Poster Session A
Systemic lupus erythematosus (SLE)
Session: (0543–0581) SLE – Diagnosis, Manifestations, & Outcomes Poster I
0561: Frequency of Anti-Ro Antibodies in Systemic Lupus Erythematosus Patients: Insights from Multicenter and National Registry
Sunday, November 12, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- LM
Lucia Mendoza Martinez, APRN (she/her/hers)
Hospital General de Agudos Dr. Enrique Tornú
Buenos Aires, ArgentinaDisclosure information not submitted.
Abstract Poster Presenter(s)
Lucia Margarita Mendoza Martinez1, Joan Manuel Dapeña1, Juan Manuel Bande1, Maria Alejandra Medina1, Silvia Papasidero1, José caracciolo1, Rosana Quintana2, Lucila Garcia2, Carla Andrea Gobbi2, Sandy Sapag Durán2, Alberto Spindler2, Analia Patricia Alvarez2, Cecilia Pisoni2, Catalina Gomez2, Raúl Hector Paniego2, María Julia Santa cruz2, Luciana Gonzalez Lucero2, Rodrigo Aguila Maldonado2, Sergio Gordon2, Julia Romero2, Gretel Rausch2, Alberto Allievi2, Alberto Omar Orden2, johana zacariaz hereter3, Roberto Baéz2, Andrea Vanesa González2, Juan Manuel Vandale2, Mario Alberto Goñi2, Victor Caputo4, María Silvia Larroudé2, Graciela Gómez2, Josefina Marin2, Victoria Collado2, Gazzoni María Victoria2, Marcos David Zelaya2, Mónica Sacnún2, Romina Rojas Tessel2, Maira Alejandra Arias Saavedra2, Maximiliano Machado Escobar2, Pablo Astesana2, Ursula Vanesa Paris2, Bernando A. Pons-Estel2 and Mercedes García5, 1Hospital General de Agudos Dr. Enrique Tornú, Buenos Aires, Argentina, 2Study Group of the Argentine Society of Rheumatology for Systemic Lupus Erythematosus, Buenos Aires, Argentina, 3Hospital Italiano de Buenos Aires, Buenos Aires, Argentina, 4Hospital Militar Central, Buenos Aires, Argentina, 5Hospital Interzonal General de Agudos José de San Martín, La Plata, Argentina
Background/Purpose: Anti-Ro antibodies can be detected in 40% of patients with systemic lupus erythematosus (SLE) and have been associated with various clinical manifestations of the disease as well as maternal-fetal complications. The aim of this study was to describe the frequency of anti-Ro antibodies in SLE patients and to assess their association with disease characteristics, maternal-fetal complications, treatment, morbidity and mortality.
Methods: Retrospective observational study. Data from SLE patients of the National Registry of the Argentine Society of Rheumatology (RELESSAR) were used. Sociodemographic data, comorbidities, disease characteristics, maternal-fetal complications, SLE morbidity and mortality, activity and damage scores were recorded.
Statistical analysis: Descriptive statistics. Appropriate tests (Chi2, Fisher, Student's t-test or Wilcoxon), and univariate/multivariate logistic regression analyses were conducted to identify factors associated with presence of anti-Ro antibodies.
