Poster Session A
Immunobiology
Session: (0066–0095) T Cell Biology & Targets in Autoimmune & Inflammatory Disease Poster
0092: Multi-omics Immune Profiling of Cytotoxic T Cells from Ankylosing Spondylitis Patients Identified a Subset of Clonally Exapnded CTLs That Evade Immune Exhaustion
Sunday, November 12, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- ZQ
Zoya Qaiyum, MSc
Krembil Research Institute
Toronto, ON, CanadaDisclosure information not submitted.
Abstract Poster Presenter(s)
Michael Tang1, Zoya Qaiyum2, Melissa Lim1 and Robert Inman1, 1University Health Network, Toronto, ON, Canada, 2Krembil Research Institute, Toronto, ON, Canada
Background/Purpose: Sustained chronic inflammation in the spine and of the sacroiliac joints is a key feature in Ankylosing Spondylitis (AS). A central role of CTL involvement in AS inflammation is supported by recent discoveries of chronically activated CTLs with distinctive cytotoxic and integrin expression profiles enriched at sites of inflammation. Yet, the immunophenotype of the CTL compartment which may perpetuate AS inflammation has not been fully defined. The microenvironment that is conducive to AS chronic inflammation is reminiscent to settings found in chronic viral infection and cancer. Here, we seek to unravel the complex heterogeneity of CTLs in AS patients to determine whether CTLs in AS are truly immunologically exhausted. We hypothesize that a loss of CTL immune homeostasis and evasion of T cell exhaustion contribute to autoinflammation in AS.
Methods: 8 pairs of PBMCs and SFMCs from patients with active (mean BASDAI=8) Axial Spondyloarthritis meeting the ASAS classification criteria were analysed. Additionally, age and sex-matched healthy control PBMCs (n=18) were analysed. We applied multi-omics single-cell immune profiling strategies to delineate the complex heterogeneity of CTLs in AS. Mass cytometry (CyTOF) of PBMCs and SFMCs was used to simultaneously measure protein expression of more than 30 surface and intracellular markers. Single-cell RNA sequencing (scRNAseq) and T cell receptor sequencing (scTCRseq) of FACS-sorted mature CTLs were used to immune profile AS CTL transcriptome and to perform clonal andTCR diversity analyses.
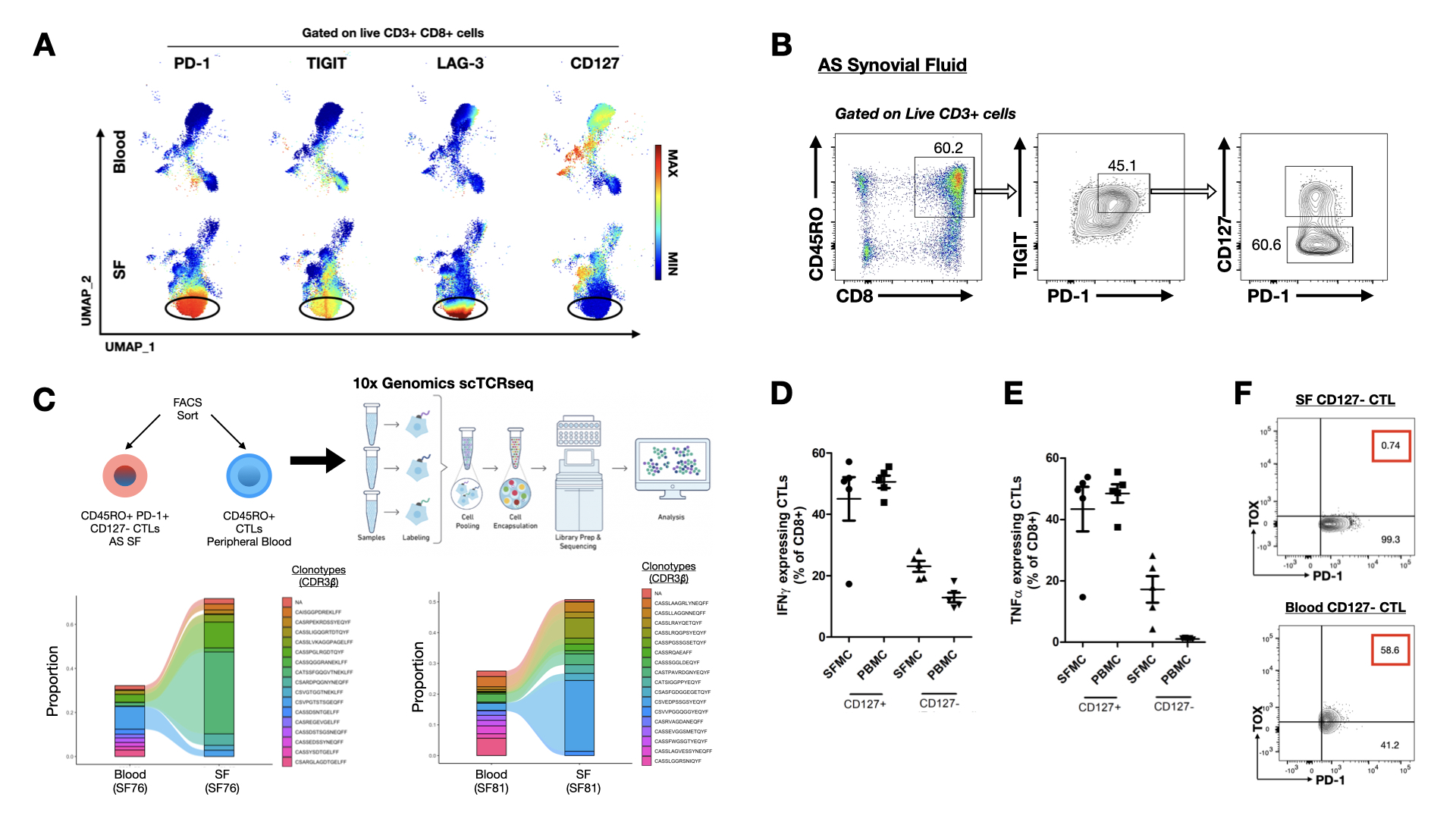
