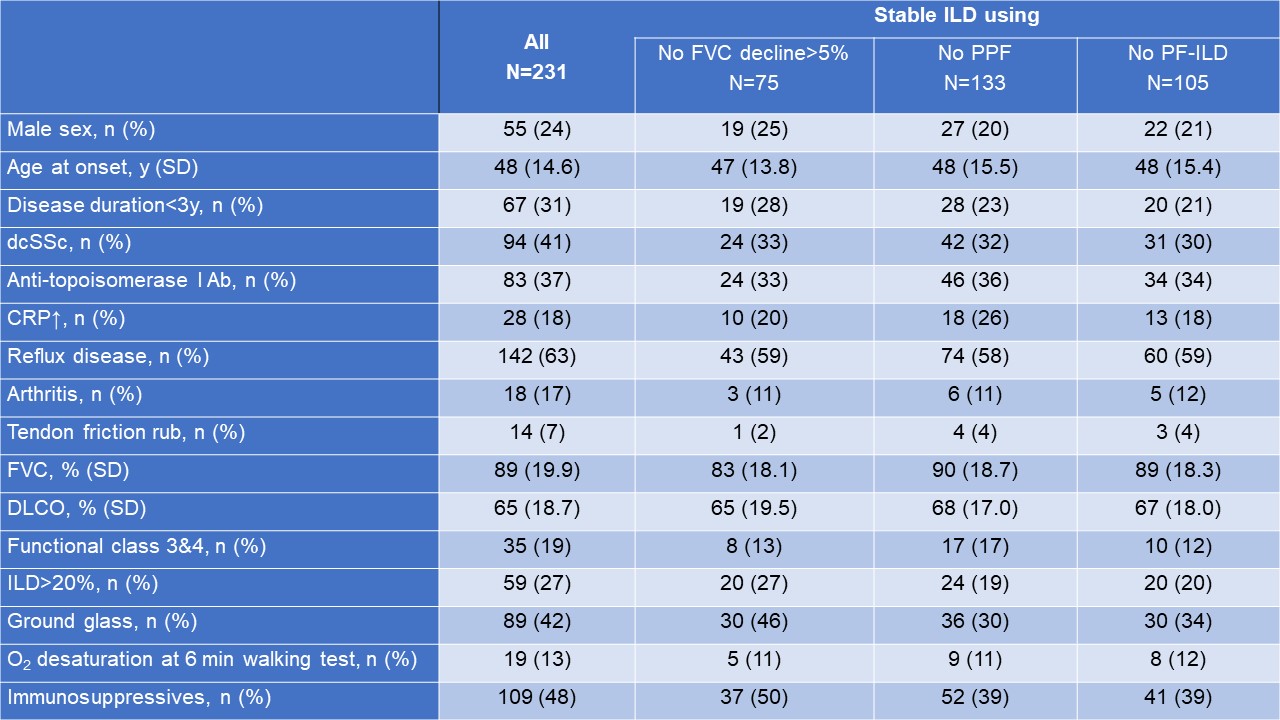
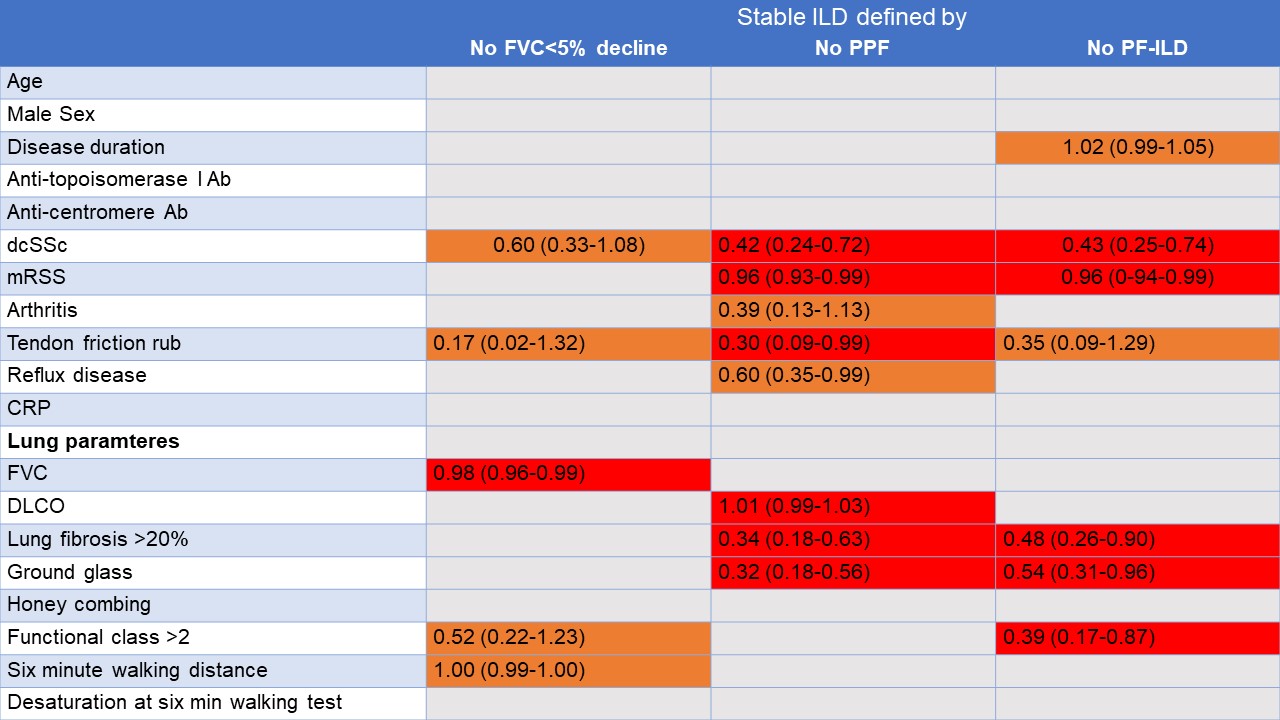
Results: In total, 231 SSc-ILD patients were included (Table 1). We identified 75 (32%) patients with stable ILD over mean three years defined by no FVC decline≥5%, 133 (58%) defined by no PPF guideline criteria and 105 (45%) by no INBUILD PF-ILD criteria. Factors predicting long-term stable ILD varied in univariable logistic regression depending on which definition was applied and no consistent factors could be identified (Table 2). Multivariable logistic regression models also varied based on the applied definition. Stable ILD as defined by no FVC >5% decline was predicted by lower baseline FVC (Figure 1A). Stable ILD defined by no PPF criteria and no PF-ILD criteria was significantly predicted by the absence of dcSSc and of ground glass opacities on HRCT (Figure 1B and C).
Conclusion: Long-term stable ILD in SSc occurs but varies based on which definition is applied. Prediction of stability is challenging, highlighting the necessity of comprehensive disease assessment and monitoring.

A. Hoffmann-Vold: Arxx Therapeutics, 2, Boehringer-Ingelheim, 2, 5, 6, 12, Support for travel, Genentech, 2, Janssen, 2, 5, 6, Medscape, 2, 6, 12, Support for travel, Roche, 2, 6, 12, Support for travel; L. Petelytska: None; H. Fretheim: actelion, 5, bayer, 2, Boehringer-Ingelheim, 6, GlaxoSmithKlein(GSK), 5; T. Aaløkken: Boehringer-Ingelheim, 6; M. Becker: Amgen, 6, Bayer, 6, GSK, 6, Mepha, 6, MSD, 6, Novartis, 6, Vifor, 6; C. Brunborg: None; C. Bruni: AbbVie/Abbott, 5, Boehringer-Ingelheim, 2, 12, Travel Support, Eli Lilly, 6; C. Clarenbach: AstraZeneca, 1, 6, Boehringer-Ingelheim, 1, 6, CSL Behring, 1, 6, Daiichi Sankyo, 1, GlaxoSmithKlein(GSK), 1, 6, Grifols, 1, 6, Merck/MSD, 1, OM Pharma, 1, 6, Sanofi, 1, 6, Vifor, 1, 6; P. Diep: Boehringer-Ingelheim, 