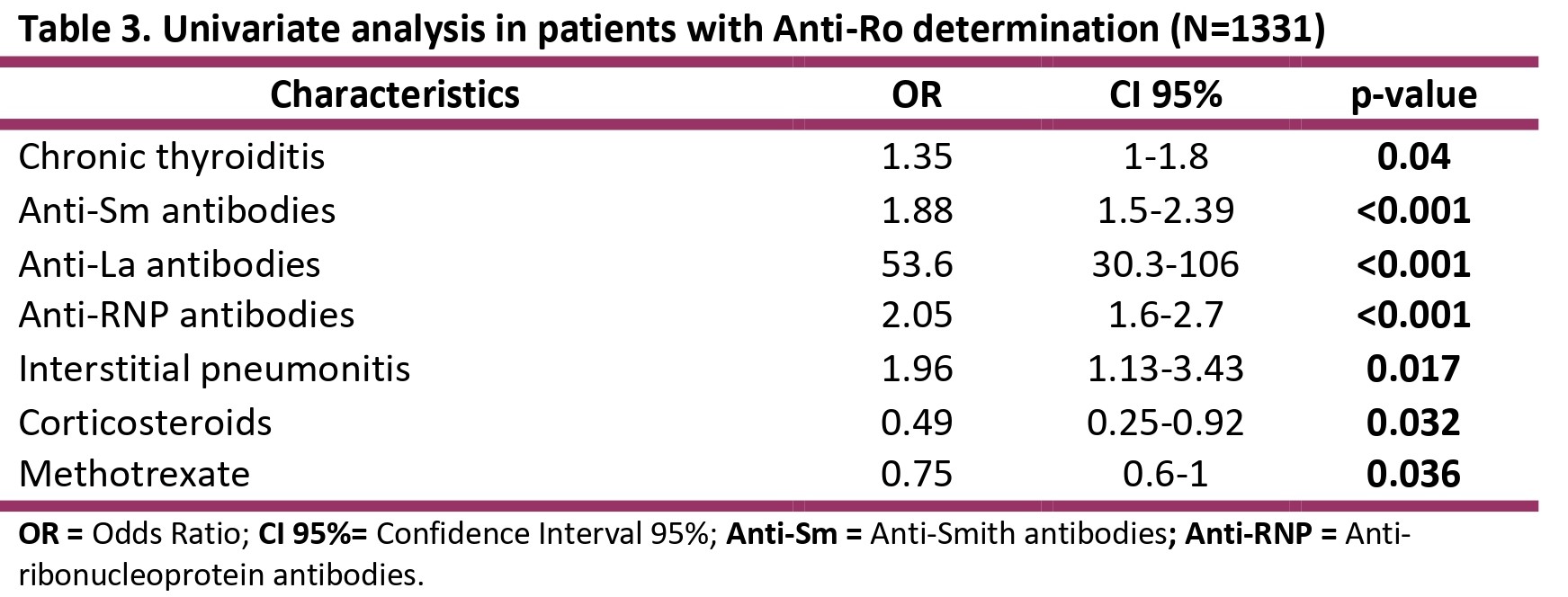
Results: A total of 1513 patients were included. Patient characteristics are shown in Table 1. Anti-Ro antibodies were requested in 88% (n=1331), of which 41% (n=552) were positive. Characteristics of patients with positive and negative Anti-Ro antibodies can be observed inTable 2. Anti-Ro antibodies were associated with hypocomplementemia (p< 0.05), anti-La antibodies (< 0.001), anti-Sm antibodies (p< 0.001), anti-RNP antibodies (p< 0.001) and Sjogren's syndrome (p< 0.001). Also were associated with Raynaud's phenomenon (p< 0.004), pericarditis (p< 0.005) and interstitial pneumonitis (p< 0.02). More infections (p< 0.01) and a greater use of corticosteroids (p< 0.04) and methotrexate (p< 0.04) were observed. Univariate analysis in patients with Anti-Ro determination is shown in table 3. In the multivariate model, the independent variables were anti-Sm antibodies (OR 1.9, 95% CI 1.4-2.5), anti-La antibodies (OR 48, 95%CI 26-99), chronic thyroiditis (OR 1.9, 95%CI 1.3-2.7) and the use of methotrexate (OR 0.66, 95%CI 0.5-0.9).
Conclusion: In this SLE patients, anti-Ro positivity was associated with the presence of other antibodies, chronic thyroiditis and methotrexate use, but not with any specific clinical profile, maternal-fetal complications or SLE-related morbidity and mortality.

.jpg)
L. Mendoza Martinez: None; J. Dapeña: None; J. Bande: None; M. Medina: None; S. Papasidero: None; J. caracciolo: None; R. Quintana: None; L. Garcia: None; C. Gobbi: None; S. Sapag Durán: None; A. Spindler: None; A. Alvarez: None; C. Pisoni: None; C. Gomez: None; R. Paniego: None; M. Santa cruz: None; L. Gonzalez Lucero: None; R. Aguila Maldonado: None; S. Gordon: None; J. Romero: None; G. Rausch: None; A. Allievi: None; A. Orden: None; j. zacariaz hereter: None; R. Baéz: None; A. González: None; J. Vandale: None; M. Goñi: None; V. Caputo: None; M. Larroudé: None; G. Gómez: None; J. Marin: None; V. Collado: None; G. María Victoria: None; M. Zelaya: None; M. Sacnún: None; R. Rojas Tessel: None; M. Arias Saavedra: None; M. Machado Escobar: None; P. Astesana: None; U. Paris: None; B. Pons-Estel: None; M. García: GSK, 6, Janssen, 6, Pfizer, 6.
