Poster Session A
Epidemiology, health policy and outcomes
Session: (0325–0344) Patient Outcomes, Preferences, & Attitudes Poster I
0338: Comparative Disease Burden in Patients with Rheumatoid Arthritis, Psoriatic Arthritis or Ankylosing Spondylitis: Data from COVAD Patient-reported E-survey
Sunday, November 12, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- DB
Diego Benavent, MD
Hospital Universitario de Bellvitge
Madrid, SpainDisclosure information not submitted.
Abstract Poster Presenter(s)
Diego Benavent1, Marco Fornaro2, Florenzo Iannone3, Lorenzo Cavagna4, Masataka Kuwana5, Naveen R6, Vishwesh Agarwal7, Jessica Day8, Mrudula Joshi9, Sreoshy Saha10, Kshitij Jagtap11, Wanruchada Katchamart12, Phonpen Akarawatcharangura Goo13, Binit Vaidya14, Tsvetelina Velikova15, Parikshit Sen16, Samuel Shinjo17, Ai Lyn Tan18, Nelly Ziade19, Marcin Milchert20, Abraham Edgar Gracia-Ramos21, Carlo Caballero22, Hector Chinoy23, Vikas Agarwal6, Rohit Aggarwal24, Latika Gupta25 and Vincenzo Venerito26, 1Hospital la Paz, Madrid, Spain, 2University of Bari, Grottaglie, Italy, 3Rheumatology Unit, Department of Precision and Regenerative Medicine and Ionian Area, University of Bari "Aldo Moro", Bari, Italy, 4Fondazione IRCCS Policlinico San Matteo, Pavia, Italy, 5Nippon Medical School Graduate School of Medicine, Tokyo, Japan, 6Sanjay Gandhi Postgraduate Institute of Medical Sciences (SGPGIMS), Lucknow, India, 7Mahatma Gandhi Missions Medical College, Lucknow, India, 8Walter and Eliza Hall Institute, Melbourne, Australia, 9Byramjee Jeejeebhoy Government Medical College and Sassoon General Hospitals, Pune, India, 10Mymensingh Medical College, Faridpur, Bangladesh, 11Seth Gordhandhas Sunderdas Medical College and King Edwards Memorial Hospital, Mumbai, India, 12Mahidol University, Bangkok, Thailand, 13Department of Medicine, Queen Savang Vadhana Memorial Hospital, Chonburi, Thailand, 14National Center for Rheumatic Diseases (NCRD), Ratopul, Kathmandu, Nepal., Kathmandu, India, 15Department of Clinical Immunology, Medical Faculty, University Hospital "Lozenetz", Sofia University St. Kliment Ohridski, Sofia, Bulgaria, 16Maulana Azad Medical College, 2-Bahadurshah Zafar Marg, New Delhi, Delhi-110002, India., Dalhi, India, 17Faculdade de Medicina FMUSP, Universidade de Sao Paulo, São Paulo, Brazil, 18University of Leeds, Leeds, United Kingdom, 19Saint-Joseph University, Beirut, Lebanon, 20Department of Internal Medicine, Rheumatology, Diabetology, Geriatrics and Clinical Immunology, Pomeranian Medical University in Szczecin, Szczecin, Poland, 21Department of Internal Medicine, General Hospital, National Medical Center "La Raza", Instituto Mexicano del Seguro Social, Av. Jacaranda S/N, Col. La Raza, Del. Azcapotzalco, C.P. 02990, Mexico City, Mexico, 22Universidad del Norte, Barranquilla, Colombia, 23The University of Manchester, Sale, United Kingdom, 24University of Pittsburgh, Pittsburgh, PA, 25Royal Wolverhampton Trust, Wolverhampton/University of Manchester, United Kingdom, 26Rheumatology Department, Università degli Studi di Bari, Bari, Italy
Background/Purpose: Rheumatoid arthritis (RA) and spondyloarthritis (SpA), including either Psoriatic Arthritis (PsA) and Ankylosing Spondylitis (AS), are some of the most commonly diagnosed autoimmune rheumatic diseases in rheumatologists' routine clinical practice. Both can lead to joint destruction and substantial disability. Understanding patients' health and functional status is crucial to provide personalized management strategies to optimize disease control and enhance the quality of life. We aimed to compare disease burden in patients with RA, PsA or AS by assessing Patient-Reported Outcome Measurement Information System (PROMIS) Physical Health, Global Mental Health, Physical Function and Fatigue 4a together with Visual Analogue Scale (VAS) Pain.
