Abstract Session
Epidemiology, health policy and outcomes
Session: Abstracts: Patient Outcomes, Preferences, & Attitudes II: Patient Experience (2533–2538)
2537: Implementation of a Best Practice Advisory to Improve Infection Screening Prior to New Prescriptions of Biologics and Targeted Synthetic Drugs
Tuesday, November 14, 2023
5:00 PM - 5:10 PM PT
Location: Room 33A-C
- HB
Hailey Baker, MD, MS (she/her/hers)
Yale University School of Medicine, Yale New Haven Hospital
New Haven, CT, United StatesDisclosure information not submitted.
Presenting Author(s)
Hailey Baker1, Rebecca Fine1, Fenn Suter2, Heather Allore3, Betty Hsiao4, Vaidehi Chowdhary5, Elizabeth Lavelle6, Ping Chen6, Richard Hintz6, Lisa Suter7 and Abhijeet Danve8, 1Yale New Haven Hospital, New Haven, CT, 2Georgetown University, Washington, DC, 3Yale University, Yale School of Medicine, New Haven, CT, 4Yale-New Haven Medical Center, New Haven, CT, 5Yale University, New Haven, CT, 6Yale New Haven Health, New Haven, CT, 7Yale School of Medicine, New Haven, CT, 8Yale University School of Medicine, Glastonbury, CT
Background/Purpose: Biologic and targeted synthetic DMARDs (b/tsDMARDs) are widely used to treat patients with various autoimmune inflammatory diseases (ARD). Use of a b/tsDMARD in patients with pre-existing tuberculosis (TB), hepatitis B (HBV), or hepatitis C (HCV) can lead to significant morbidity and mortality. Many national and international societies recommend screening for one or all of these exposures prior to initiation of certain b/tsDMARDs. However, recommended timing intervals of screening are not clear and adherence to these recommendations varies widely. This quality improvement initiative focused on improving screening for TB, HBV, and HCV prior to new prescription of a b/tsDMARD using a best practice advisory (BPA) in the electronic health records (EHR).
