Poster Session A
Systemic lupus erythematosus (SLE)
Session: (0543–0581) SLE – Diagnosis, Manifestations, & Outcomes Poster I
0548: Clinical and Interferon Biomarkers to Exclude Imminent Autoimmune Disease in ANA Positive Individuals
Sunday, November 12, 2023
9:00 AM - 11:00 AM PT
Location: Poster Hall
- MM
Md Yuzaiful Md Yusof, PhD, MBChB, MRCP (he/him/his)
University of Leeds
Leeds, United KingdomDisclosure information not submitted.
Abstract Poster Presenter(s)
Md Yuzaiful Md Yusof1, Sabih Ul-Hassan1, Zoe Wigston1, Antonios Psarras2, Jack Arnold1, Lucy Carter3, Paul Emery4 and Ed Vital1, 1University of Leeds, Leeds, United Kingdom, 2Oxford University, Oxford, United Kingdom, 3University of Leeds, Hartlepool, United Kingdom, 4Leeds Institute of Rheumatic and Musculoskeletal Medicine, University of Leeds, and NIHR Leeds Biomedical Research Centre, Leeds Teaching Hospitals NHS Trust, Leeds, United Kingdom
Background/Purpose: Many rheumatologists find it challenging to safely discharge "At-Risk" ANA-positive people at the first visit due to lack of cardinal signs and prediction tools. "Watch and wait" approach is not cost-effective and delay appropriate care of non-immune pathology. We previously showed that higher IFN-Score-B and family history were predictive of progression to meeting classification criteria at 12-months[1]. However, classification criteria may undergo revision and not capture all significant outcomes. Our study objectives were to describe the 3-year outcomes of At-Risk cohort and assess discriminative ability of baseline clinical and IFN-Score-B in predicting various progression endpoints at 1 and 3 years.
Methods: We conducted a prospective cohort study in At-Risk people (ANA-positive ≥1:80, new referral with non-specific symptoms of ≤1 year and treatment naïve). Patients were assessed at baseline, then annually for 3 years. We used multiple RMD classification criteria, including the revised 2019 EULAR/ACR for SLE, and need for therapy, to group patients as below: i) Absolute non-progressor (ANP) (no clinical criteria); ii) Undifferentiated