Methods: Data were obtained in the international COVID vaccination in autoimmune rheumatic diseases study second e-survey (COVAD study). Demographics, AIRD diagnosis, disease activity, PROMIS Global Physical health, PROMIS Global Mental Health, PROMIS Physical Function SF10 and PROMIS Fatigue 4a scores were extracted from the COVAD study database. For this study, we only included patients with self-reported RA or spondyloarthritis (either PsA or AS) undergoing active treatment with conventional synthetic disease-modifying anti-rheumatic drugs (DMARDs) and/or biologic DMARDs, who answered all the survey questions. Active disease was defined as the patient's perception of their disease as active in the four weeks before the first dose of COVID-19 vaccination. Analysis of Variance with Bartlett's and Tukey's test was used to compare continuous variables between groups.
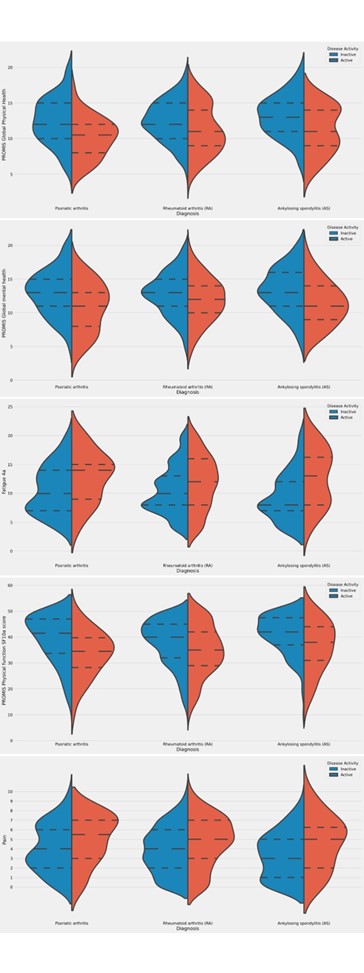
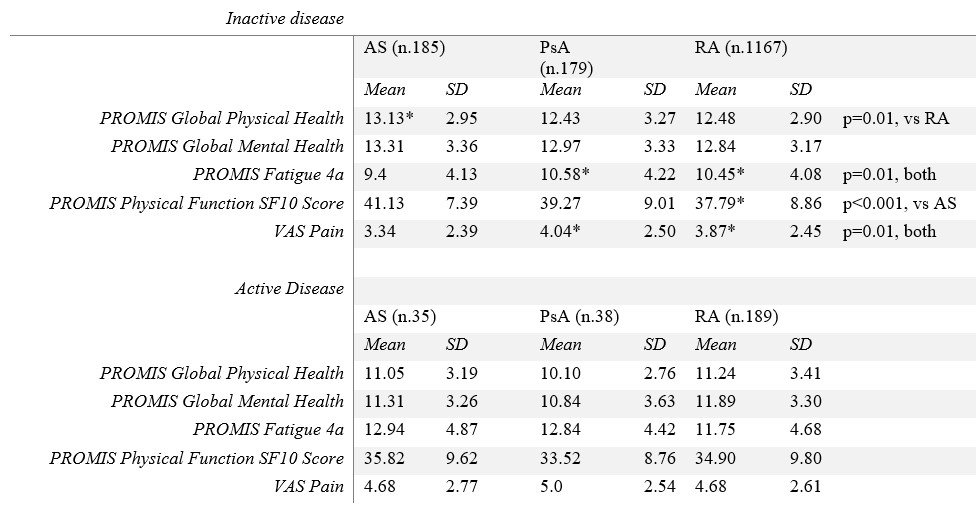
Results: From January to June 2022, 1907 patients with RA, female 87.62% (1671/1907), with mean age (±SD) 50.95 ± 13.67, 311 patients with PsA, female 67.20% (209/311), with a mean age of 50.42 ± 12.70, and 343 patients with AS, male 63.27% (217/343), with a mean age of 43.13 ± 12.75 years, responded to the COVAD e-survey. When assessed in those with active disease, neither physical health, global mental health, physical function, fatigue, nor pain were different among groups (Table 1, Figure 1). Patients with inactive AS had higher mean global physical health scores than RA patients (13.13 ± 2.93 VS RA 12.48 ± 2.90, p=0.01, Table 1). Nevertheless, those with inactive RA or PsA showed more severe fatigue (PsA 10.58 ± 2.22, RA 10.45 ± 4.08, vs AS 9.4 ± 4.13, p =0.01 for both). Patients with inactive RA also reported poorer physical function and more residual pain than those with AS (37.79 ± 8.86 VS 41.13 ± 7.79, p< 0.001; 3.87 ± 2.45 VS 3.34 ± 2.39, p=0.01, respectively). Similarly, residual pain was perceived as higher in patients with inactive PsA than those with AS (4.04 ± 2.50 VS 3.34 ± 2.39, p=0.01).
