Poster Session A
Rheumatoid arthritis (RA)
Session: (0380–0422) RA – Diagnosis, Manifestations, and Outcomes Poster I
0391: Biologic Use Regulates the Impact of Inflammation on Ischemic Cardiovascular Risk in Rheumatoid Arthritis
Sunday, November 12, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall

George Karpouzas, MD (he/him/his)
Harbor-UCLA Medical Center
Torrance, CA, United StatesDisclosure information not submitted.
Abstract Poster Presenter(s)
George Karpouzas1, Sarah Ormseth2, Piet Van Riel3, Elena Myasoedova4, Miguel A Gonzalez-Gay5, Alfonso Corrales6, Solbritt Rantapää-Dahlqvist7, Petros Sfikakis8, Patrick Dessein9, Linda Tsang9, Carol Hitchon10, Hani El Gabalawi10, Virginia Pascual Ramos11, Irazú Contreras Yañez12, Iris Colunga13, Dionicio A. Galarza-Delgado13, José Ramón Azpiri-López13, Silvia Rolefstad14, Anne Grete Semb15, Durga P Misra16, Ellen Margrethe Hauge17 and GEORGE KITAS18, 1Harbor-UCLA Medical Center, Torrance, CA, 2The Lundquist Institute, Torrance, CA, 3Radboud University Medical Center, Drunen, Netherlands, 4Mayo Clinic, Rochester, MN, 5IDIVAL and School of Medicine, UC, Santander; Department of Rheumatology, IIS-Fundación Jiménez Díaz, Madrid, Santander, Spain, 6Rheumatology Department, Immunopathology Group, Hospital Universitario Marqués de Valdecilla-IDIVAL, Santander, Spain, 7Department of Public Health and Clinical Medicine/Rheumatology, Umeå University, Umeå, Sweden, 8National Kapodistrian University of Athens Medical School, Athens, Greece, 9University of Witwatersrand, Johannesburg, South Africa, 10University of Manitoba, Winnipeg, MB, Canada, 11Instituto Nacional de Ciencias Medicas y Nutricion Salvador Zubiran, Mexico City, Mexico, 12Instituto Nacional de Ciencias Médicas y Nutrición Salvador Zubirán, Mexico City, Mexico, 13Hospital Universitario UANL, Monterrey, Mexico, 14Diakonhjemmet Hospital, Oslo, Norway,, Oslo, Norway, 15Preventive Cardio-Rheuma clinic, Dept Rheum, Diakonhjemmet Hospital, Oslo, Norway, 16Sanjay Gandhi Postgraduate Institute of Medical Sciences (SGPGIMS), Lucknow, India, 17Aarhus University Hospital, Aarhus, Denmark, 18The Dudley Group NHS Foundation Trust, Birmingham, United Kingdom
Background/Purpose: Chronic inflammation contributes to enhanced cardiovascular risk in rheumatoid arthritis (RA). Biologic disease modifying antirheumatic drugs (bDMARDs) control inflammation in many conventional synthetic DMARD non-responders and improve outcomes. We explored whether baseline bDMARD use may influence the impact of disease activity and systemic inflammation on long-term cardiovascular risk in RA.
Methods: We studied 4370 patients free of cardiovascular disease upon registration to An International Cardiovascular Consortium for people with RA (ATACC-RA) and followed prospectively. Outcomes included (a) major adverse cardiovascular events (MACE) defined as non-fatal myocardial infarction, non-fatal stroke, or cardiovascular death and (b) any ischemic cardiovascular events (CVE) comprising MACE, coronary revascularization, stable angina pectoris, transient ischemic attack and peripheral arterial disease with or without revascularization. Missing data were imputed using multiple imputation with 10 repetitions. Multivariable Cox models stratified by center evaluated the impact of disease activity (DAS28-CRP), inflammation (CRP), bDMARD use and their respective interactions on CVE risk after adjusting for age, gender, hypertension, diabetes, family history, smoking and total cholesterol to high-density lipoprotein ratio. Two corroborating sensitivity analyses were performed; the first included patients enrolled in the cohort on or after January 1, 2000, when bDMARD use became more prevalent. The second used inverse probability of treatment weights to balance differences in bDMARD treated and untreated patients.