CTD (U-CTD) (≥1 clinical criteria but not full RMD criteria). This group was subdivided into those requiring immunosuppressant (IS) excluding antimalarials only and those who did not; iii) Year 1 progressor (meeting criteria for RMD by 1 year); iv) Late progressor (meeting criteria for RMD in Years 2-3); v) Clinically significant disease (CSD) (progressor OR U-CTD on IS). Bloods were analysed for two IFN-stimulated gene expression scores previously described[2]. Discrimination of single or combined clinical and IFN-Score-B markers were assessed using ROC curve analyses.
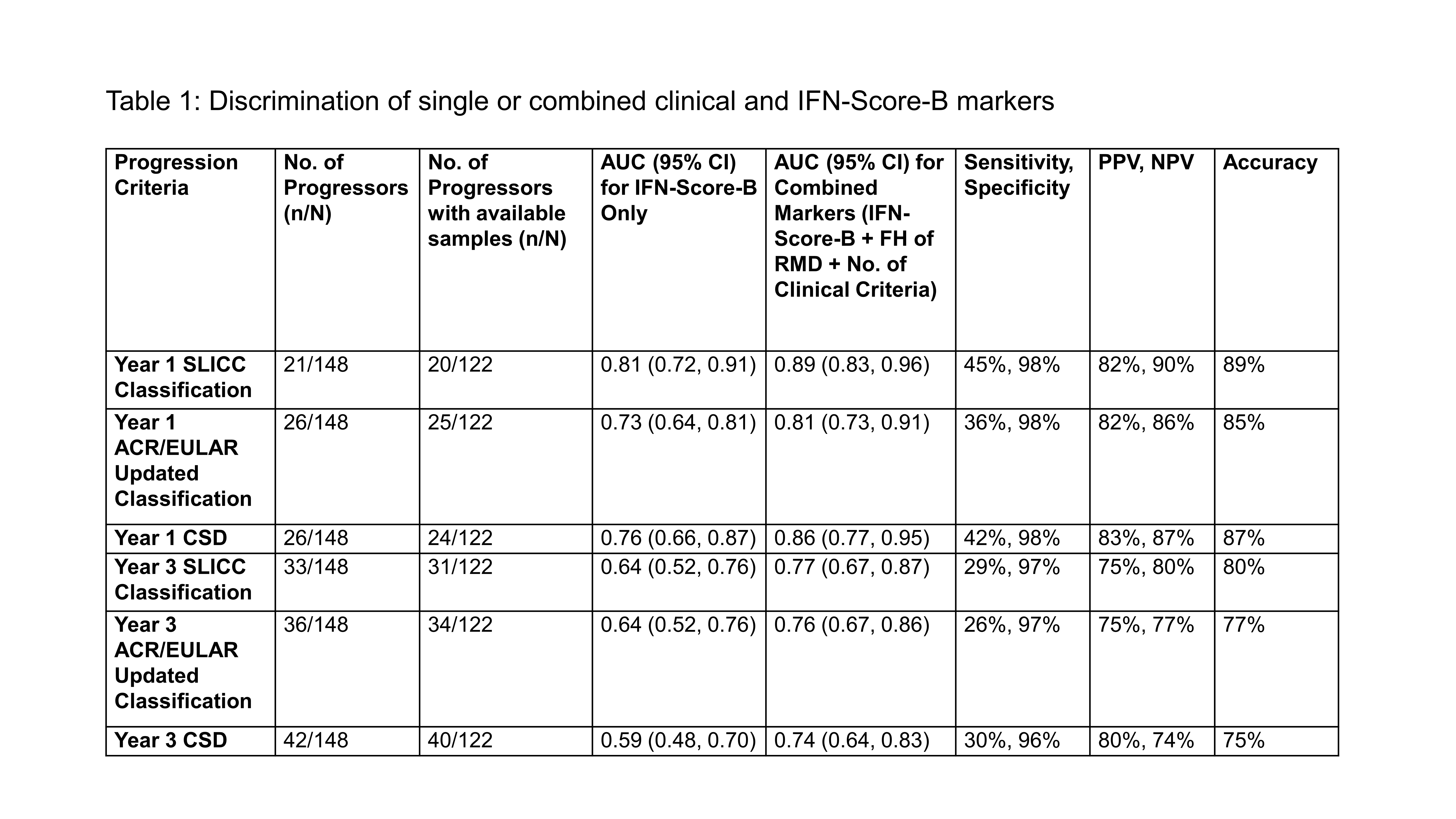
Results: Of 148 patients, mean (SD) age was 47 (15) years, 132 (89%) were female, 107 (72%) were Caucasians, 48 (32%) had a family history of RMD, 56 (38%) were anti-dsDNA+ and 8 (6%) had low C3 and/or C4. No. of clinical criteria at baseline were 0 (30%), 1 (64%) and 2 (6%). Outcomes were: Year 1 progressors: 21 (14%) [SLE=14; pSS=6; AS=1]; Late progressors: 12 (8%) [SLE=10; pSS=1; AS=1] of which only 2/12 in Year 3; U-CTD on IS: 8 (5%); U-CTD on antimalarials only: 20 (14%); U-CTD not on therapy: 50 (34%) and ANP: 37 (25%). Thus, 41 (28%) was classified as CSD by 3-year. For the prediction of Year 1 progressor, in addition to baseline IFN-Score-B and family history, multivariable regression showed no. of clinical criteria was associated with increased risk, OR 7.8 (95% CI 1.7-36.3). Table 1 showed that combined baseline markers (IFN-Score-B, family history and no. of clinical criteria) had good accuracy in predicting various definitions of progression at Years 1 and 3 as per AUROC.
Conclusion: About a quarter of At-Risk people developed CSD by 3-year, mostly did so within Year 1. Combined baseline clinical and IFN biomarkers had high specificity and could be used to risk stratify new ANA-positive referrals to rheumatology to exclude imminent or future disease/requirement of immunosuppressant. A validation study with cost-effectiveness analysis of these markers is in progress and would help translate their use in clinical practice.
