Poster Session A
Fibrosing rheumatic diseases (scleroderma, MCTD, IgG4-related disease, scleroderma mimics)
Session: (0609–0672) Systemic Sclerosis & Related Disorders – Clinical Poster I: Research
0643: Progression of Interstitial Lung Disease in Systemic Sclerosis Does Not Predict Further Progression
Sunday, November 12, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- AH
Anna-Maria Hoffmann-Vold, MD, PhD
Oslo University Hospital
Oslo, NorwayDisclosure information not submitted.
Abstract Poster Presenter(s)
Anna-Maria Hoffmann-Vold1, Liubov Petelytska2, Havard Fretheim1, Trond Mogens Aaløkken3, Mike Becker4, Hilde Jenssen Bjørkekjær5, Cathrine Brunborg6, Cosimo Bruni7, Christian Clarenbach8, Phuong Phuong Diep9, Rucsandra Dobrota7, Michael Durheim10, Muriel Elhai11, Thomas Frauenfelder12, Suzana Jordan7, Emily Langballe13, Oyvind Midtvedt1, Carina Mihai7, Oyvind Molberg14 and Oliver Distler7, 1Oslo University Hospital, Oslo, Norway, 2Bogomolets National Medical University, Kyiv, Ukraine, 3Dept of Radiology, Oslo University Hospital, Oslo, Norway, 4Department of Rheumatology, University Hospital Zurich, University of Zurich, Zürich, Switzerland, 5Department of Rheumatology, Hospital of Southern Norway, Kristiansand & University of Oslo, Institute of Clinical Medicine, Oslo, Norway, 6Oslo Centre for Biostatistics and Epidemiology, Research Support Services, Oslo University Hospital - Rikshospitalet, Oslo, Norway, 7Department of Rheumatology, University Hospital Zurich, University of Zurich, Zurich, Switzerland, 8Dept of Pulmonology, University Hospital Zurich, University of Zurich, Zurich, Switzerland, 9Dept of Respiratory diseases, University Hospital Oslo, Oslo, Norway, 10Dept of Respiratory diseases, Oslo University Hospital, Oslo, Norway, 11University Hospital zurich, University of Zurich, Zurich, Switzerland, 12Dept of Radiology, University Hospital Zurich, University of Zurich, Zurich, Switzerland, 13Universitetet i Oslo, Oslo, Norway, 14Dept of Rheumatology, University Hospital Oslo, Oslo, Norway