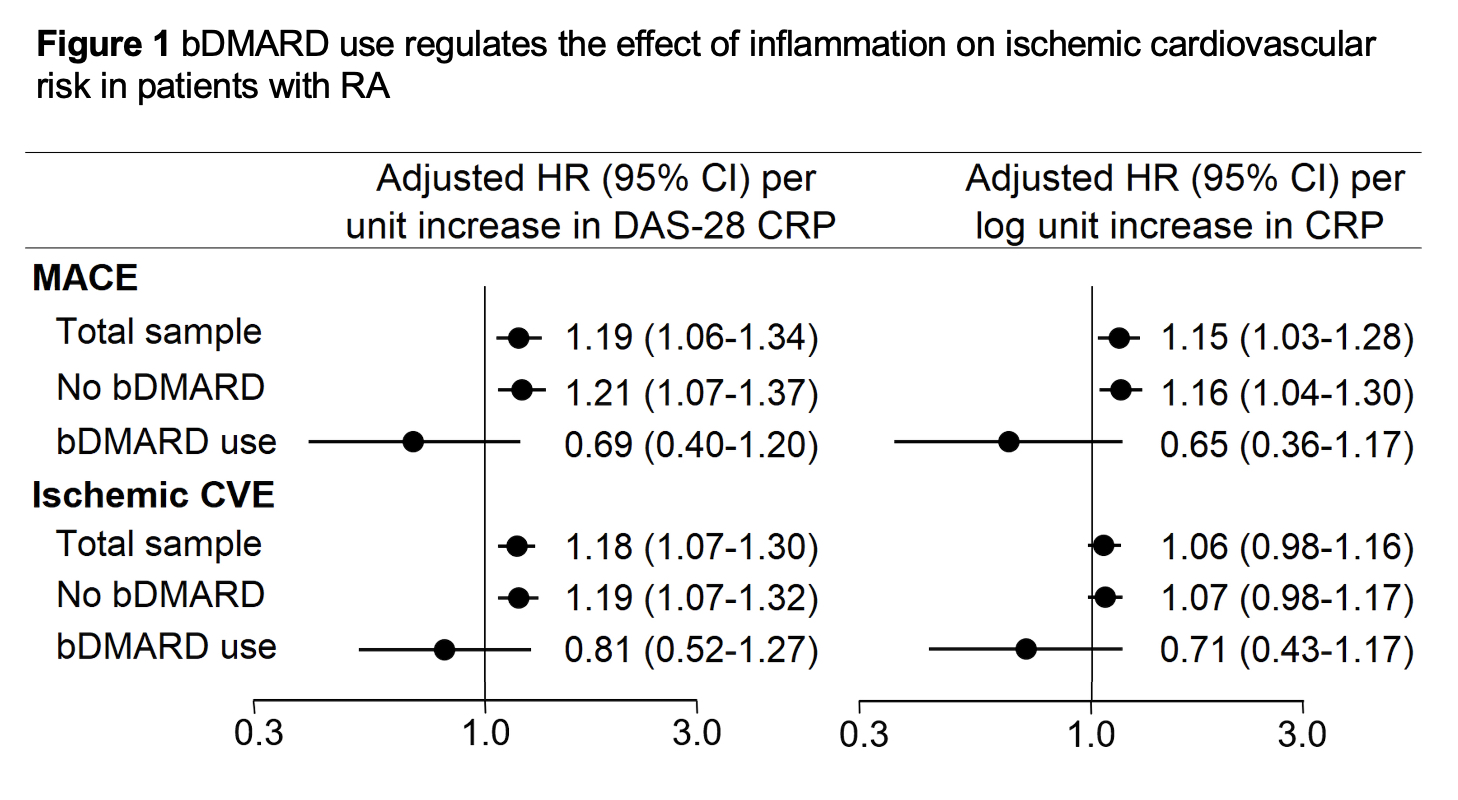
Results: Throughout 26,534 patient years, 239 first MACE and 362 total ischemic CVE were recorded. Among bDMARD nonusers, incidence of MACE and any ischemic CVE was 9.3 (95% CI 8.2-10.6) and 14.2 (12.8-15.8) events/1000PY. Rates for bDMARD users were [5.4 (95% CI 2.9-10.1) and 8.2 (5.0-13.6) events/1000PY. In the entire cohort, DAS-28 CRP and CRP(ln) associated with greater risk of MACE [(adjusted hazards ratio [aHR] 1.19 (95%CI 1.06-1.34), p=0.004 and HR 1.15 (1.02-1.28), p=0.017], while for all ischemic CVE the association was significant for DAS28-CRP [aHR 1.1 (95%CI 1.07-1.30)], but not CRP(ln) [aHR 1.06 (0.97-1.16)]. In bDMARD nonusers, higher DAS28-CRP and CRP(ln) associated with greater risk of MACE [aHR 1.21 (95%CI 1.07-1.37), p=0.002 and aHR 1.16 (1.04-1.30), p=0.009]. However, this was not the case in bDMARD users [p-for-interaction= 0.017 and 0.011 correspondingly, Figures 1 and 2]. In contrast, no significant interaction between DAS28-CRP or CRP and bDMARD use on any ischemic CVE risk was observed (p-for-interaction= 0.167 and 0.237 respectively). Both sensitivity analyses yielded similar results.
Conclusion: Conclusion: Higher disease activity and systemic inflammation at baseline associated with greater risk of MACE in bDMARD nonusers but not in users. This may suggest the presence of additional bDMARD-specific benefits directly on atherosclerotic plaque —such as plaque stabilization— above and beyond effects on systemic inflammation.