Methods: Patients aged 18 years or older with at least one visit with a clinician (attending, fellow, or APRN) in the section of Rheumatology in the designated time frame were included. Upon a new prescription of a b/tsDMARD, clinicians were alerted via a BPA pop-up in the EHR about the last available results if any, for TB, HBV, and HCV, and allowed for quick ordering as appropriate. This BPA was implemented on December 1, 2020. Baseline screening proportions for TB (QuantiFERON), HBV, and HCV for patients prescribed a new b/tsDMARD from October 1, 2017 to November 30, 2020 (pre-BPA period) were compared with those of patients with prescriptions from December 1, 2020 to March 3, 2022 (post-BPA period).
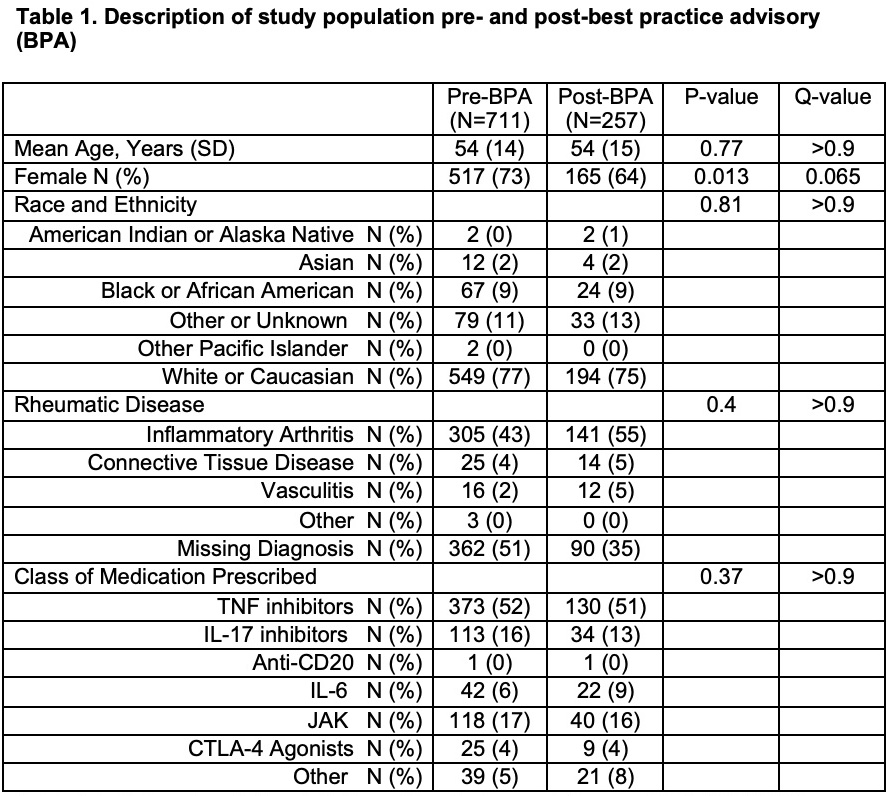
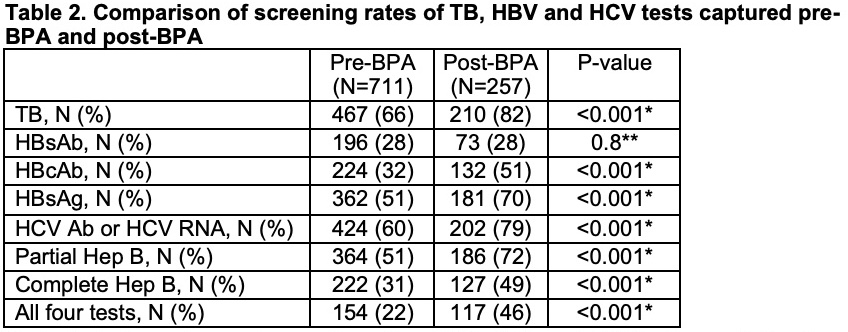
Results: A total of 711 patients pre-BPA and 257 patients post-BPA implementation were included in the study. The BPA implementation was associated with statistically significant improvement in screening for TB, HBsAg, HBcAb, and HCV Ab or HCV viral load (Table 1). Partial hepatitis B testing (screening for HBcAb or HBsAg) was significantly changed by the BPA (pre-BPA 364 of 711 (51%) vs post-BPA 186 of 257 (72%), p=< 0.001). Complete hepatitis B testing (screening for both HBcAb and HBsAg) also improved after BPA implementation (pre-BPA 222 of 711 (31%) vs post-BPA 127 of 257 (49%), p< 0.001). A total of 154 patients (22%) in the pre-BPA and 117 patients (46%) in the post-BPA periods were appropriately screened for all three infectious diseases, which was statistically significant (p< 0.001). Multivariable logistic analysis revealed in the pre-BPA period, relative to attending physicians, fellows were significantly more likely to order testing (HBcAb p=< 0.001, HBsAb p=< 0.001, HBsAg p=0.005, partial Hep B testing p=0.006, complete Hep B testing p=< 0.001, HCV Ab or HCV RNA p=0.002, all four tests p=< 0.001) after accounting for multiple comparisons. In the pre-BPA period, each year of age decreased the adjusted odds of TB testing by 2% (OR 0.98; 95% CI 0.97, 1.0; p=0.006) and males had lower adjusted odds for HBcAb testing than females (OR 0.58; 95% CI 0.39, 0.85). In the post-BPA period, there were no statistically significant findings for any patient or clinician variable.
Conclusion: Implementation of a BPA in the EHR can improve infectious disease screening for patients with ARD initiating b/tsDMARDs and has potential to improve patient safety and eliminate screening discrepancies.
