Abstract Session
Fibrosing rheumatic diseases (scleroderma, MCTD, IgG4-related disease, scleroderma mimics)
Session: Abstracts: Systemic Sclerosis & Related Disorders III: Clinical Trials (2593–2598)
2597: Fecal Microbiota Transplantation in Patients with Systemic Sclerosis and Lower Gastrointestinal Tract Symptoms: Data from the ReSScue Phase 2 Randomized Clinical Trial
Wednesday, November 15, 2023
12:00 PM - 12:10 PM PT
Location: Room 32A-B
- AH
Anna-Maria Hoffmann-Vold, MD, PhD
Oslo University Hospital
Oslo, NorwayDisclosure information not submitted.
Presenting Author(s)
Anna-Maria Hoffmann-Vold1, Havard Fretheim1, Imon Barua1, Maylen Nordgård Carstens1, Henriette Didriksen2, Vikas Sarna1, Knut AE Lundin1, Oliver Distler3, Dinesh Khanna4, Elizabeth Volkmann5, Oyvind Midtvedt1, Tore Midtvedt6, Alvilde Dhainaut7, Anne-Kristine H Halse8, Gunnstein Bakland9, Inge Olsen1, Maiju E Pesonen1 and Oyvind Molberg10, 1Oslo University Hospital, Oslo, Norway, 2Oslo University Hospital, Moss, Norway, 3Department of Rheumatology, University Hospital Zurich, University of Zurich, Zurich, Switzerland, 4University of Michigan, Ann Arbor, MI, 5University of California Los Angeles, Los Angeles, CA, 6Karolinska Institutet, Stockholm, Sweden, 7St. Olav, Trondheim, Norway, 8University Hospital Bergen, Bergen, Norway, 9University Hospital of North Norway, Tromsø, Norway, 10Dept of Rheumatology, University Hospital Oslo, Oslo, Norway
Background/Purpose: Lower gastrointestinal tract (GIT) complications are common in patients with systemic sclerosis (SSc), associate with a high disease burden, and current treatment alternatives are limited. Patients with SSc have also an altered intestinal microbiota composition. This provides a rational for the investigation of fecal microbiota transplantation (FMT) in SSc patients with lower GIT symptoms. In this randomized mutlicenter double-blind clinical trial (RCT) we assessed the safety and efficacy of a standardized intestinal microbiota infusion in SSc patients with lower GIT symptoms.
Methods: Patients with SSc and moderate to severe bloating and/or diarrhea assessed by the UCLA SCTC GIT score 2.0 were enrolled in a multicenter, double-blind, randomized, placebo-controlled phase 2 trial. Patients were randomized to receive FMT with an intestinal infusion of a standardized fecal microbiota culture (ACHIM) or placebo at weeks 0 and 2. At week 12, all patients received a FMT infusion and were followed in an open lable phase until week 20. The primary outcome was change between baseline and week 12 in UCLA GIT score item diarrhea or bloating measured as the average marginal effect (AME), depending on which was the worst symptom at baseline; evaluated separately for each patient. Comprehensive predefined subgroup analyses were conducted. Secondary outcomes were safety and tolerability and total UCLA GIT score. Other outcome measures included the change in UCLA GIT score from week 12 to week 20.