Results: Unsupervised clustering analysis of CyTOF data identified a subset of CTLs that co-express checkpoint receptors PD-1, TIGIT, and LAG-3 in SF CTLs of AS patients; while these markers are downregulated in the peripheral CTLs compared to healthy controls, implicating CTL dysregulation. This subset of SF CTLs downregulates CD127 expression, while retaining the capacity for CTL effector functions including the ability to express granzyme B, perforin and produce IFNγ and TNFαupon stimulation. Additionally, CD8+ T cell tissue residency markers (CD103, CD69, and CD49a) are highly upregulated in this subset of SF CTLs. scRNAseq of FACS-sorted CD45RO+ PD-1+ TIGIT+ CTLs revealed that transcripts that are normally expressed in canonically exhausted CTLs (e.g. CD244, HAVCR2, ENTPD1, NT5E, TOX) are downregulated. Differential gene analysis revealed that the top 5 genes (IFI6, MX1, ISG16, MT2A, IFITM1) upregulated in these cells are related to the interferon signalling pathway. Single cell TCR sequencing demonstrated that this CTL subset is clonally expanded at sites of inflammation. Intracellular flow cytometry confirmed that this CTL subset downregulated the master transcription factor regulating T cell exhaustion- TOX, suggesting that these CTLs are not truly immunologically exhausted.
Conclusion: Multi-omics immune profiling of CTLs from AS patients identified a subset of clonally expanded CTLs in SF that appear to resist immune exhaustion, while retaining classical cytotoxic capacities. This findings implicates that dysregulation of T cell exhaustion and homeostasis could potentially exacerbate AS autoinflammation.
M. Tang: None; Z. Qaiyum: None; M. Lim: Aria Pharmaceuticals, 5; R. Inman: AbbVie, 2, Eli Lilly, 2, Janssen, 2, Novartis, 2, Sandoz, 2.
Background/Purpose: Sustained chronic inflammation in the spine and of the sacroiliac joints is a key feature in Ankylosing Spondylitis (AS). A central role of CTL involvement in AS inflammation is supported by recent discoveries of chronically activated CTLs with distinctive cytotoxic and integrin expression profiles enriched at sites of inflammation. Yet, the immunophenotype of the CTL compartment which may perpetuate AS inflammation has not been fully defined. The microenvironment that is conducive to AS chronic inflammation is reminiscent to settings found in chronic viral infection and cancer. Here, we seek to unravel the complex heterogeneity of CTLs in AS patients to determine whether CTLs in AS are truly immunologically exhausted. We hypothesize that a loss of CTL immune homeostasis and evasion of T cell exhaustion contribute to autoinflammation in AS.
