Poster Session A
Immunobiology
Session: (0066–0095) T Cell Biology & Targets in Autoimmune & Inflammatory Disease Poster
0093: Single Cell RNA-seq and Mass Cytometry Reveal a Cytotoxic CD8 Effector T Cell Population Associated with Interstitial Lung Disease in Systemic Sclerosis Patients
Sunday, November 12, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- YC
Ye Cao, PhD
Brigham and Women's Hospital
Boston, MA, United StatesDisclosure information not submitted.
Abstract Poster Presenter(s)
Ye Cao1, Takanori Sasaki2, Richard Ainsworth3, Kim Taylor4, Nunzio Bottini5, Mehreen Elahee6, Edy Kim7, Francesco Boin3 and Deepak Rao7, 1Brigham and Women's Hospital, Boston, MA, 2Brigham and Women's Hospital and Harvard Medical School, Boston, MA, 3Cedars-Sinai Medical Center, Los Angeles, CA, 4University of California San Francisco, San Francisco, CA, 5University of California, San Francisco, CA, 6University of Pittsburgh Medical Center, Pittsburgh, PA, 7Brigham and Women's Hospital, Boston, MA
Background/Purpose: Interstitial lung disease (ILD) is a major cause of morbidity and mortality in systemic sclerosis (SSc). We aimed to identify features of circulating immune cells associated with SSc-ILD to develop biomarkers and to find treatment targets of SSc-ILD.