6, Roche, 6; R. Dobrota: Actelion, 5, 6, Amgen, 5, Articulum Fellowship, sponsored by Pfizer, 5, Boehringer-Ingelheim, 6; M. Durheim: Boehringer-Ingelheim, 2, 5, 6, Roche, 6; M. Elhai: AstraZeneca, 12, Travel to Congress support, Janssen, 12, Congress support; T. Frauenfelder: Boehringer-Ingelheim, 6; S. Jordan: None; E. Langballe: None; C. Mihai: Boehringer-Ingelheim, 2, 5, 6, Janssen, 2, MED Talks Switzerland, 2, Mepha, 2, PlayToKnow AG, 2; O. Midtvedt: None; O. Molberg: None; O. Distler: 4P-Pharma, 2, 5, 6, AbbVie, 2, 5, 6, Acceleron, 2, 5, 6, Alcimed, 2, 5, 6, Altavant Sciences, 2, 5, 6, Amgen, 2, 5, 6, AnaMar, 2, 5, 6, Arxx, 2, 5, 6, AstraZeneca, 2, 5, 6, Bayer, 2, 5, 6, Blade Therapeutics, 2, 5, 6, Boehringer Ingelheim, 2, 5, 6, Citus AG, 12, Co-Founder, Corbus Pharmaceuticals, 2, 5, 6, CSL Behring, 2, 5, 6, Galapagos, 2, 5, 6, Galderma, 2, 5, 6, Glenmark, 2, 5, 6, Gossamer, 2, 5, 6, Horizon Therapeutics, 2, 5, 6, Janssen, 2, 5, 6, Kymera, 2, 5, 6, Lupin, 2, 5, 6, Medscape, 2, 5, 6, Miltenyi Biotec, 2, 5, 6, Mitsubishi Tanabe, 2, 5, 6, MSD, 2, 5, 6, Novartis, 2, 5, 6, Patent issued “mir-29 for the treatment of systemic sclerosis” (US8247389, EP2331143), 10, Prometheus Biosciences, 2, 5, 6, Redx Pharma, 2, 5, 6, Roivant, 2, 5, 6, Topadur, 2, 5, 6.
Background/Purpose: Progression of interstitial lung disease (ILD) reduces long-term survival in patients with systemic sclerosis (SSc), and aggressive treatment and tight monitoring should be considered. Conversely, identifying stable SSc-ILD patients over time is important in clinical practice to avoid overtreatment and facilitate inclusion into clinical trials. The objective was to determine predictive factors for stable ILD in SSc.