Methods: 8 pairs of PBMCs and SFMCs from patients with active (mean BASDAI=8) Axial Spondyloarthritis meeting the ASAS classification criteria were analysed. Additionally, age and sex-matched healthy control PBMCs (n=18) were analysed. We applied multi-omics single-cell immune profiling strategies to delineate the complex heterogeneity of CTLs in AS. Mass cytometry (CyTOF) of PBMCs and SFMCs was used to simultaneously measure protein expression of more than 30 surface and intracellular markers. Single-cell RNA sequencing (scRNAseq) and T cell receptor sequencing (scTCRseq) of FACS-sorted mature CTLs were used to immune profile AS CTL transcriptome and to perform clonal andTCR diversity analyses.
Results: Unsupervised clustering analysis of CyTOF data identified a subset of CTLs that co-express checkpoint receptors PD-1, TIGIT, and LAG-3 in SF CTLs of AS patients; while these markers are downregulated in the peripheral CTLs compared to healthy controls, implicating CTL dysregulation. This subset of SF CTLs downregulates CD127 expression, while retaining the capacity for CTL effector functions including the ability to express granzyme B, perforin and produce IFNγ and TNFαupon stimulation. Additionally, CD8+ T cell tissue residency markers (CD103, CD69, and CD49a) are highly upregulated in this subset of SF CTLs. scRNAseq of FACS-sorted CD45RO+ PD-1+ TIGIT+ CTLs revealed that transcripts that are normally expressed in canonically exhausted CTLs (e.g. CD244, HAVCR2, ENTPD1, NT5E, TOX) are downregulated. Differential gene analysis revealed that the top 5 genes (IFI6, MX1, ISG16, MT2A, IFITM1) upregulated in these cells are related to the interferon signalling pathway. Single cell TCR sequencing demonstrated that this CTL subset is clonally expanded at sites of inflammation. Intracellular flow cytometry confirmed that this CTL subset downregulated the master transcription factor regulating T cell exhaustion- TOX, suggesting that these CTLs are not truly immunologically exhausted.
Conclusion: Multi-omics immune profiling of CTLs from AS patients identified a subset of clonally expanded CTLs in SF that appear to resist immune exhaustion, while retaining classical cytotoxic capacities. This findings implicates that dysregulation of T cell exhaustion and homeostasis could potentially exacerbate AS autoinflammation.

Mass cytometry and single cell TCR sequencing identified a population of clonally expanded SF CTL that evades immune exhaustion. A) Live CD3+ CD8+ cells from single file representations of paired AS SFMC and PBMC (n=8) were entered into the UMAP algorithm for high dimensional analysis of immune checkpoint molecules. UMAP projections display expression levels of indicated surface molecules. B) Representative flow cytometric analyses of CD127+ and CD127- PD-1+ TIGIT+ CTL from SF of AS patient. C) Schematic workflow of single cell TCR sequencing. Mature CD45RO expressing CTLs were sorted from matched SFMC and PBMC from 2 AS patients. Alluvial plots demonstrating proportion of clonotypes based on amino acid sequences of CDR3b chains of CTLs from blood and SF of 2 AS patients. D-E) Cytokine release assay; CD127+ and CD127- PD-1+ TIGIT+ CTLs were FACS sorted from paired SFMC and PBMC samples, then stimulated with PMA/ionomycin for 4 hours in the presence of Brefeldin A. Intracellular IFNg and TNFa were measured by flow cytometry. Graphs display proportion of CD8+ T cells expressing the cytokines. F) Flow cytometric analysis to quantify frequency of TOX+ CD127- PD-1+ CTL.

