Abstract Session
Hematologic- and oncologic-associated rheumatic syndromes
Session: Abstracts: Immunological Complications of Medical Therapy (2509–2514)
2510: Autoantibody-positivity Before Treatment Is Associated with Immune-related Adverse Events in Melanoma Patients Treated with anti-PD-1
Tuesday, November 14, 2023
4:15 PM - 4:25 PM PT
Location: Room 25A-C
- Dv
Diane van der Woude, MD, PhD
Leiden University Medical Center
Leiden, NetherlandsDisclosure information not submitted.
Presenting Author(s)
Tineke van Wesemael1, Jessica Borgers2, Kyra Gelderman3, Els Verdegaal4, C.M. Korse5, M.J.P. Welters6, Ellen Kapiteijn7, W.J. van Houdt2, Sjoerd van der Burg6, René Toes1, J.V. Thienen2, John Haanen2 and Diane van der Woude1, 1Leiden University Medical Center, Leiden, Netherlands, 2Department of Medical Oncology, Netherlands Cancer Institute, Amsterdam, Netherlands, 3Sanquin Diagnostic Services, Amsterdam, Netherlands, 4Department of Medical Oncology, Oncode Institute, Leiden University Medical Center, Amsterdam, Netherlands, 5Department of Laboratory Medicine, Antoni Van Leeuwenhoek, The Netherlands Cancer Institute, Amsterdam, Netherlands, 6Department of Medical Oncology, Oncode Institute, Leiden University Medical Center, Leiden, Netherlands, 7Department of Medical Oncology, Leiden University Medical Center, Leiden, Netherlands
Background/Purpose: PD-1 immune checkpoint inhibitors are able to re-activate restrained anti-tumor T-cell responses in melanoma patients. However, PD-1 inhibition may also activate auto-reactive T-cells leading to immune-related adverse events (irAEs). Autoantibodies might also play a role in anti-PD-1 induced irAEs and could potentially be used to identify patients at risk of developing these irAEs. Therefore, we investigated the association between autoantibody-positivity and treatment toxicity and response in melanoma patients treated with anti-PD-1.
Methods: This multicenter, retrospective study included 143 advanced melanoma patients treated with anti-PD-1. Until six months after treatment initiation, toxicities grade ≥2 and recurrences/responses were captured. Autoantibody measurements, comprising IgM rheumatoid factor (RF), antinuclear antibodies (ANA), anti-cyclic citrullinated peptide antibodies (anti-CCP2), anti-thyroid peroxidase (anti-TPO) and anti-thyroglobulin (anti-Tg) were performed in an ISO15189 accredited laboratory at baseline and after three months of treatment.
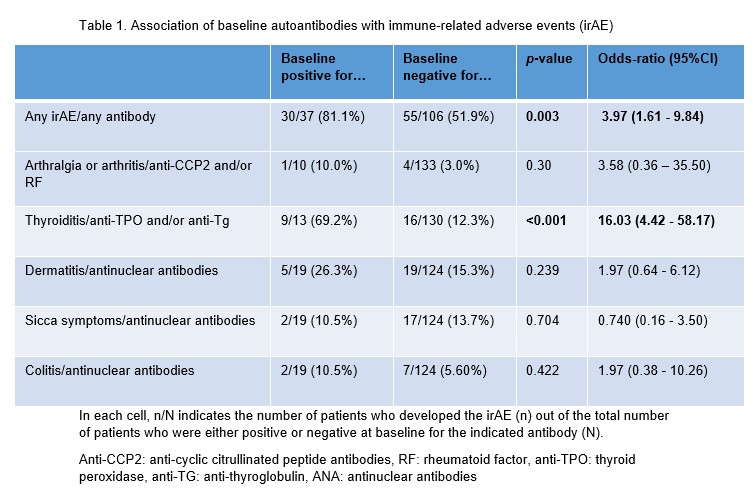
Results: A total of 169 irAEs were experienced by 86 patients, the most common being thyroiditis (n=25), dermatitis (n=24), and sicca complaints (n=19). More irAE (30/37 [81%]) were observed in patients with pre-existing detectable autoantibodies than in patients without these autoantibodies (55/106 [52%], p = 0.003) (table 1). This was mainly due to the strong association of anti-thyroid antibodies and thyroid dysfunction (69% in anti-TPO and/or anti-Tg positive patients versus 12% in anti-TPO and/or anti-Tg negative patients, p < 0.001). No association between anti-CCP2, RF or ANA and arthritis, dermatitis, sicca or colitis was observed. The association with thyroid antibodies was strongest in female patients: of seropositive female patients 7/7 (100%) developed thyroiditis, compared to only 10/48 (21%) of anti-TPO and anti-Tg negative patients.
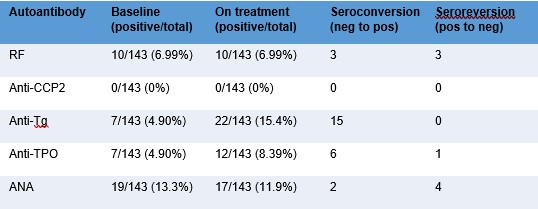
Seroconversion during anti-PD-1-treatment was only seen for thyroid antibodies. Here the combined positivity for anti-TPO and/or anti-Tg increased from 13/143 (9.1%) at baseline to 24/143 (17%) on treatment (p = 0.003). For the other autoantibodies the total number of seropositive patients stayed stable or decreased on treatment (table 2). Seroconversion of anti-Tg and/or anti-TPO was associated with thyroid dysfunction in female patients: 6/10 [60%] of seroconverted patients developed thyroid dysfunction compared to 3/38 [8%] patients that remained seronegative (p = 0.001). Autoantibody-positivity was not associated with disease recurrence (p = 0.45) or response (p = 0.69).