Methods: We included all SSc patients who had ILD on HRCT from two expert SSc centers with well characterized SSc cohorts. Consecutive annual lung function tests including forced vital capacity (FVC) and diffusing capacity for carbon monoxide (DLCO), and comprehensive serial clinical and imaging assessments were evaluated. Patients were defined as long-term stable ILD if no progression was observed over three years, using the following definitions for ILD progression:
(A) FVC decline ≥5% over 12 months
(B) 2022 ATS/ERS/JRS/ALAT guideline progressive pulmonary fibrosis (PPF) criteria with (1) worsening of respiratory symptoms; (2) absolute decline in FVC ≥5% or in DLCO≥10% and (3) disease progression on HRCT over 12 months
(C) INBUILD progressive fibrosing ILD (PF-ILD) criteria with (1) relative FVC decline ≥10%, (2) relative FVC decline ≥5-< 10% and worsening of respiratory symptoms or an increased extent of fibrosis on HRCT, or (3) worsening of respiratory symptoms and an increased extent of fibrosis within 24 months.
Multivariable logistic regression was applied, adjusting for known risk factors for ILD progression, including treatment, to identify predictors of stable ILD.
Results: In total, 231 SSc-ILD patients were included (Table 1). We identified 75 (32%) patients with stable ILD over mean three years defined by no FVC decline≥5%, 133 (58%) defined by no PPF guideline criteria and 105 (45%) by no INBUILD PF-ILD criteria. Factors predicting long-term stable ILD varied in univariable logistic regression depending on which definition was applied and no consistent factors could be identified (Table 2). Multivariable logistic regression models also varied based on the applied definition. Stable ILD as defined by no FVC >5% decline was predicted by lower baseline FVC (Figure 1A). Stable ILD defined by no PPF criteria and no PF-ILD criteria was significantly predicted by the absence of dcSSc and of ground glass opacities on HRCT (Figure 1B and C).