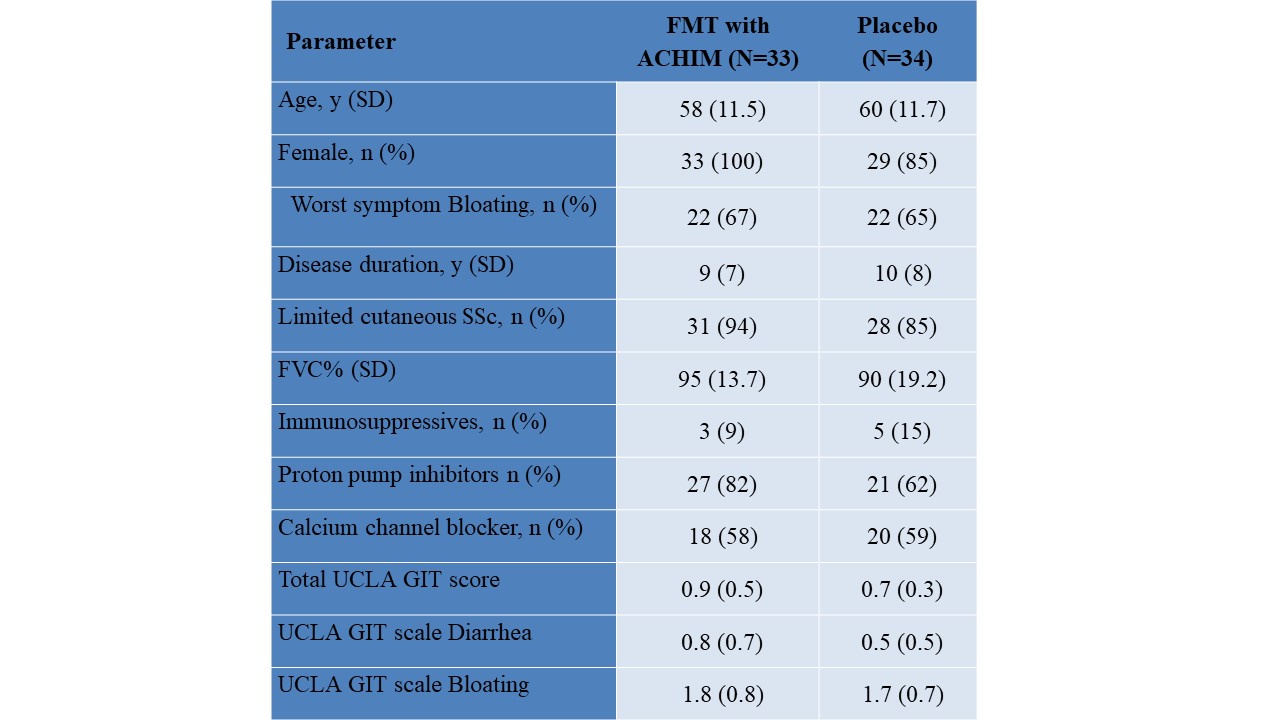
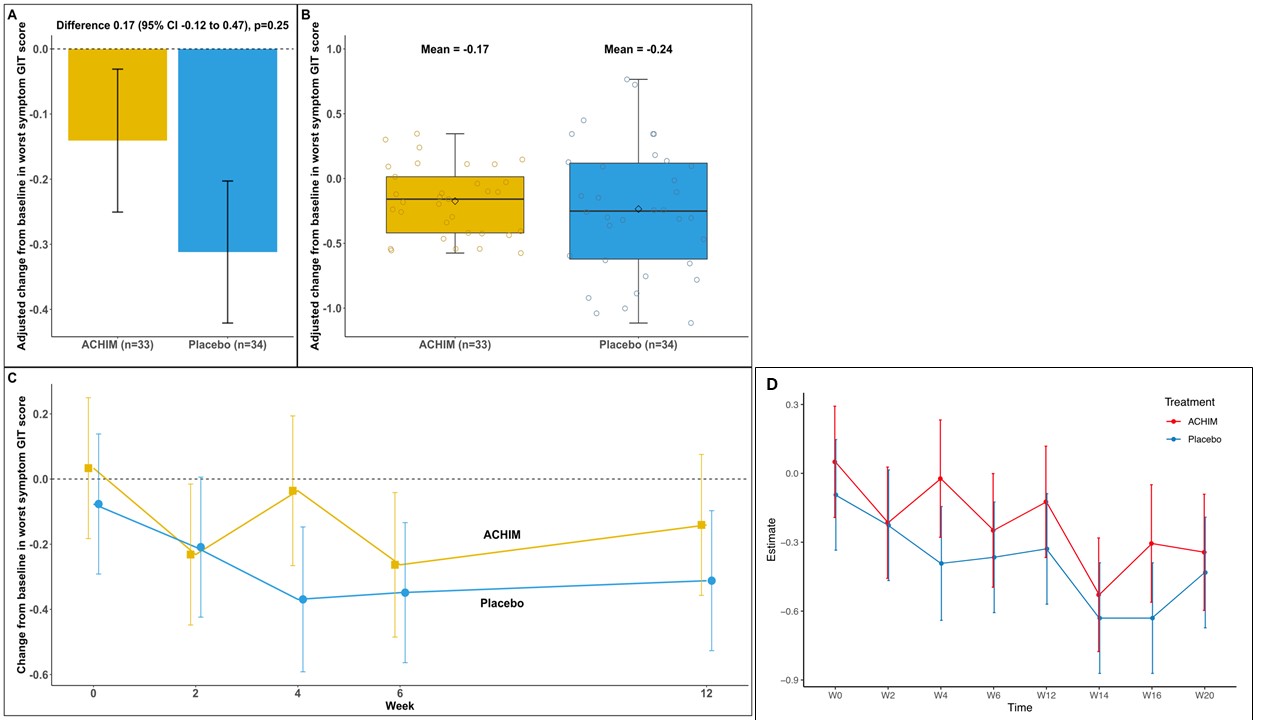
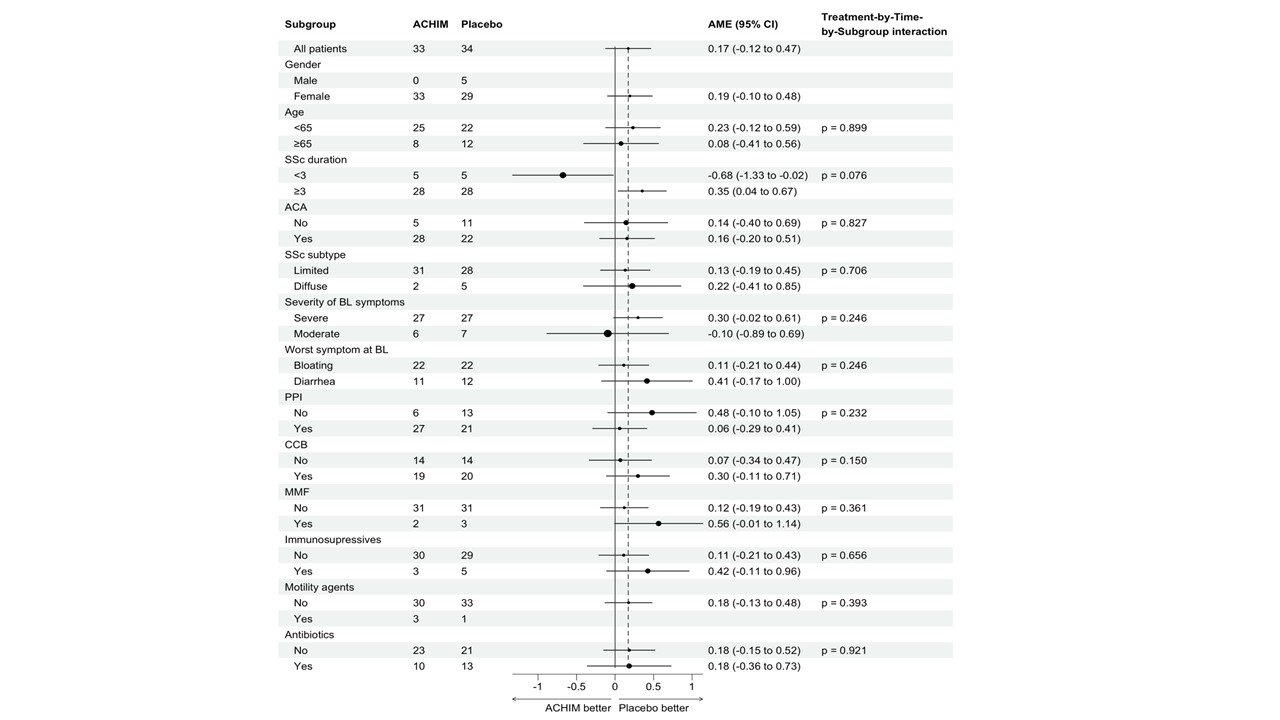
Results: A total of 65 patients were randomized to receive FMT or placebo. Baseline characteristics in the groups were comparable (Table). There was no significant difference in the change in lower GIT symptoms from week 0 to week 12 between the groups (AME=0.17 (-0.12, 0.47), p=0.25) and no significant differences in any of the predefined subgroup analyses (Figure 1A-C and 2). Similarly, no significant difference was observed in the total GIT score between the two groups (AME=0.09 (-0.04, 0.23), p=0.17). Furthermore, during the open lable period, there was no statistical difference in change between the ACHIM and the placebo group (AME= -0.04 (-0.32, 0.23), p=0.077) shown in Figure 1D. Participants treated with ACHIM, 16 (37%) and with placebo 19 (42%) experienced any side effects. These were in general mild and short-lasting, with abdominal pain as the most frequent side effect present in 5 (15%) in ACHIM and 2 (6%) in placebo. Time to resolved pain was 2days in both groups. One patient experienced an intramural perforation during gastroscopy and needed IV antibiotics but fully recovered.
Conclusion: We were unable to find indications that FMT improves lower GIT symptoms in SSc patients, but the treatment was found to be safe.