Conclusion: Autoantibody-positivity prior to anti-PD-1 therapy is associated with the development of irAEs in melanoma patients, indicating that anti-PD-1 therapy not only leads to loss of tolerance to tumor antigens but also to nontumor autoantigens. Baseline positivity and seroconversion of anti-thyroid antibodies were associated with the development of thyroiditis.
T. van Wesemael: None; J. Borgers: None; K. Gelderman: None; E. Verdegaal: None; C. Korse: None; M. Welters: None; E. Kapiteijn: None; W. van Houdt: None; S. van der Burg: None; R. Toes: Bristol-Myers Squibb(BMS), 5; J. Thienen: None; J. Haanen: None; D. van der Woude: None.
Background/Purpose: PD-1 immune checkpoint inhibitors are able to re-activate restrained anti-tumor T-cell responses in melanoma patients. However, PD-1 inhibition may also activate auto-reactive T-cells leading to immune-related adverse events (irAEs). Autoantibodies might also play a role in anti-PD-1 induced irAEs and could potentially be used to identify patients at risk of developing these irAEs. Therefore, we investigated the association between autoantibody-positivity and treatment toxicity and response in melanoma patients treated with anti-PD-1.
Methods: This multicenter, retrospective study included 143 advanced melanoma patients treated with anti-PD-1. Until six months after treatment initiation, toxicities grade ≥2 and recurrences/responses were captured. Autoantibody measurements, comprising IgM rheumatoid factor (RF), antinuclear antibodies (ANA), anti-cyclic citrullinated peptide antibodies (anti-CCP2), anti-thyroid peroxidase (anti-TPO) and anti-thyroglobulin (anti-Tg) were performed in an ISO15189 accredited laboratory at baseline and after three months of treatment.
Results: A total of 169 irAEs were experienced by 86 patients, the most common being thyroiditis (n=25), dermatitis (n=24), and sicca complaints (n=19). More irAE (30/37 [81%]) were observed in patients with pre-existing detectable autoantibodies than in patients without these autoantibodies (55/106 [52%], p = 0.003) (table 1). This was mainly due to the strong association of anti-thyroid antibodies and thyroid dysfunction (69% in anti-TPO and/or anti-Tg positive patients versus 12% in anti-TPO and/or anti-Tg negative patients, p < 0.001). No association between anti-CCP2, RF or ANA and arthritis, dermatitis, sicca or colitis was observed. The association with thyroid antibodies was strongest in female patients: of seropositive female patients 7/7 (100%) developed thyroiditis, compared to only 10/48 (21%) of anti-TPO and anti-Tg negative patients.
Seroconversion during anti-PD-1-treatment was only seen for thyroid antibodies. Here the combined positivity for anti-TPO and/or anti-Tg increased from 13/143 (9.1%) at baseline to 24/143 (17%) on treatment (p = 0.003). For the other autoantibodies the total number of seropositive patients stayed stable or decreased on treatment (table 2). Seroconversion of anti-Tg and/or anti-TPO was associated with thyroid dysfunction in female patients: 6/10 [60%] of seroconverted patients developed thyroid dysfunction compared to 3/38 [8%] patients that remained seronegative (p = 0.001). Autoantibody-positivity was not associated with disease recurrence (p = 0.45) or response (p = 0.69).
Conclusion: Autoantibody-positivity prior to anti-PD-1 therapy is associated with the development of irAEs in melanoma patients, indicating that anti-PD-1 therapy not only leads to loss of tolerance to tumor antigens but also to nontumor autoantigens. Baseline positivity and seroconversion of anti-thyroid antibodies were associated with the development of thyroiditis.

Table 1. Association of baseline autoantibodies with immune-related adverse events (irAE).
In each cell, n/N indicates the number of patients who developed the irAE (n) out of the total number of patients who were either positive or negative at baseline for the indicated antibody (N).
Anti-CCP2: anti-cyclic citrullinated peptide antibodies, RF: rheumatoid factor, anti-TPO: thyroid peroxidase, anti-TG: anti-thyroglobulin, ANA: antinuclear antibodies
In each cell, n/N indicates the number of patients who developed the irAE (n) out of the total number of patients who were either positive or negative at baseline for the indicated antibody (N).
Anti-CCP2: anti-cyclic citrullinated peptide antibodies, RF: rheumatoid factor, anti-TPO: thyroid peroxidase, anti-TG: anti-thyroglobulin, ANA: antinuclear antibodies

Table 2. Number of autoantibody-positive patients per antibody (RF, anti-CCP2, anti-TG, anti-TPO and ANA) at baseline and on treatment.
Anti-CCP2: anti-cyclic citrullinated peptide antibodies, RF: rheumatoid factor, anti-TPO: thyroid peroxidase, anti-TG: anti-thyroglobulin, ANA: antinuclear antibodies
Anti-CCP2: anti-cyclic citrullinated peptide antibodies, RF: rheumatoid factor, anti-TPO: thyroid peroxidase, anti-TG: anti-thyroglobulin, ANA: antinuclear antibodies
T. van Wesemael: None; J. Borgers: None; K. Gelderman: None; E. Verdegaal: None; C. Korse: None; M. Welters: None; E. Kapiteijn: None; W. van Houdt: None; S. van der Burg: None; R. Toes: Bristol-Myers Squibb(BMS), 5; J. Thienen: None; J. Haanen: None; D. van der Woude: None.