Conclusion: The burden of disease is approximately similar in patients with active RA, PsA or AS. However, patients with inactive RA and PsA report a considerably greater disease burden compared to those with AS.
D. Benavent: Abbvie, 5, Galapagos, 6, Janssen, 6, Novartis, 5, Roche, 6; M. Fornaro: None; F. Iannone: Abbvie, 2, 5, BMS, 2, 5, Janssen, 2, 5, Lilly, 2, 5, MSD, 2, 5, Novartis, 2, 5, Pfizer, 2, 5, Roche, 2, 5, UCB, 2, 5; L. Cavagna: None; M. Kuwana: AbbVie/Abbott, 6, Asahi-Kasei, 5, 6, Astellas, 6, AstraZeneca, 2, Boehringer-Ingelheim, 2, 5, 6, Chugai, 2, 5, 6, Corbus, 2, Eisai, 6, GlaxoSmithKlein(GSK), 2, Horizon, 2, Janssen, 6, Kissei, 2, MBL, 2, 5, Mitsubishi Tanabe, 2, 5, 6, Mochida, 2, 6, Nippon Shinyaku, 6, Ono, 5, 6; N. R: None; V. Agarwal: None; J. Day: CSL limited, 5; M. Joshi: None; S. Saha: None; K. Jagtap: None; W. Katchamart: None; P. Akarawatcharangura Goo: None; B. Vaidya: None; T. Velikova: AstraZeneca, 6, Pfizer, 6; P. Sen: None; S. Shinjo: None; A. Tan: Abbvie, 1, 6, Gilead, 6, Janssen, 6, Lilly, 6, Novartis, 6, Pfizer, 6, UCB, 6; N. Ziade: Abbvie, 6, Boehringer-Ingelheim, 6, Eli Lilly, 6, Janssen, 6, Newbridge, 6, Novartis, 6, Pfizer, 6, Pierre Fabre, 6, Roche, 6, sanofi, 6; M. Milchert: None; A. Gracia-Ramos: None; C. Caballero: None; H. Chinoy: AstraZeneca, 1, Biogen, 2, Eli Lilly, 5, GlaxoSmithKlein(GSK), 6, Novartis, 2, Orphazyme, 2, Pfizer, 1, UCB, 6; V. Agarwal: None; R. Aggarwal: Actigraph, 2, Alexion, 2, ANI Pharmaceuticals, 2, Argenx, 2, AstraZeneca, 2, Boehringer-Ingelheim, 2, 5, Bristol-Myers Squibb(BMS), 2, 5, CabalettaBio, 2, Capella Bioscience, 2, Corbus, 2, CSL Behring, 2, EMD Serono, 2, 5, Galapagos, 2, Horizon Therapeutics, 2, I-Cell, 2, Janssen, 2, 5, Kezar, 2, Kyverna, 2, Mallinckrodt, 5, Merck, 2, Octapharma, 2, Pfizer, 2, 5, Q32, 5, Roivant, 2, Sanofi, 2, Teva, 2; L. Gupta: None; V. Venerito: None.
Background/Purpose: Rheumatoid arthritis (RA) and spondyloarthritis (SpA), including either Psoriatic Arthritis (PsA) and Ankylosing Spondylitis (AS), are some of the most commonly diagnosed autoimmune rheumatic diseases in rheumatologists' routine clinical practice. Both can lead to joint destruction and substantial disability. Understanding patients' health and functional status is crucial to provide personalized management strategies to optimize disease control and enhance the quality of life. We aimed to compare disease burden in patients with RA, PsA or AS by assessing Patient-Reported Outcome Measurement Information System (PROMIS) Physical Health, Global Mental Health, Physical Function and Fatigue 4a together with Visual Analogue Scale (VAS) Pain.
Methods: Data were obtained in the international COVID vaccination in autoimmune rheumatic diseases study second e-survey (COVAD study). Demographics, AIRD diagnosis, disease activity, PROMIS Global Physical health, PROMIS Global Mental Health, PROMIS Physical Function SF10 and PROMIS Fatigue 4a scores were extracted from the COVAD study database. For this study, we only included patients with self-reported RA or spondyloarthritis (either PsA or AS) undergoing active treatment with conventional synthetic disease-modifying anti-rheumatic drugs (DMARDs) and/or biologic DMARDs, who answered all the survey questions. Active disease was defined as the patient's perception of their disease as active in the four weeks before the first dose of COVID-19 vaccination. Analysis of Variance with Bartlett's and Tukey's test was used to compare continuous variables between groups.