.jpg)
H. Baker: None; R. Fine: None; F. Suter: None; H. Allore: NIH/NCATS, 5, NIH/NIA, 5, NIH/NIAID, 5; B. Hsiao: Rheumatology Research Foundation, 5; V. Chowdhary: ACR Guideline Committee (unpaid), 1, MCIC Vermont, 5; E. Lavelle: None; P. Chen: None; R. Hintz: None; L. Suter: Centers for Medicare Medicaid Services, 12, Salary support from unrelated CMS ontract, unrelated NIH grant to Elena Losina/BWH, 1; A. Danve: Abbvie, 2, Amgen, 2, Janssen, 2, Lilly, 5, Medscape, 6, Novartis, 2, 5, Spondylitis Association of America, 5, Spondyloarthritis Research and Treatment Network, 5, UCB, 1.
Background/Purpose: Biologic and targeted synthetic DMARDs (b/tsDMARDs) are widely used to treat patients with various autoimmune inflammatory diseases (ARD). Use of a b/tsDMARD in patients with pre-existing tuberculosis (TB), hepatitis B (HBV), or hepatitis C (HCV) can lead to significant morbidity and mortality. Many national and international societies recommend screening for one or all of these exposures prior to initiation of certain b/tsDMARDs. However, recommended timing intervals of screening are not clear and adherence to these recommendations varies widely. This quality improvement initiative focused on improving screening for TB, HBV, and HCV prior to new prescription of a b/tsDMARD using a best practice advisory (BPA) in the electronic health records (EHR).
Methods: Patients aged 18 years or older with at least one visit with a clinician (attending, fellow, or APRN) in the section of Rheumatology in the designated time frame were included. Upon a new prescription of a b/tsDMARD, clinicians were alerted via a BPA pop-up in the EHR about the last available results if any, for TB, HBV, and HCV, and allowed for quick ordering as appropriate. This BPA was implemented on December 1, 2020. Baseline screening proportions for TB (QuantiFERON), HBV, and HCV for patients prescribed a new b/tsDMARD from October 1, 2017 to November 30, 2020 (pre-BPA period) were compared with those of patients with prescriptions from December 1, 2020 to March 3, 2022 (post-BPA period).
Results: A total of 711 patients pre-BPA and 257 patients post-BPA implementation were included in the study. The BPA implementation was associated with statistically significant improvement in screening for TB, HBsAg, HBcAb, and HCV Ab or HCV viral load (Table 1). Partial hepatitis B testing (screening for HBcAb or HBsAg) was significantly changed by the BPA (pre-BPA 364 of 711 (51%) vs post-BPA 186 of 257 (72%), p=< 0.001). Complete hepatitis B testing (screening for both HBcAb and HBsAg) also improved after BPA implementation (pre-BPA 222 of 711 (31%) vs post-BPA 127 of 257 (49%), p< 0.001). A total of 154 patients (22%) in the pre-BPA and 117 patients (46%) in the post-BPA periods were appropriately screened for all three infectious diseases, which was statistically significant (p< 0.001). Multivariable logistic analysis revealed in the pre-BPA period, relative to attending physicians, fellows were significantly more likely to order testing (HBcAb p=< 0.001, HBsAb p=< 0.001, HBsAg p=0.005, partial Hep B testing p=0.006, complete Hep B testing p=< 0.001, HCV Ab or HCV RNA p=0.002, all four tests p=< 0.001) after accounting for multiple comparisons. In the pre-BPA period, each year of age decreased the adjusted odds of TB testing by 2% (OR 0.98; 95% CI 0.97, 1.0; p=0.006) and males had lower adjusted odds for HBcAb testing than females (OR 0.58; 95% CI 0.39, 0.85). In the post-BPA period, there were no statistically significant findings for any patient or clinician variable.
Conclusion: Implementation of a BPA in the EHR can improve infectious disease screening for patients with ARD initiating b/tsDMARDs and has potential to improve patient safety and eliminate screening discrepancies.