References: 1. Md Yusof et al. ARD 2018, 2. El-Sherbiny et al. Sci Rep 2018
M. Md Yusof: Novartis, 6, Roche, 6, UCB, 1; S. Ul-Hassan: None; Z. Wigston: None; A. Psarras: None; J. Arnold: None; L. Carter: UCB, 1; P. Emery: Boehringer Ingelheim, 2, Eli Lilly, 2, Novartis, 2; E. Vital: F. Hoffmann-La Roche Ltd, 2, Genentech, Inc., 2, Sandoz, 5.
Background/Purpose: Many rheumatologists find it challenging to safely discharge "At-Risk" ANA-positive people at the first visit due to lack of cardinal signs and prediction tools. "Watch and wait" approach is not cost-effective and delay appropriate care of non-immune pathology. We previously showed that higher IFN-Score-B and family history were predictive of progression to meeting classification criteria at 12-months[1]. However, classification criteria may undergo revision and not capture all significant outcomes. Our study objectives were to describe the 3-year outcomes of At-Risk cohort and assess discriminative ability of baseline clinical and IFN-Score-B in predicting various progression endpoints at 1 and 3 years.
Methods: We conducted a prospective cohort study in At-Risk people (ANA-positive ≥1:80, new referral with non-specific symptoms of ≤1 year and treatment naïve). Patients were assessed at baseline, then annually for 3 years. We used multiple RMD classification criteria, including the revised 2019 EULAR/ACR for SLE, and need for therapy, to group patients as below: i) Absolute non-progressor (ANP) (no clinical criteria); ii) Undifferentiated CTD (U-CTD) (≥1 clinical criteria but not full RMD criteria). This group was subdivided into those requiring immunosuppressant (IS) excluding antimalarials only and those who did not; iii) Year 1 progressor (meeting criteria for RMD by 1 year); iv) Late progressor (meeting criteria for RMD in Years 2-3); v) Clinically significant disease (CSD) (progressor OR U-CTD on IS). Bloods were analysed for two IFN-stimulated gene expression scores previously described[2]. Discrimination of single or combined clinical and IFN-Score-B markers were assessed using ROC curve analyses.
Results: Of 148 patients, mean (SD) age was 47 (15) years, 132 (89%) were female, 107 (72%) were Caucasians, 48 (32%) had a family history of RMD, 56 (38%) were anti-dsDNA+ and 8 (6%) had low C3 and/or C4. No. of clinical criteria at baseline were 0 (30%), 1 (64%) and 2 (6%). Outcomes were: Year 1 progressors: 21 (14%) [SLE=14; pSS=6; AS=1]; Late progressors: 12 (8%) [SLE=10; pSS=1; AS=1] of which only 2/12 in Year 3; U-CTD on IS: 8 (5%); U-CTD on antimalarials only: 20 (14%); U-CTD not on therapy: 50 (34%) and ANP: 37 (25%). Thus, 41 (28%) was classified as CSD by 3-year. For the prediction of Year 1 progressor, in addition to baseline IFN-Score-B and family history, multivariable regression showed no. of clinical criteria was associated with increased risk, OR 7.8 (95% CI 1.7-36.3). Table 1 showed that combined baseline markers (IFN-Score-B, family history and no. of clinical criteria) had good accuracy in predicting various definitions of progression at Years 1 and 3 as per AUROC.
Conclusion: About a quarter of At-Risk people developed CSD by 3-year, mostly did so within Year 1. Combined baseline clinical and IFN biomarkers had high specificity and could be used to risk stratify new ANA-positive referrals to rheumatology to exclude imminent or future disease/requirement of immunosuppressant. A validation study with cost-effectiveness analysis of these markers is in progress and would help translate their use in clinical practice.
References: 1. Md Yusof et al. ARD 2018, 2. El-Sherbiny et al. Sci Rep 2018

M. Md Yusof: Novartis, 6, Roche, 6, UCB, 1; S. Ul-Hassan: None; Z. Wigston: None; A. Psarras: None; J. Arnold: None; L. Carter: UCB, 1; P. Emery: Boehringer Ingelheim, 2, Eli Lilly, 2, Novartis, 2; E. Vital: F. Hoffmann-La Roche Ltd, 2, Genentech, Inc., 2, Sandoz, 5.