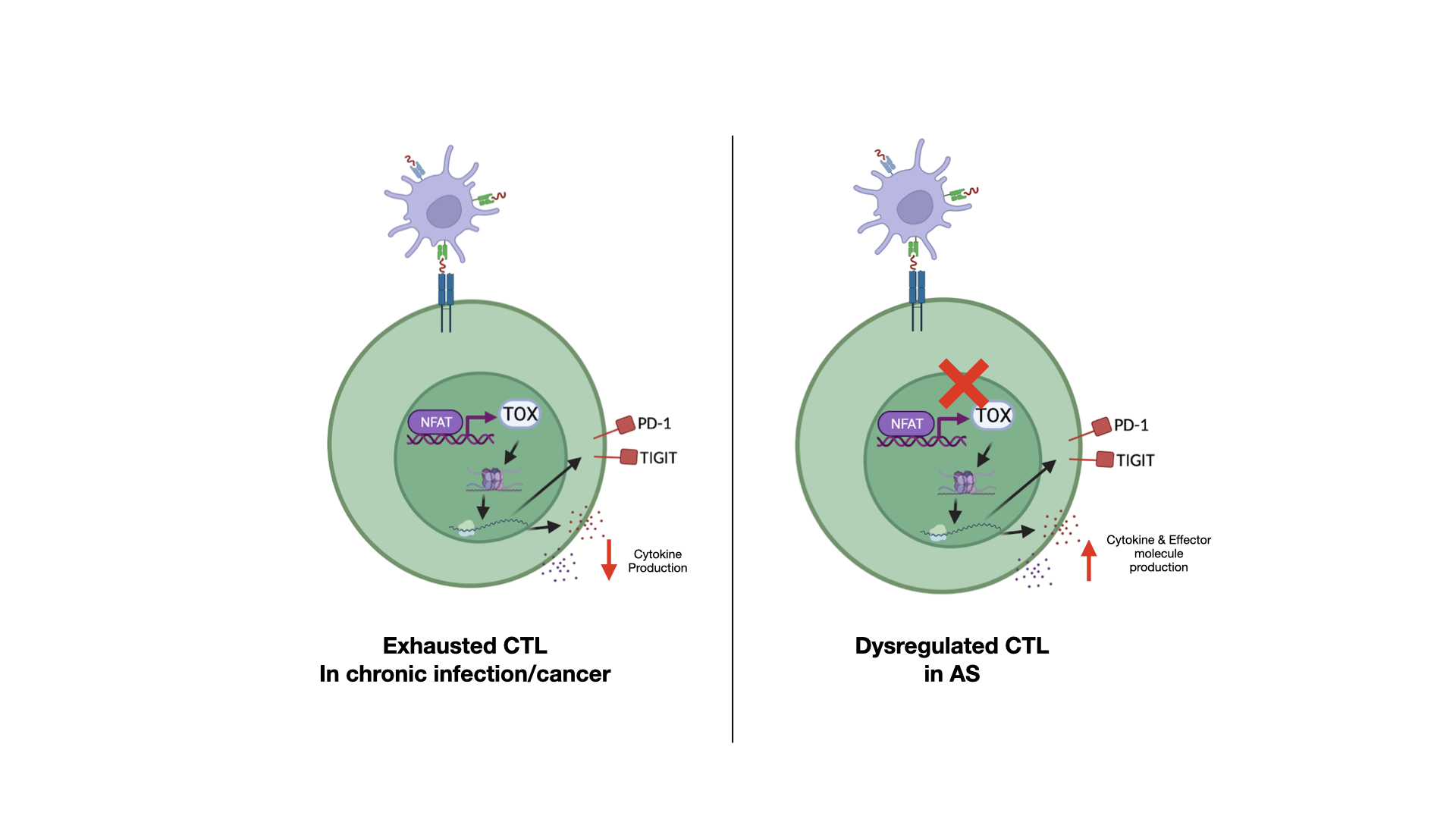
Dysregulation of CTLs in AS synovial fluid. In contrast to canonically exhausted CTLs in settings of chronic inflammation such as chronic viral infection and cancer, a distinct subset of CTLs expressing classical immune checkpoint markers appear to resist immune exhaustion, retain effector functions, and progressively perpetuate autoinflammation in AS.
M. Tang: None; Z. Qaiyum: None; M. Lim: Aria Pharmaceuticals, 5; R. Inman: AbbVie, 2, Eli Lilly, 2, Janssen, 2, Novartis, 2, Sandoz, 2.