Background/Purpose: Anti-Ro antibodies can be detected in 40% of patients with systemic lupus erythematosus (SLE) and have been associated with various clinical manifestations of the disease as well as maternal-fetal complications. The aim of this study was to describe the frequency of anti-Ro antibodies in SLE patients and to assess their association with disease characteristics, maternal-fetal complications, treatment, morbidity and mortality.
Methods: Retrospective observational study. Data from SLE patients of the National Registry of the Argentine Society of Rheumatology (RELESSAR) were used. Sociodemographic data, comorbidities, disease characteristics, maternal-fetal complications, SLE morbidity and mortality, activity and damage scores were recorded.
Statistical analysis: Descriptive statistics. Appropriate tests (Chi2, Fisher, Student's t-test or Wilcoxon), and univariate/multivariate logistic regression analyses were conducted to identify factors associated with presence of anti-Ro antibodies.
Results: A total of 1513 patients were included. Patient characteristics are shown in Table 1. Anti-Ro antibodies were requested in 88% (n=1331), of which 41% (n=552) were positive. Characteristics of patients with positive and negative Anti-Ro antibodies can be observed inTable 2. Anti-Ro antibodies were associated with hypocomplementemia (p< 0.05), anti-La antibodies (< 0.001), anti-Sm antibodies (p< 0.001), anti-RNP antibodies (p< 0.001) and Sjogren's syndrome (p< 0.001). Also were associated with Raynaud's phenomenon (p< 0.004), pericarditis (p< 0.005) and interstitial pneumonitis (p< 0.02). More infections (p< 0.01) and a greater use of corticosteroids (p< 0.04) and methotrexate (p< 0.04) were observed. Univariate analysis in patients with Anti-Ro determination is shown in table 3. In the multivariate model, the independent variables were anti-Sm antibodies (OR 1.9, 95% CI 1.4-2.5), anti-La antibodies (OR 48, 95%CI 26-99), chronic thyroiditis (OR 1.9, 95%CI 1.3-2.7) and the use of methotrexate (OR 0.66, 95%CI 0.5-0.9).
Conclusion: In this SLE patients, anti-Ro positivity was associated with the presence of other antibodies, chronic thyroiditis and methotrexate use, but not with any specific clinical profile, maternal-fetal complications or SLE-related morbidity and mortality.

Table 1. Patient characteristics (N=1513)
.jpg)
Table 2. Characteristics of patients with positive and negative anti-Ro antibodies

Table 3. Univariate analysis in patients with Anti-Ro determination (N=1331)
L. Mendoza Martinez: None; J. Dapeña: None; J. Bande: None; M. Medina: None; S. Papasidero: None; J. caracciolo: None; R. Quintana: None; L. Garcia: None; C. Gobbi: None; S. Sapag Durán: None; A. Spindler: None; A. Alvarez: None; C. Pisoni: None; C. Gomez: None; R. Paniego: None; M. Santa cruz: None; L. Gonzalez Lucero: None; R. Aguila Maldonado: None; S. Gordon: None; J. Romero: None; G. Rausch: None; A. Allievi: None; A. Orden: None; j. zacariaz hereter: None; R. Baéz: None; A. González: None; J. Vandale: None; M. Goñi: None; V. Caputo: None; M. Larroudé: None; G. Gómez: None; J. Marin: None; V. Collado: None; G. María Victoria: None; M. Zelaya: None; M. Sacnún: None; R. Rojas Tessel: None; M. Arias Saavedra: None; M. Machado Escobar: None; P. Astesana: None; U. Paris: None; B. Pons-Estel: None; M. García: GSK, 6, Janssen, 6, Pfizer, 6.