Methods: We employed single cell RNA-seq (scRNAseq) using cryopreserved peripheral blood mononuclear cells (PBMC) from 20 SSc patients without ILD and 38 SSc patients with ILD and implemented covarying neighborhood analysis (CNA) to identify cell features associated with SSc-ILD. Detailed surface maker expression was also examined using mass cytometry (CyTOF) with a 39-marker panel using PBMC from 18 controls, 53 SSc patients with ILD and 29 SSc patients without ILD. Differentiation paths and inducing factors were explored through RNA velocity analysis and in-vitro culture.
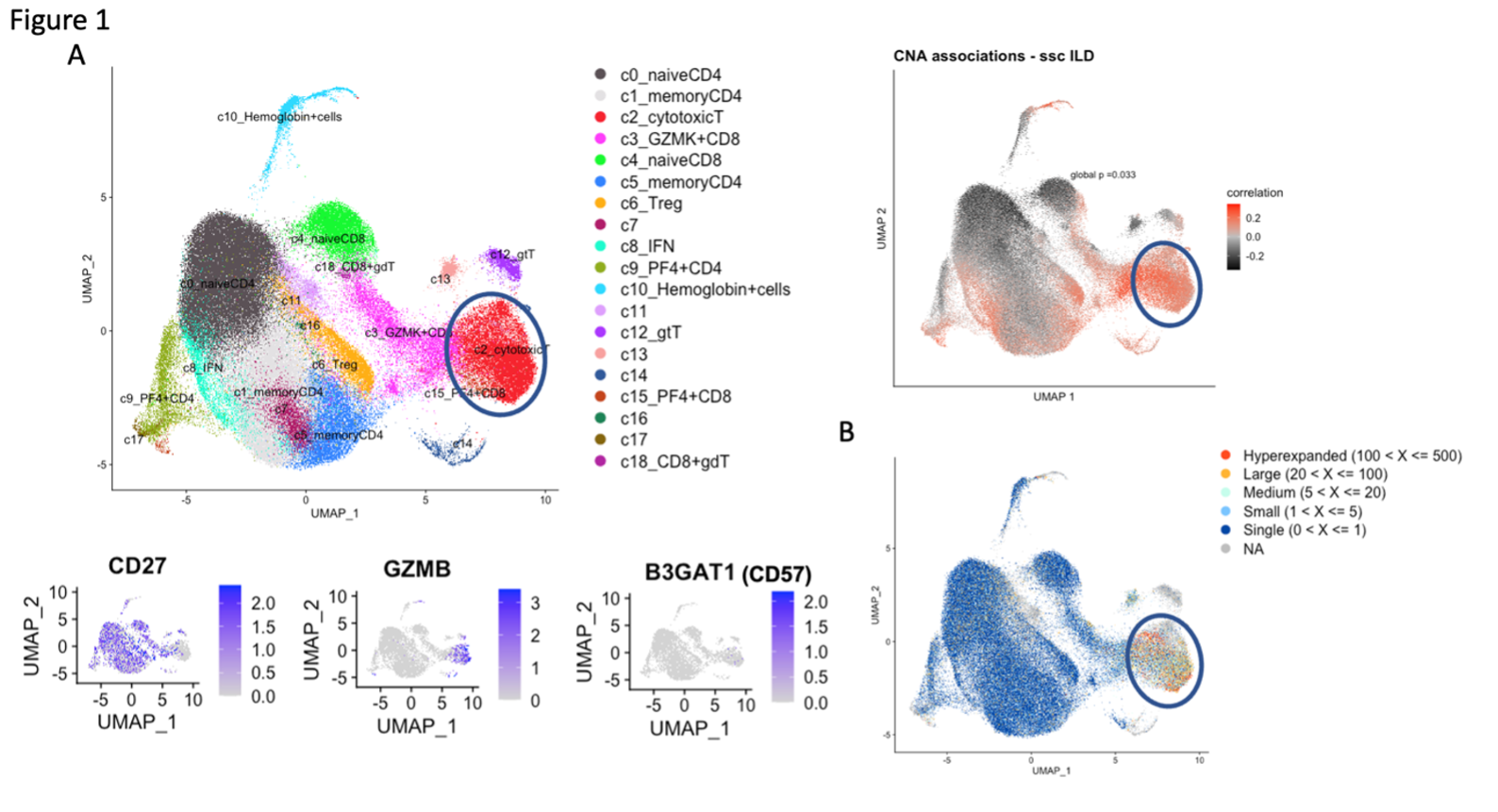
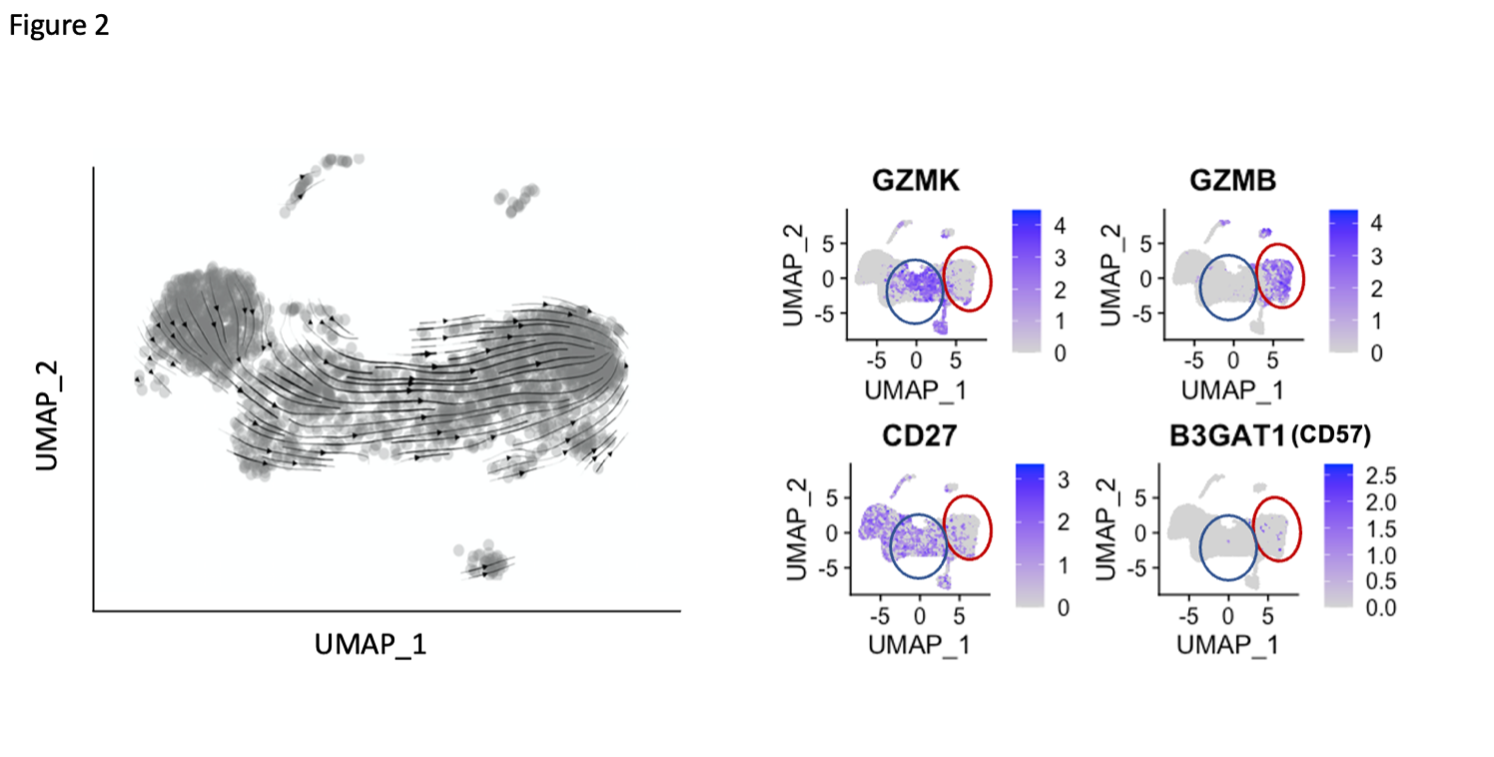
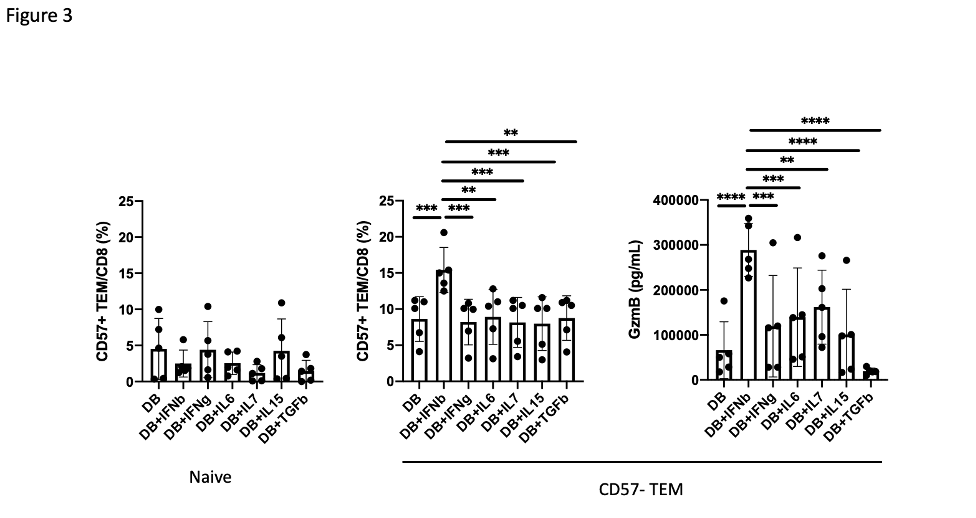
Results: A broad analysis of the scRNAseq dataset across mononuclear cells highlighted a significant enrichment in a specific CD8 T cell phenotype, captured as CD8 T cell cluster 2, in patients with ILD compared to those without ILD. This cluster expressed CD8A and GZMB but not CD27 or GZMK (Figure 1A). T cell receptor repertoire analysis indicated cluster 2 was clonally expanded, suggesting the antigen recognition of this CD8 T cell population in SSc-ILD (Figure 