.jpg)
G. Karpouzas: Janssen, 1, Pfizer, 5, Scipher, 1; S. Ormseth: None; P. Van Riel: None; E. Myasoedova: None; M. Gonzalez-Gay: AbbVie/Abbott, 5, 6, Amgen, 5, 6, Pfizer, 5, 6; A. Corrales: None; S. Rantapää-Dahlqvist: None; P. Sfikakis: AbbVie/Abbott, 2, 5, Amgen, 2, 5, Boehringer-Ingelheim, 2, 5, Celgene, 2, 5, Eli Lilly, 2, 5, Janssen, 2, 5, Novartis, 2, 5, Pfizer, 2, 5; P. Dessein: None; L. Tsang: None; C. Hitchon: Astra Zeneca, 1, Pfizer, 5; H. El Gabalawi: None; V. Pascual Ramos: None; I. Contreras Yañez: None; I. Colunga: None; D. Galarza-Delgado: None; J. Azpiri-López: None; S. Rolefstad: None; A. Semb: None; D. Misra: None; E. Hauge: AbbVie/Abbott, 5, 6, Galapagos, 5, Novartis, 6, Novo Nordic Foundation, 5, 6, Sanofi, 6, Sobi, 6; G. KITAS: None.
Background/Purpose: Chronic inflammation contributes to enhanced cardiovascular risk in rheumatoid arthritis (RA). Biologic disease modifying antirheumatic drugs (bDMARDs) control inflammation in many conventional synthetic DMARD non-responders and improve outcomes. We explored whether baseline bDMARD use may influence the impact of disease activity and systemic inflammation on long-term cardiovascular risk in RA.