Background/Purpose: In clinical practice, we often wait for progression of interstitial lung disease (ILD) in systemic sclerosis (SSc) to initiate or escalate therapy. Similarly, progressive SSc-ILD patients are recruited into trials to enrich for further progression. These strategies assume that patients with recent ILD progression have a higher risk for further progression. Here, we assessed whether ILD progression predicts subsequent progression using four definitions of progressive disease.
Methods: We included all SSc patients from two expert SSc centers who had ILD on HRCT and consecutive annual forced vital capacity (FVC) measurements. For the primary analysis, ILD progression was defined as absolute FVC decline ≥5% over 12 months. Patients were grouped into progressors (FVC ≥5% decline) and non-progressors (all others). At the next annual follow up visit, all patients were again assessed for ILD progression. In secondary analyses, we applied other definitions of progression such as:
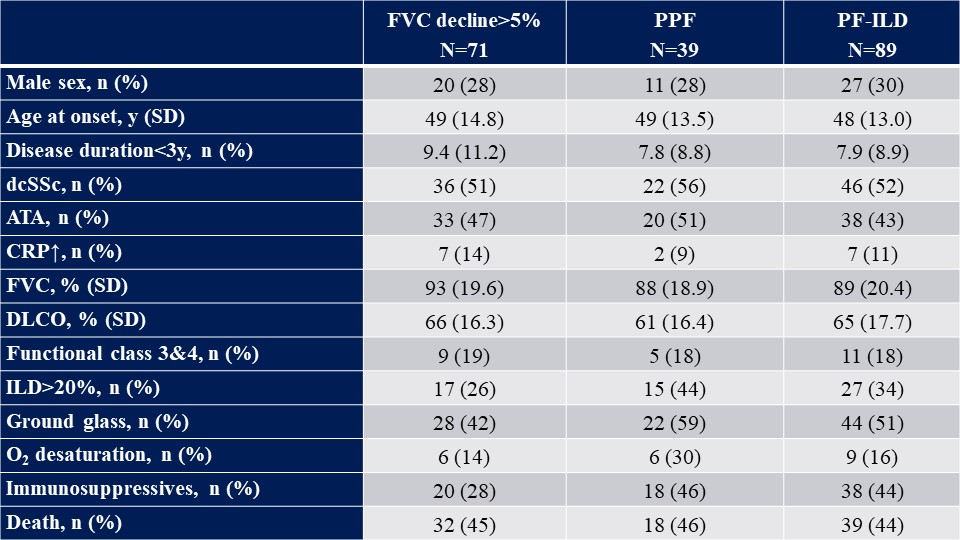
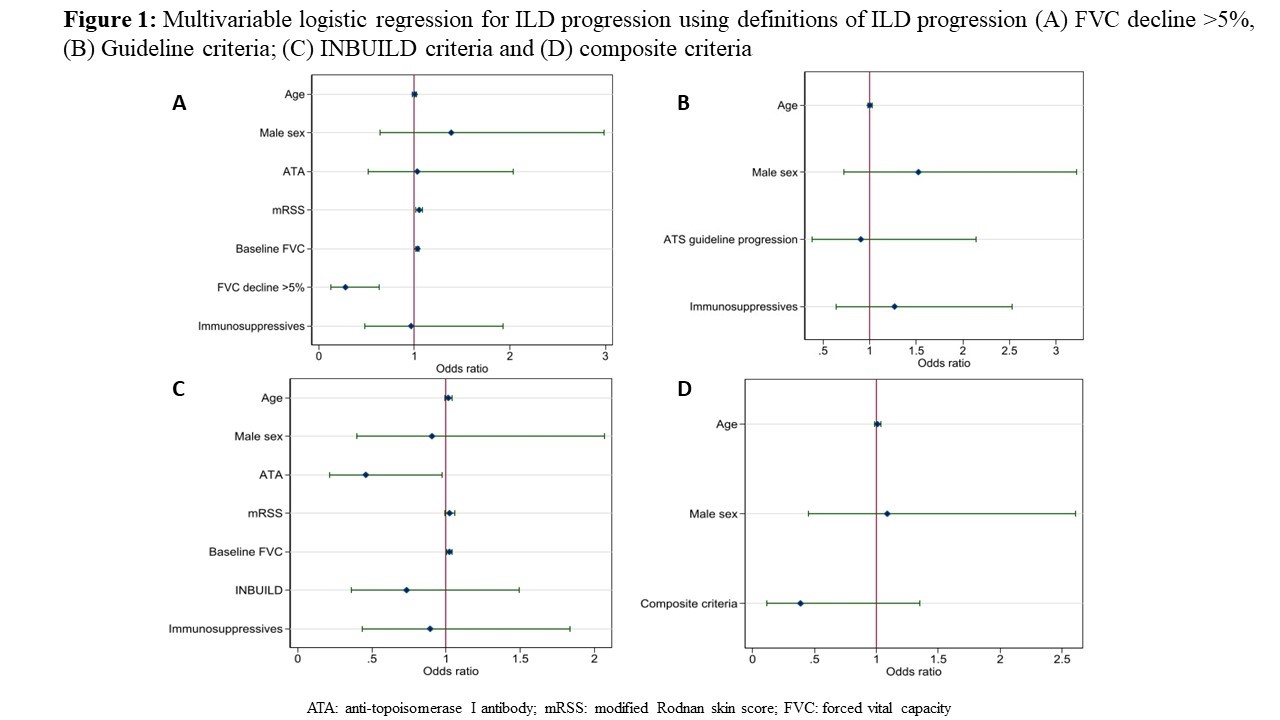
Results: In total, 231 SSc-ILD patients were included (Table 1). At 12 months, 71 (30.7%) showed FVC decline >5% and were classified as progressors. In multivariable logistic regression ILD progression was significantly protective for further progression at the next annual follow-up (Odds Ratio 0.28, 95%CI 0.12-0.63, p=0.002, Figure1a). When other definitions of progression were applied, similar results were obtained: 39 (19%) fulfilled the PPF guideline criteria, 89 (39%) the PF-ILD criteria and 33 (14%) the composite criteria. Multivariable regression analysis adjusted for age, sex and treatment (PPF), for age and sex (composite) and for age, sex, anti-Topoisomerase I antibody (ATA), modified Rodnan skin Score (mRSS), baseline FVC and treatment (PF-ILD) showed the same direction as the primary analysis (Figure1b-d).
Conclusion: SSc-ILD progression does not predict further progression using any definition of ILD progression. These results challenge current treatment practice, since waiting for progression to initiate or escalate treatment does not seem to be the adequate strategy.