A. Hoffmann-Vold: Arxx Therapeutics, 2, Boehringer-Ingelheim, 2, 5, 6, 12, Support for travel, Genentech, 2, Janssen, 2, 5, 6, Medscape, 2, 6, 12, Support for travel, Roche, 2, 6, 12, Support for travel; H. Fretheim: actelion, 5, bayer, 2, Boehringer-Ingelheim, 6, GlaxoSmithKlein(GSK), 5; I. Barua: None; M. Nordgård Carstens: None; H. Didriksen: None; V. Sarna: None; K. Lundin: Falk Pharma, 2, GSK, 2, Takeda, 2, 6, Topas, 2; O. Distler: 4P-Pharma, 2, 5, 6, AbbVie, 2, 5, 6, Acceleron, 2, 5, 6, Alcimed, 2, 5, 6, Altavant Sciences, 2, 5, 6, Amgen, 2, 5, 6, AnaMar, 2, 5, 6, Arxx, 2, 5, 6, AstraZeneca, 2, 5, 6, Bayer, 2, 5, 6, Blade Therapeutics, 2, 5, 6, Boehringer Ingelheim, 2, 5, 6, Citus AG, 12, Co-Founder, Corbus Pharmaceuticals, 2, 5, 6, CSL Behring, 2, 5, 6, Galapagos, 2, 5, 6, Galderma, 2, 5, 6, Glenmark, 2, 5, 6, Gossamer, 2, 5, 6, Horizon Therapeutics, 2, 5, 6, Janssen, 2, 5, 6, Kymera, 2, 5, 6, Lupin, 2, 5, 6, Medscape, 2, 5, 6, Miltenyi Biotec, 2, 5, 6, Mitsubishi Tanabe, 2, 5, 6, MSD, 2, 5, 6, Novartis, 2, 5, 6, Patent issued “mir-29 for the treatment of systemic sclerosis” (US8247389, EP2331143), 10, Prometheus Biosciences, 2, 5, 6, Redx Pharma, 2, 5, 6, Roivant, 2, 5, 6, Topadur, 2, 5, 6; D. Khanna: AbbVie, 12, DSMB, AstraZeneca, 2, Boehringer-Ingelheim, 2, Bristol-Myers Squibb, 2, 5, CSL Behring, 2, Genentech, 2, Horizon Therapeutics, 2, 5, Janssen, 2, 6, Pfizer, 5, Prometheus, 2; E. Volkmann: Boehringer-Ingelheim, 2, 5, 6, CSL Behring, 2, GlaxoSmithKline, 2, Horizon, 5, Prometheus, 5, Roche, 2; O. Midtvedt: None; T. Midtvedt: ACHIM, 8, 10, 11; A. Dhainaut: None; A. Halse: None; G. Bakland: UCB, 2; I. Olsen: None; M. Pesonen: None; O. Molberg: None.
Background/Purpose: Lower gastrointestinal tract (GIT) complications are common in patients with systemic sclerosis (SSc), associate with a high disease burden, and current treatment alternatives are limited. Patients with SSc have also an altered intestinal microbiota composition. This provides a rational for the investigation of fecal microbiota transplantation (FMT) in SSc patients with lower GIT symptoms. In this randomized mutlicenter double-blind clinical trial (RCT) we assessed the safety and efficacy of a standardized intestinal microbiota infusion in SSc patients with lower GIT symptoms.
Methods: Patients with SSc and moderate to severe bloating and/or diarrhea assessed by the UCLA SCTC GIT score 2.0 were enrolled in a multicenter, double-blind, randomized, placebo-controlled phase 2 trial. Patients were randomized to receive FMT with an intestinal infusion of a standardized fecal microbiota culture (ACHIM) or placebo at weeks 0 and 2. At week 12, all patients received a FMT infusion and were followed in an open lable phase until week 20. The primary outcome was change between baseline and week 12 in UCLA GIT score item diarrhea or bloating measured as the average marginal effect (AME), depending on which was the worst symptom at baseline; evaluated separately for each patient. Comprehensive predefined subgroup analyses were conducted. Secondary outcomes were safety and tolerability and total UCLA GIT score. Other outcome measures included the change in UCLA GIT score from week 12 to week 20.