1B). In parallel, analysis of an overlapping set of PBMC samples by mass cytometry demonstrated that CD57+ CD27- CD56- CD45RO+ CCR7- effector memory CD8 T cells (CD57+ TEM) were significantly increased in the SSc-ILD cohort (Control: 0.74%, SSc without ILD: 1.42%, SSc with ILD: 2.71%), suggesting that a similar CD8 T cell population associated with SSc-ILD was captured by scRNAseq and mass cytometry. In contrast to CD57+ TEM cells, TEMRA were not statistically different between SSc patients with and without ILD. Independent flow cytometry analysis confirmed that CD57+ TEM highly expressed GZMB but not GZMK. On the contrary, CD57- CD27+ TEM (CD57- TEM) expressed GZMK but not GZMB, indicating there are two distinct populations in CD8 TEM. RNA velocity analysis of CD8 T cell clusters suggested that CD57+ TEM may differentiate from CD57- TEM (Figure 2). In-vitro culture experiments demonstrated that type I IFN facilitates differentiation from CD57- TEM to CD57+ TEM that highly produce GZMB (Figure 3).
Conclusion: ScRNAseq and mass cytometry analyses revealed that a cytotoxic effector memory CD8 T cell subset is expanded in SSc-ILD, and that a type I IFN signal may drive differentiation towards this phenotype.