Results: From January to June 2022, 1907 patients with RA, female 87.62% (1671/1907), with mean age (±SD) 50.95 ± 13.67, 311 patients with PsA, female 67.20% (209/311), with a mean age of 50.42 ± 12.70, and 343 patients with AS, male 63.27% (217/343), with a mean age of 43.13 ± 12.75 years, responded to the COVAD e-survey. When assessed in those with active disease, neither physical health, global mental health, physical function, fatigue, nor pain were different among groups (Table 1, Figure 1). Patients with inactive AS had higher mean global physical health scores than RA patients (13.13 ± 2.93 VS RA 12.48 ± 2.90, p=0.01, Table 1). Nevertheless, those with inactive RA or PsA showed more severe fatigue (PsA 10.58 ± 2.22, RA 10.45 ± 4.08, vs AS 9.4 ± 4.13, p =0.01 for both). Patients with inactive RA also reported poorer physical function and more residual pain than those with AS (37.79 ± 8.86 VS 41.13 ± 7.79, p< 0.001; 3.87 ± 2.45 VS 3.34 ± 2.39, p=0.01, respectively). Similarly, residual pain was perceived as higher in patients with inactive PsA than those with AS (4.04 ± 2.50 VS 3.34 ± 2.39, p=0.01).
Conclusion: The burden of disease is approximately similar in patients with active RA, PsA or AS. However, patients with inactive RA and PsA report a considerably greater disease burden compared to those with AS.

Figure 1. Violin plots showing kernel densities, quartiles and median for Patient-Reported Outcome Measures for patients with RA, PsA and AS, stratified by disease activity status.

Table 1. Patient-Reported Outcome Measures between groups.
D. Benavent: Abbvie, 5, Galapagos, 6, Janssen, 6, Novartis, 5, Roche, 6; M. Fornaro: None; F. Iannone: Abbvie, 2, 5, BMS, 2, 5, Janssen, 2, 5, Lilly, 2, 5, MSD, 2, 5, Novartis, 2, 5, Pfizer, 2, 5, Roche, 2, 5, UCB, 2, 5; L. Cavagna: None; M. Kuwana: AbbVie/Abbott, 6, Asahi-Kasei, 5, 6, Astellas, 6, AstraZeneca, 2, Boehringer-Ingelheim, 2, 5, 6, Chugai, 2, 5, 6, Corbus, 2, Eisai, 6, GlaxoSmithKlein(GSK), 2, Horizon, 2, Janssen, 6, Kissei, 2, MBL, 2, 5, Mitsubishi Tanabe, 2, 5, 6, Mochida, 2, 6, Nippon Shinyaku, 6, Ono, 5, 6; N. R: None; V. Agarwal: None; J. Day: CSL limited, 5; M. Joshi: None; S. Saha: None; K. Jagtap: None; W. Katchamart: None; P. Akarawatcharangura Goo: None; B. Vaidya: None; T. Velikova: AstraZeneca, 6, Pfizer, 6; P. Sen: None; S. Shinjo: None; A. Tan: Abbvie, 1, 6, Gilead, 6, Janssen, 6, Lilly, 6, Novartis, 6, Pfizer, 6, UCB, 6; N. Ziade: Abbvie, 6, Boehringer-Ingelheim, 6, Eli Lilly, 6, Janssen, 6, Newbridge, 6, Novartis, 6, Pfizer, 6, Pierre Fabre, 6, Roche, 6, sanofi, 6; M. Milchert: None; A. Gracia-Ramos: None; C. Caballero: None; H. Chinoy: AstraZeneca, 1, Biogen, 2, Eli Lilly, 5, GlaxoSmithKlein(GSK), 6, Novartis, 2, Orphazyme, 2, Pfizer, 1, UCB, 6; V. Agarwal: None; R. Aggarwal: Actigraph, 2, Alexion, 2, ANI Pharmaceuticals, 2, Argenx, 2, AstraZeneca, 2, Boehringer-Ingelheim, 2, 5, Bristol-Myers Squibb(BMS), 2, 5, CabalettaBio, 2, Capella Bioscience, 2, Corbus, 2, CSL Behring, 2, EMD Serono, 2, 5, Galapagos, 2, Horizon Therapeutics, 2, I-Cell, 2, Janssen, 2, 5, Kezar, 2, Kyverna, 2, Mallinckrodt, 5, Merck, 2, Octapharma, 2, Pfizer, 2, 5, Q32, 5, Roivant, 2, Sanofi, 2, Teva, 2; L. Gupta: None; V. Venerito: None.