.jpg)
Figure 1 is a visualization of the finalized BPA after subsequent PDSA cycles identified areas of improvement in the clinical decision support system. This final version of the BPA includes seven lab result components at the top, five options for quick ordering of infectious disease screening, four lab components for general monitoring while on drug therapy (CBC, CMP, CRP, and ESR), and five acknowledgement reasons for why a clinician is or is not ordering the labs. TB= ”Tuberculosis”; HCV=”hepatitis C virus”; PCR= “polymerase chain reaction”; LTBI = “latent tuberculosis infection”; PTB = “pulmonary tuberculosis”; HEPBSAG= “Hepatitis B surface Antigen”; HEPBSAGQ= “Hepatitis B surface Antigen, Quest Diagnostics”; HEPBCAB=”Hepatitis B core Antibody”; LABHEP= represents multiple test components for Hepatitis C; HEPCAB=”Hepatitis C Antibody”; QUATBAU= ”TB QuantiFERON Gold”; QUANTGOLD1= ”TB QuantiFERON Gold”; QGOLD4TINCUB= ”TB QuantiFERON Gold”

Table 1 includes descriptive statistics of the study population pre-BPA (10/1/2017 – 11/30/2020) and post-BPA (12/1/2020 – 3/3/2022) implementation. A Bonferroni correction was used to correct for multiple comparisons and reduce risk of false positive results, which is represented by the Q-values. BPA=”best practice advisory”; TNF= “Tumor necrosis factor”; CTLA4= “cytotoxic T-lymphocyte associated protein 4”; IL-17= “Interleukin 17”; IL-6= “Interleukin 6”; JAK= “Janus kinase inhibitor”. Inflammatory arthritides included rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, and Adult-onset Still’s disease. Connective tissue diseases included were systemic lupus erythematosus, Sjogren’s syndrome, mixed connective tissue disease, and systemic sclerosis. Vasculitides included anti-neutrophil cytoplasmic autoantibody (ANCA)-associated vasculitis, Bechet’s disease, giant cell arteritis, and Takayasu arteritis. The “Other” category included diagnoses for polymyalgia rheumatica and Familial Mediterranean Fever.

Table 2 displays observed screening rates of TB, HBV, and HCV before and after the initiation of the BPA on 12/1/2020. Partial Hep B testing was defined as having either HBsAg or HBcAb, but not both. Complete Hep B testing was defined as having both HBsAg and HBcAb. To be considered as having testing of “All Four Tests” a patient had to have QuantiFERON Gold, HBsAg, HBcAb, and HCV Ab or HCV quantitative RNA performed within the specified time. We used a Bonferroni correction to correct for multiple comparisons and reduce risk of false positive results, which is represented by Q-values. BPA= “Best Practice Advisory”; TB= “Tuberculosis”; HBsAb= ”Hepatitis B surface antibody”; HBcAb= ”Hepatitis B core antibody”; HBsAg= ”Hepatitis B surface antigen”; HCV= ”Hepatitis C virus”; HBV= ”Hepatitis B virus.”
*Q-value=<0.001
**Q-value=>0.9
*Q-value=<0.001
**Q-value=>0.9
H. Baker: None; R. Fine: None; F. Suter: None; H. Allore: NIH/NCATS, 5, NIH/NIA, 5, NIH/NIAID, 5; B. Hsiao: Rheumatology Research Foundation, 5; V. Chowdhary: ACR Guideline Committee (unpaid), 1, MCIC Vermont, 5; E. Lavelle: None; P. Chen: None; R. Hintz: None; L. Suter: Centers for Medicare Medicaid Services, 12, Salary support from unrelated CMS ontract, unrelated NIH grant to Elena Losina/BWH, 1; A. Danve: Abbvie, 2, Amgen, 2, Janssen, 2, Lilly, 5, Medscape, 6, Novartis, 2, 5, Spondylitis Association of America, 5, Spondyloarthritis Research and Treatment Network, 5, UCB, 1.