Methods: We studied 4370 patients free of cardiovascular disease upon registration to An International Cardiovascular Consortium for people with RA (ATACC-RA) and followed prospectively. Outcomes included (a) major adverse cardiovascular events (MACE) defined as non-fatal myocardial infarction, non-fatal stroke, or cardiovascular death and (b) any ischemic cardiovascular events (CVE) comprising MACE, coronary revascularization, stable angina pectoris, transient ischemic attack and peripheral arterial disease with or without revascularization. Missing data were imputed using multiple imputation with 10 repetitions. Multivariable Cox models stratified by center evaluated the impact of disease activity (DAS28-CRP), inflammation (CRP), bDMARD use and their respective interactions on CVE risk after adjusting for age, gender, hypertension, diabetes, family history, smoking and total cholesterol to high-density lipoprotein ratio. Two corroborating sensitivity analyses were performed; the first included patients enrolled in the cohort on or after January 1, 2000, when bDMARD use became more prevalent. The second used inverse probability of treatment weights to balance differences in bDMARD treated and untreated patients.
Results: Throughout 26,534 patient years, 239 first MACE and 362 total ischemic CVE were recorded. Among bDMARD nonusers, incidence of MACE and any ischemic CVE was 9.3 (95% CI 8.2-10.6) and 14.2 (12.8-15.8) events/1000PY. Rates for bDMARD users were [5.4 (95% CI 2.9-10.1) and 8.2 (5.0-13.6) events/1000PY. In the entire cohort, DAS-28 CRP and CRP(ln) associated with greater risk of MACE [(adjusted hazards ratio [aHR] 1.19 (95%CI 1.06-1.34), p=0.004 and HR 1.15 (1.02-1.28), p=0.017], while for all ischemic CVE the association was significant for DAS28-CRP [aHR 1.1 (95%CI 1.07-1.30)], but not CRP(ln) [aHR 1.06 (0.97-1.16)]. In bDMARD nonusers, higher DAS28-CRP and CRP(ln) associated with greater risk of MACE [aHR 1.21 (95%CI 1.07-1.37), p=0.002 and aHR 1.16 (1.04-1.30), p=0.009]. However, this was not the case in bDMARD users [p-for-interaction= 0.017 and 0.011 correspondingly, Figures 1 and 2]. In contrast, no significant interaction between DAS28-CRP or CRP and bDMARD use on any ischemic CVE risk was observed (p-for-interaction= 0.167 and 0.237 respectively). Both sensitivity analyses yielded similar results.
Conclusion: Conclusion: Higher disease activity and systemic inflammation at baseline associated with greater risk of MACE in bDMARD nonusers but not in users. This may suggest the presence of additional bDMARD-specific benefits directly on atherosclerotic plaque —such as plaque stabilization— above and beyond effects on systemic inflammation.

.jpg)
G. Karpouzas: Janssen, 1, Pfizer, 5, Scipher, 1; S. Ormseth: None; P. Van Riel: None; E. Myasoedova: None; M. Gonzalez-Gay: AbbVie/Abbott, 5, 6, Amgen, 5, 6, Pfizer, 5, 6; A. Corrales: None; S. Rantapää-Dahlqvist: None; P. Sfikakis: AbbVie/Abbott, 2, 5, Amgen, 2, 5, Boehringer-Ingelheim, 2, 5, Celgene, 2, 5, Eli Lilly, 2, 5, Janssen, 2, 5, Novartis, 2, 5, Pfizer, 2, 5; P. Dessein: None; L. Tsang: None; C. Hitchon: Astra Zeneca, 1, Pfizer, 5; H. El Gabalawi: None; V. Pascual Ramos: None; I. Contreras Yañez: None; I. Colunga: None; D. Galarza-Delgado: None; J. Azpiri-López: None; S. Rolefstad: None; A. Semb: None; D. Misra: None; E. Hauge: AbbVie/Abbott, 5, 6, Galapagos, 5, Novartis, 6, Novo Nordic Foundation, 5, 6, Sanofi, 6, Sobi, 6; G. KITAS: None.