Results: A total of 65 patients were randomized to receive FMT or placebo. Baseline characteristics in the groups were comparable (Table). There was no significant difference in the change in lower GIT symptoms from week 0 to week 12 between the groups (AME=0.17 (-0.12, 0.47), p=0.25) and no significant differences in any of the predefined subgroup analyses (Figure 1A-C and 2). Similarly, no significant difference was observed in the total GIT score between the two groups (AME=0.09 (-0.04, 0.23), p=0.17). Furthermore, during the open lable period, there was no statistical difference in change between the ACHIM and the placebo group (AME= -0.04 (-0.32, 0.23), p=0.077) shown in Figure 1D. Participants treated with ACHIM, 16 (37%) and with placebo 19 (42%) experienced any side effects. These were in general mild and short-lasting, with abdominal pain as the most frequent side effect present in 5 (15%) in ACHIM and 2 (6%) in placebo. Time to resolved pain was 2days in both groups. One patient experienced an intramural perforation during gastroscopy and needed IV antibiotics but fully recovered.
Conclusion: We were unable to find indications that FMT improves lower GIT symptoms in SSc patients, but the treatment was found to be safe.

Table: Baseline characteristics of patients included in the randomized double-blimd placebo controlled ReSScue trial

Figure 1: Change in worst symptom GIT score between baseline and week 12. Panel A shows the marginal means of the adjusted changes in the worst symptom GIT score over 12 weeks together with their standard errors for ACHIM and placebo groups as well as the adjusted between-group difference estimated from the primary model. Panel B shows the box-plots of the predicted (adjusted) individual changes in worst symptom GIT score over 12 weeks for both treatment groups given by the primary model. Panel C shows the mean change from baseline to 12 weeks in the worst symptom GIT score. The bars indicate 95% confidence intervals. Panel D shows the mean change from baseline to 20 weeks in the worst symptom GIT score. The bars indicate 95% confidence intervals

Figure 2: Pre-specified subgroup analyses of the change in the worst symptom GIT score from baseline to week 12 (primary endpoint) based on baseline characteristics and concomitant medication.
A. Hoffmann-Vold: Arxx Therapeutics, 2, Boehringer-Ingelheim, 2, 5, 6, 12, Support for travel, Genentech, 2, Janssen, 2, 5, 6, Medscape, 2, 6, 12, Support for travel, Roche, 2, 6, 12, Support for travel; H. Fretheim: actelion, 5, bayer, 2, Boehringer-Ingelheim, 6, GlaxoSmithKlein(GSK), 5; I. Barua: None; M. Nordgård Carstens: None; H. Didriksen: None; V. Sarna: None; K. Lundin: Falk Pharma, 2, GSK, 2, Takeda, 2, 6, Topas, 2; O. Distler: 4P-Pharma, 2, 5, 6, AbbVie, 2, 5, 6, Acceleron, 2, 5, 6, Alcimed, 2, 5, 6, Altavant Sciences, 2, 5, 6, Amgen, 2, 5, 6, AnaMar, 2, 5, 6, Arxx, 2, 5, 6, AstraZeneca, 2, 5, 6, Bayer, 2, 5, 6, Blade Therapeutics, 2, 5, 6, Boehringer Ingelheim, 2, 5, 6, Citus AG, 12, Co-Founder, Corbus Pharmaceuticals, 2, 5, 6, CSL Behring, 2, 5, 6, Galapagos, 2, 5, 6, Galderma, 2, 5, 6, Glenmark, 2, 5, 6, Gossamer, 2, 5, 6, Horizon Therapeutics, 2, 5, 6, Janssen, 2, 5, 6, Kymera, 2, 5, 6, Lupin, 2, 5, 6, Medscape, 2, 5, 6, Miltenyi Biotec, 2, 5, 6, Mitsubishi Tanabe, 2, 5, 6, MSD, 2, 5, 6, Novartis, 2, 5, 6, Patent issued “mir-29 for the treatment of systemic sclerosis” (US8247389, EP2331143), 10, Prometheus Biosciences, 2, 5, 6, Redx Pharma, 2, 5, 6, Roivant, 2, 5, 6, Topadur, 2, 5, 6; D. Khanna: AbbVie, 12, DSMB, AstraZeneca, 2, Boehringer-Ingelheim, 2, Bristol-Myers Squibb, 2, 5, CSL Behring, 2, Genentech, 2, Horizon Therapeutics, 2, 5, Janssen, 2, 6, Pfizer, 5, Prometheus, 2; E. Volkmann: Boehringer-Ingelheim, 2, 5, 6, CSL Behring, 2, GlaxoSmithKline, 2, Horizon, 5, Prometheus, 5, Roche, 2; O. Midtvedt: None; T. Midtvedt: ACHIM, 8, 10, 11; A. Dhainaut: None; A. Halse: None; G. Bakland: UCB, 2; I. Olsen: None; M. Pesonen: None; O. Molberg: None.