A. Hoffmann-Vold: Arxx Therapeutics, 2, Boehringer-Ingelheim, 2, 5, 6, 12, Support for travel, Genentech, 2, Janssen, 2, 5, 6, Medscape, 2, 6, 12, Support for travel, Roche, 2, 6, 12, Support for travel; L. Petelytska: None; H. Fretheim: actelion, 5, bayer, 2, Boehringer-Ingelheim, 6, GlaxoSmithKlein(GSK), 5; T. Aaløkken: Boehringer-Ingelheim, 6; M. Becker: Amgen, 6, Bayer, 6, GSK, 6, Mepha, 6, MSD, 6, Novartis, 6, Vifor, 6; H. Jenssen Bjørkekjær: Janssen, 5; C. Brunborg: None; C. Bruni: AbbVie/Abbott, 5, Boehringer-Ingelheim, 2, 12, Travel Support, Eli Lilly, 6; C. Clarenbach: AstraZeneca, 1, 6, Boehringer-Ingelheim, 1, 6, CSL Behring, 1, 6, Daiichi Sankyo, 1, GlaxoSmithKlein(GSK), 1, 6, Grifols, 1, 6, Merck/MSD, 1, OM Pharma, 1, 6, Sanofi, 1, 6, Vifor, 1, 6; P. Diep: Boehringer-Ingelheim, 6, Roche, 6; R. Dobrota: Actelion, 5, 6, Amgen, 5, Articulum Fellowship, sponsored by Pfizer, 5, Boehringer-Ingelheim, 6; M. Durheim: Boehringer-Ingelheim, 2, 5, 6, Roche, 6; M. Elhai: AstraZeneca, 12, Travel to Congress support, Janssen, 12, Congress support; T. Frauenfelder: Boehringer-Ingelheim, 6; S. Jordan: None; E. Langballe: None; O. Midtvedt: None; C. Mihai: Boehringer-Ingelheim, 2, 5, 6, Janssen, 2, MED Talks Switzerland, 2, Mepha, 2, PlayToKnow AG, 2; O. Molberg: None; O. Distler: 4P-Pharma, 2, 5, 6, AbbVie, 2, 5, 6, Acceleron, 2, 5, 6, Alcimed, 2, 5, 6, Altavant Sciences, 2, 5, 6, Amgen, 2, 5, 6, AnaMar, 2, 5, 6, Arxx, 2, 5, 6, AstraZeneca, 2, 5, 6, Bayer, 2, 5, 6, Blade Therapeutics, 2, 5, 6, Boehringer Ingelheim, 2, 5, 6, Citus AG, 12, Co-Founder, Corbus Pharmaceuticals, 2, 5, 6, CSL Behring, 2, 5, 6, Galapagos, 2, 5, 6, Galderma, 2, 5, 6, Glenmark, 2, 5, 6, Gossamer, 2, 5, 6, Horizon Therapeutics, 2, 5, 6, Janssen, 2, 5, 6, Kymera, 2, 5, 6, Lupin, 2, 5, 6, Medscape, 2, 5, 6, Miltenyi Biotec, 2, 5, 6, Mitsubishi Tanabe, 2, 5, 6, MSD, 2, 5, 6, Novartis, 2, 5, 6, Patent issued “mir-29 for the treatment of systemic sclerosis” (US8247389, EP2331143), 10, Prometheus Biosciences, 2, 5, 6, Redx Pharma, 2, 5, 6, Roivant, 2, 5, 6, Topadur, 2, 5, 6.
Background/Purpose: In clinical practice, we often wait for progression of interstitial lung disease (ILD) in systemic sclerosis (SSc) to initiate or escalate therapy. Similarly, progressive SSc-ILD patients are recruited into trials to enrich for further progression. These strategies assume that patients with recent ILD progression have a higher risk for further progression. Here, we assessed whether ILD progression predicts subsequent progression using four definitions of progressive disease.
Methods: We included all SSc patients from two expert SSc centers who had ILD on HRCT and consecutive annual forced vital capacity (FVC) measurements. For the primary analysis, ILD progression was defined as absolute FVC decline ≥5% over 12 months. Patients were grouped into progressors (FVC ≥5% decline) and non-progressors (all others). At the next annual follow up visit, all patients were again assessed for ILD progression. In secondary analyses, we applied other definitions of progression such as:
- 2022 ATS/ERS/JRS/ALAT PPF guideline criteria with (1) worsening of respiratory symptoms; (2) absolute decline in FVC ≥5% or in DLCO ≥10% and (3) disease progression on HRCT over 12 months
- INBUILD PF-ILD criteria with (1) FVC decline ≥10%, (2) FVC decline >5%-< 10% and worsening of respiratory symptoms or an increased extent of fibrosis on HRCT, or (3) worsening of respiratory symptoms and an increased extent of fibrosis within 24 months
- Composite criteria with decline in FVC ≥10%; or FVC ≥5%-9% and DLCO >15%.