Conclusion: Long-term stable ILD in SSc occurs but varies based on which definition is applied. Prediction of stability is challenging, highlighting the necessity of comprehensive disease assessment and monitoring.

Disease characteristics of stable patients not fulfilling the different definitions for ILD progression over an observation period of 3 years

Table 2: Variables predicting significantly (red) or numerically (orange) stable ILD using the competing definitions for ILD progression assessed with univariable logistic regression

Figure 1: Variables predicting stable ILD using (A) FVC ≥5% decline, (B) PPF and (C) PF-ILD assessed with multivariable logistic regression
A. Hoffmann-Vold: Arxx Therapeutics, 2, Boehringer-Ingelheim, 2, 5, 6, 12, Support for travel, Genentech, 2, Janssen, 2, 5, 6, Medscape, 2, 6, 12, Support for travel, Roche, 2, 6, 12, Support for travel; L. Petelytska: None; H. Fretheim: actelion, 5, bayer, 2, Boehringer-Ingelheim, 6, GlaxoSmithKlein(GSK), 5; T. Aaløkken: Boehringer-Ingelheim, 6; M. Becker: Amgen, 6, Bayer, 6, GSK, 6, Mepha, 6, MSD, 6, Novartis, 6, Vifor, 6; C. Brunborg: None; C. Bruni: AbbVie/Abbott, 5, Boehringer-Ingelheim, 2, 12, Travel Support, Eli Lilly, 6; C. Clarenbach: AstraZeneca, 1, 6, Boehringer-Ingelheim, 1, 6, CSL Behring, 1, 6, Daiichi Sankyo, 1, GlaxoSmithKlein(GSK), 1, 6, Grifols, 1, 6, Merck/MSD, 1, OM Pharma, 1, 6, Sanofi, 1, 6, Vifor, 1, 6; P. Diep: Boehringer-Ingelheim, 6, Roche, 6; R. Dobrota: Actelion, 5, 6, Amgen, 5, Articulum Fellowship, sponsored by Pfizer, 5, Boehringer-Ingelheim, 6; M. Durheim: Boehringer-Ingelheim, 2, 5, 6, Roche, 6; M. Elhai: AstraZeneca, 12, Travel to Congress support, Janssen, 12, Congress support; T. Frauenfelder: Boehringer-Ingelheim, 6; S. Jordan: None; E. Langballe: None; C. Mihai: Boehringer-Ingelheim, 2, 5, 6, Janssen, 2, MED Talks Switzerland, 2, Mepha, 2, PlayToKnow AG, 2; O. Midtvedt: None; O. Molberg: None; O. Distler: 4P-Pharma, 2, 5, 6, AbbVie, 2, 5, 6, Acceleron, 2, 5, 6, Alcimed, 2, 5, 6, Altavant Sciences, 2, 5, 6, Amgen, 2, 5, 6, AnaMar, 2, 5, 6, Arxx, 2, 5, 6, AstraZeneca, 2, 5, 6, Bayer, 2, 5, 6, Blade Therapeutics, 2, 5, 6, Boehringer Ingelheim, 2, 5, 6, Citus AG, 12, Co-Founder, Corbus Pharmaceuticals, 2, 5, 6, CSL Behring, 2, 5, 6, Galapagos, 2, 5, 6, Galderma, 2, 5, 6, Glenmark, 2, 5, 6, Gossamer, 2, 5, 6, Horizon Therapeutics, 2, 5, 6, Janssen, 2, 5, 6, Kymera, 2, 5, 6, Lupin, 2, 5, 6, Medscape, 2, 5, 6, Miltenyi Biotec, 2, 5, 6, Mitsubishi Tanabe, 2, 5, 6, MSD, 2, 5, 6, Novartis, 2, 5, 6, Patent issued “mir-29 for the treatment of systemic sclerosis” (US8247389, EP2331143), 10, Prometheus Biosciences, 2, 5, 6, Redx Pharma, 2, 5, 6, Roivant, 2, 5, 6, Topadur, 2, 5, 6.