Y. Cao: None; T. Sasaki: None; R. Ainsworth: None; K. Taylor: None; N. Bottini: Thirona Bio, 2; M. Elahee: None; E. Kim: Bayer, 5, Novartis, 12, Spouse is employee; F. Boin: None; D. Rao: AstraZeneca, 2, Bristol-Myers Squibb, 2, 5, GlaxoSmithKlein(GSK), 2, Hifibio, 2, Janssen, 5, Merck, 5, Scipher Medicine, 2.
Background/Purpose: Interstitial lung disease (ILD) is a major cause of morbidity and mortality in systemic sclerosis (SSc). We aimed to identify features of circulating immune cells associated with SSc-ILD to develop biomarkers and to find treatment targets of SSc-ILD.
Methods: We employed single cell RNA-seq (scRNAseq) using cryopreserved peripheral blood mononuclear cells (PBMC) from 20 SSc patients without ILD and 38 SSc patients with ILD and implemented covarying neighborhood analysis (CNA) to identify cell features associated with SSc-ILD. Detailed surface maker expression was also examined using mass cytometry (CyTOF) with a 39-marker panel using PBMC from 18 controls, 53 SSc patients with ILD and 29 SSc patients without ILD. Differentiation paths and inducing factors were explored through RNA velocity analysis and in-vitro culture.
Results: A broad analysis of the scRNAseq dataset across mononuclear cells highlighted a significant enrichment in a specific CD8 T cell phenotype, captured as CD8 T cell cluster 2, in patients with ILD compared to those without ILD. This cluster expressed CD8A and GZMB but not CD27 or GZMK (Figure 1A). T cell receptor repertoire analysis indicated cluster 2 was clonally expanded, suggesting the antigen recognition of this CD8 T cell population in SSc-ILD (Figure 1B). In parallel, analysis of an overlapping set of PBMC samples by mass cytometry demonstrated that CD57+ CD27- CD56- CD45RO+ CCR7- effector memory CD8 T cells (CD57+ TEM) were significantly increased in the SSc-ILD cohort (Control: 0.74%, SSc without ILD: 1.42%, SSc with ILD: 2.71%), suggesting that a similar CD8 T cell population associated with SSc-ILD was captured by scRNAseq and mass cytometry. In contrast to CD57+ TEM cells, TEMRA were not statistically different between SSc patients with and without ILD. Independent flow cytometry analysis confirmed that CD57+ TEM highly expressed GZMB but not GZMK. On the contrary, CD57- CD27+ TEM (CD57- TEM) expressed GZMK but not GZMB, indicating there are two distinct populations in CD8 TEM. RNA velocity analysis of CD8 T cell clusters suggested that CD57+ TEM may differentiate from CD57- TEM (Figure 2). In-vitro culture experiments demonstrated that type I IFN facilitates differentiation from CD57- TEM to CD57+ TEM that highly produce GZMB (Figure 3).
Conclusion: ScRNAseq and mass cytometry analyses revealed that a cytotoxic effector memory CD8 T cell subset is expanded in SSc-ILD, and that a type I IFN signal may drive differentiation towards this phenotype.

Figure 1.scRNAseq analysis of our SSc-ILD cohort. A. Covarying neighborhood analysis indicated cluster 2 is associated with lung disease in SSc, which expressed GZMB and B3GAT1 (CD57) but not expressed CD27. B. T cell receptor repertoire analysis demonstrated cluster 2 is a clonally expanded population.

Figure 2. Differentiation pathway analysis of CD57+ TEM. RNA velocity analysis of CD8 T cell clusters indicated that CD57+ TEM (red circle) is differentiated from CD57- TEM (blue circle).

Figure 3. Type I IFN promotes differentiation from CD57- TEM to CD57+ TEM. Proportion of CD57+ TEM after 4 days culture of Naïve or CD57-TEM from 5 different donors with CD3/CD28 + cytokine stimulation. Granzyme B production was assessed by ELISA using culture supernatant after 4 days of culture of CD57- TEM with CD3/CD28 + cytokine stimulation. P value was calculated by One-Way repeated measures ANOVA.
Y. Cao: None; T. Sasaki: None; R. Ainsworth: None; K. Taylor: None; N. Bottini: Thirona Bio, 2; M. Elahee: None; E. Kim: Bayer, 5, Novartis, 12, Spouse is employee; F. Boin: None; D. Rao: AstraZeneca, 2, Bristol-Myers Squibb, 2, 5, GlaxoSmithKlein(GSK), 2, Hifibio, 2, Janssen, 5, Merck, 5, Scipher Medicine, 2.