Results: In total, 231 SSc-ILD patients were included (Table 1). At 12 months, 71 (30.7%) showed FVC decline >5% and were classified as progressors. In multivariable logistic regression ILD progression was significantly protective for further progression at the next annual follow-up (Odds Ratio 0.28, 95%CI 0.12-0.63, p=0.002, Figure1a). When other definitions of progression were applied, similar results were obtained: 39 (19%) fulfilled the PPF guideline criteria, 89 (39%) the PF-ILD criteria and 33 (14%) the composite criteria. Multivariable regression analysis adjusted for age, sex and treatment (PPF), for age and sex (composite) and for age, sex, anti-Topoisomerase I antibody (ATA), modified Rodnan skin Score (mRSS), baseline FVC and treatment (PF-ILD) showed the same direction as the primary analysis (Figure1b-d).
Conclusion: SSc-ILD progression does not predict further progression using any definition of ILD progression. These results challenge current treatment practice, since waiting for progression to initiate or escalate treatment does not seem to be the adequate strategy.

Table1: Baseline characteristics and death of patients fulfilling different definitions for ILD progression

A. Hoffmann-Vold: Arxx Therapeutics, 2, Boehringer-Ingelheim, 2, 5, 6, 12, Support for travel, Genentech, 2, Janssen, 2, 5, 6, Medscape, 2, 6, 12, Support for travel, Roche, 2, 6, 12, Support for travel; L. Petelytska: None; H. Fretheim: actelion, 5, bayer, 2, Boehringer-Ingelheim, 6, GlaxoSmithKlein(GSK), 5; T. Aaløkken: Boehringer-Ingelheim, 6; M. Becker: Amgen, 6, Bayer, 6, GSK, 6, Mepha, 6, MSD, 6, Novartis, 6, Vifor, 6; H. Jenssen Bjørkekjær: Janssen, 5; C. Brunborg: None; C. Bruni: AbbVie/Abbott, 5, Boehringer-Ingelheim, 2, 12, Travel Support, Eli Lilly, 6; C. Clarenbach: AstraZeneca, 1, 6, Boehringer-Ingelheim, 1, 6, CSL Behring, 1, 6, Daiichi Sankyo, 1, GlaxoSmithKlein(GSK), 1, 6, Grifols, 1, 6, Merck/MSD, 1, OM Pharma, 1, 6, Sanofi, 1, 6, Vifor, 1, 6; P. Diep: Boehringer-Ingelheim, 6, Roche, 6; R. Dobrota: Actelion, 5, 6, Amgen, 5, Articulum Fellowship, sponsored by Pfizer, 5, Boehringer-Ingelheim, 6; M. Durheim: Boehringer-Ingelheim, 2, 5, 6, Roche, 6; M. Elhai: AstraZeneca, 12, Travel to Congress support, Janssen, 12, Congress support; T. Frauenfelder: Boehringer-Ingelheim, 6; S. Jordan: None; E. Langballe: None; O. Midtvedt: None; C. Mihai: Boehringer-Ingelheim, 2, 5, 6, Janssen, 2, MED Talks Switzerland, 2, Mepha, 2, PlayToKnow AG, 2; O. Molberg: None; O. Distler: 4P-Pharma, 2, 5, 6, AbbVie, 2, 5, 6, Acceleron, 2, 5, 6, Alcimed, 2, 5, 6, Altavant Sciences, 2, 5, 6, Amgen, 2, 5, 6, AnaMar, 2, 5, 6, Arxx, 2, 5, 6, AstraZeneca, 2, 5, 6, Bayer, 2, 5, 6, Blade Therapeutics, 2, 5, 6, Boehringer Ingelheim, 2, 5, 6, Citus AG, 12, Co-Founder, Corbus Pharmaceuticals, 2, 5, 6, CSL Behring, 2, 5, 6, Galapagos, 2, 5, 6, Galderma, 2, 5, 6, Glenmark, 2, 5, 6, Gossamer, 2, 5, 6, Horizon Therapeutics, 2, 5, 6, Janssen, 2, 5, 6, Kymera, 2, 5, 6, Lupin, 2, 5, 6, Medscape, 2, 5, 6, Miltenyi Biotec, 2, 5, 6, Mitsubishi Tanabe, 2, 5, 6, MSD, 2, 5, 6, Novartis, 2, 5, 6, Patent issued “mir-29 for the treatment of systemic sclerosis” (US8247389, EP2331143), 10, Prometheus Biosciences, 2, 5, 6, Redx Pharma, 2, 5, 6, Roivant, 2, 5, 6, Topadur, 2, 5, 6.



